Last updated: April 25, 2026
Market dynamics and financial trajectory for PENICILLIN
How is penicillin priced and financed across markets?
Penicillin’s market structure is defined by three forces: (1) generation-level product lifecycle (old-generation versus newer biosynthetic/semi-synthetic supply chains), (2) antibiotic stewardship and hospital procurement rules, and (3) the procurement and tender discipline that governs hospital spending.
Price and cost drivers
- Input cost volatility: fermentation feedstocks, upstream enzyme supply, and contract-manufacturing rates shape gross margin more than brand economics.
- Form and route: injectable penicillins (hospital use) command higher administered cost but face tighter tendering; oral formulations are more exposed to payer and pharmacy channel competition.
- Regulatory label constraints: indications and safety restrictions limit sales velocity even when global demand rises.
- Competition from generics and therapeutically substitutable antibiotics: pricing compresses quickly once patent exclusivity ends.
Procurement-driven cash conversion
- Most penicillin revenue is tied to government and hospital tender cycles, producing uneven quarter-to-quarter billing but stable annual demand where stewardship policies permit use.
- Working capital is heavily affected by distributor inventory rules and tender payment terms. Hospital payables often dominate cash timing even when list prices are stable.
What are the demand mechanics for penicillin?
Penicillin demand is less about consumer pull and more about institutional use for bacterial infections and prophylaxis, with utilization constrained by resistance patterns and stewardship.
Demand-side mechanics
- Hospital and emergency utilization: highest in acute infection workflows, with prescribing concentrated in a narrower formulary when stewardship is active.
- Resistance and guideline adherence: penicillin use tightens when guidelines prioritize beta-lactamase resistant options for certain pathogen profiles.
- Seasonality and outbreak effects: localized spikes can occur during respiratory infection waves, but antibiotics-in-use programs can cap usage.
- Supply continuity: penicillin is sensitive to upstream fermentation disruptions; procurement planners adjust orders to maintain stock coverage targets.
How do regulations and stewardship change penicillin volume?
Antibiotic stewardship programs shift penicillin from broad use to more targeted prescribing, affecting both volume and product mix.
Policy impacts
- Formulary restriction: hospitals often require criteria-based approval for older antibiotics.
- Stewardship monitoring: prescribing audits reduce “empiric default” behavior and increase culture-guided prescribing.
- Reimbursement scrutiny: payer policies increasingly align payment with guideline-concordant therapy rather than broad antibiotic availability.
What does the competitive landscape look like?
Penicillin competes on cost and availability more than differentiation, with the market dominated by generics once intellectual property protections lapse.
Competitive dynamics
- Generic substitution: price competition intensifies post-exclusivity through automatic substitution and tender award criteria.
- Therapeutic substitution: even when penicillin is available, clinicians may choose alternatives if resistance risk is higher or if dosing convenience favors another beta-lactam class.
- Regional manufacturing footprint: supply chains concentrate in a limited number of producers, making regional pricing dependent on manufacturing capacity and export controls.
How does lifecycle maturity affect financial trajectory?
Penicillin is a historically first-wave antibiotic; its market behavior typically follows a mature, low-growth pattern once generics dominate. Financial trajectories tend to show stable revenue bases with margin compression.
Typical mature-drug trajectory
- Revenue: steady or modest decline long-term due to mix shift to newer alternatives, offset by ongoing demand for susceptible infections.
- Margin: structurally pressured by tender pricing and generic competition, with occasional spikes tied to raw material shortages or supply disruptions.
- Capex strategy: less innovation-led spending and more capacity maintenance and compliance-driven investments.
- R&D: limited for “new” penicillin unless tied to novel combinations, new delivery systems, or expanded indications with modern trials.
What metrics best map penicillin financial performance to market dynamics?
For penicillin, financial performance correlates more with supply reliability, procurement wins, and cost-of-goods than with marketing-driven growth.
Key monitoring indicators
- Hospital tender wins and awarded volumes (top-line stability)
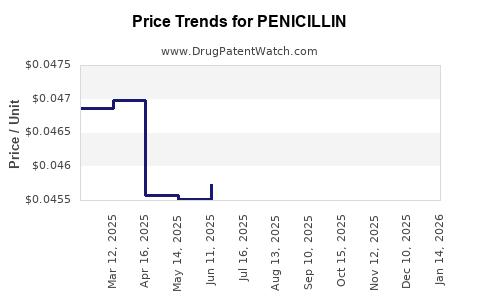
- Price per unit by route and strength (pricing power vs compression)
- Gross margin trend vs raw material and manufacturing rates (cost pressure)
- Inventory turns and receivables days (cash conversion under tender payment terms)
- Regulatory and supply disruptions (short-term inventory pricing and backorder revenue timing)
Where do financial outcomes come from: volume, price, or mix?
For mature antibiotics like penicillin, the split typically trends as follows:
- Volume: constrained by stewardship and substitution.
- Price: pressured by generic competition and tender procurement.
- Mix: improves revenue stability when manufacturers maintain coverage for injectable hospital products versus losing higher-margin formulations.
Net effect: revenue is more resilient than profitability, and profitability is more sensitive to supply chain cost swings than to demand.
What is the expected investment and business implication for R&D?
If R&D targets penicillin itself, the business case usually depends on one of two lanes:
- Differentiation through formulation or delivery to reduce dosing burden or improve stability (higher unit economics, if tender rules allow).
- Combination strategy to restore effectiveness against beta-lactamase mediated resistance or to align with modern guideline pathways (access to formulary despite substitution).
For standalone “new” penicillin molecules, the commercial upside is typically limited because the market is already saturated by generics and therapeutic alternatives.
Key Takeaways
- Penicillin’s market is governed by institutional procurement, stewardship constraints, and generic pricing discipline, not brand-driven demand.
- Financial trajectory is typically mature-drug steady revenue with margin compression, where cost-of-goods and supply continuity drive profitability more than growth.
- Competitive pressure comes from both generic substitution and therapeutic substitution, forcing economics to hinge on tenders and manufacturing reliability.
- R&D that can win formulary placement usually relies on formulation, delivery, or combination strategy rather than expecting blockbuster behavior from a legacy molecule.
FAQs
1) Is penicillin a growth market or a mature market?
It behaves as a mature market: revenue stability can persist, but long-run growth is constrained by stewardship, resistance-driven substitution, and generic competition.
2) What most strongly affects penicillin profit margins?
Manufacturing and upstream input costs, plus tender award pricing and inventory/working-capital timing.
3) How do hospital tender cycles influence financial reporting?
They can create lumpy quarterly billing and cash timing, driven by awarded volumes, shipment schedules, and payment terms tied to procurement cycles.
4) Why does antibiotic stewardship reduce penicillin utilization?
Stewardship programs tighten prescribing to guideline-concordant, often culture-guided treatment, reducing broad empiric use.
5) What R&D path is most commercially plausible for penicillin?
Projects tied to formulation/delivery improvements or combination strategies that support formulary access and clinical differentiation.
References
[1] World Health Organization. WHO Model List of Essential Medicines. World Health Organization. https://www.who.int/teams/health-product-and-policy-standards/medicines/essential-medicines
[2] U.S. Food and Drug Administration. Drug Development and Approval Process (general reference for regulation and labeling concepts). FDA. https://www.fda.gov/patients/drug-development-approval-process
[3] Centers for Disease Control and Prevention. Antibiotic Use and Stewardship (general stewardship framework). CDC. https://www.cdc.gov/antibiotic-use/
[4] European Medicines Agency. Guidelines and regulatory framework for medicines (general). EMA. https://www.ema.europa.eu/en/