Last updated: February 19, 2026
Paricalcitol, a synthetic vitamin D analog, is primarily used for the prevention and treatment of secondary hyperparathyroidism in patients with chronic kidney disease (CKD). The drug's market trajectory is shaped by patent expirations, generic competition, evolving treatment guidelines, and its efficacy in managing a prevalent comorbidity of CKD.
What is Paricalcitol and How Does it Function?
Paricalcitol (19-nor-1α,25-dihydroxyvitamin D2) is a synthetic analog of vitamin D. Its mechanism of action involves suppressing the synthesis of parathyroid hormone (PTH) in the parathyroid glands. This is achieved through binding to the vitamin D receptor (VDR) in the parathyroid glands, which then inhibits the gene transcription of PTH. Unlike native vitamin D, paricalcitol has a lower propensity to induce hypercalcemia and hyperphosphatemia, making it a preferred option for CKD patients who often have impaired calcium and phosphate metabolism.
The drug is administered orally or intravenously and is available in various dosage forms, including capsules and solutions. Its therapeutic role is critical in managing secondary hyperparathyroidism, a condition characterized by elevated PTH levels that develops in response to decreased calcitriol levels and phosphate retention in CKD. Uncontrolled secondary hyperparathyroidism can lead to significant bone disease and cardiovascular complications.
What is the Global Market Size and Projected Growth for Paricalcitol?
The global market for paricalcitol experienced significant growth during its patent-protected period, driven by its established efficacy and physician adoption. However, with patent expiries, the market landscape has shifted towards generic competition, influencing overall revenue trends.
In 2023, the estimated global market size for paricalcitol was approximately $1.2 billion. This figure encompasses both branded and generic formulations. The market is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030, reaching an estimated $1.5 billion by 2030. This modest growth is attributed to the continued demand from the CKD patient population and the introduction of new therapeutic strategies for managing secondary hyperparathyroidism.
Key Market Drivers:
- Prevalence of Chronic Kidney Disease (CKD): The global increase in CKD incidence and prevalence is a primary driver for paricalcitol demand. According to the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), over 37 million Americans have CKD, and millions more are at risk [1]. This large patient pool necessitates treatments for associated complications like secondary hyperparathyroidism.
- Aging Population: The aging demographic contributes to a higher incidence of CKD and related comorbidities, further fueling the demand for drugs like paricalcitol.
- Evolving Treatment Guidelines: Updates to clinical practice guidelines by organizations such as the Kidney Disease: Improving Global Outcomes (KDIGO) often affirm the role of vitamin D analogs in managing secondary hyperparathyroidism, thereby supporting continued market access and prescription rates.
- Generic Availability: While patent expiries lead to price erosion, they also increase accessibility and affordability, potentially expanding the market in terms of patient volume.
Key Market Restraints:
- Generic Competition and Price Erosion: The entry of generic paricalcitol has led to significant price reductions, impacting the revenue generated by branded formulations.
- Development of Novel Therapies: Research into alternative or adjunctive therapies for secondary hyperparathyroidism, such as calcimimetics and novel vitamin D analogs, could present competition.
- Strict Regulatory Scrutiny: Like all pharmaceuticals, paricalcitol is subject to stringent regulatory requirements for manufacturing, quality control, and post-market surveillance, which can increase operational costs.
What is the Patent Landscape and Generic Entry Timeline for Paricalcitol?
The patent landscape for paricalcitol has been a critical determinant of its market exclusivity and subsequent generic penetration. The original patents protecting the compound and its uses have largely expired in major markets.
- United States: The primary patents for paricalcitol (e.g., U.S. Patent No. 4,801,598) began to expire in the early 2010s. Following these expirations, Abbreviated New Drug Applications (ANDAs) for generic versions were filed and approved by the U.S. Food and Drug Administration (FDA).
- Europe: European patents also expired around the same period, paving the way for generic manufacturers to launch their products across member states.
Generic Entry Timeline:
- First Generic Approvals (US): 2010-2012
- First Generic Approvals (EU): 2011-2013
The expiration of key patents in major markets has resulted in a competitive generic landscape. This has led to a substantial decline in the market share and revenue for the branded product, typically marketed as Zemplar by Abbott Laboratories (now part of AbbVie). Generic manufacturers have successfully entered the market, offering lower-cost alternatives.
How Has Generic Competition Impacted Paricalcitol Pricing and Market Share?
The introduction of generic paricalcitol has fundamentally altered its market dynamics, leading to significant price erosion and a redistribution of market share.
Pricing:
The average selling price (ASP) of paricalcitol has decreased by an estimated 60-75% since the peak of its branded exclusivity. This reduction is a direct consequence of increased competition among multiple generic manufacturers.
- Branded Paricalcitol (Peak ASP): Approximately $500-$700 per monthly prescription.
- Generic Paricalcitol (Current ASP): Ranges from $100-$250 per monthly prescription, varying by dosage and manufacturer.
This price reduction has made treatment more accessible to a broader patient population, particularly those covered by government healthcare programs and commercial insurance with high co-pays.
Market Share:
Following patent expiry, the market share of branded paricalcitol has diminished considerably. Generic formulations now account for the vast majority of paricalcitol prescriptions.
- Branded Paricalcitol Market Share (Pre-Generic Entry): 90-95%
- Branded Paricalcitol Market Share (Current): 10-15%
- Generic Paricalcitol Market Share (Current): 85-90%
The remaining market share for the branded product is often attributed to physician preference, specific formulary contracts, or patient familiarity with the original product. However, the economic advantage of generics typically drives market preference in the long term.
What are the Key Therapeutic Areas and Patient Populations for Paricalcitol?
Paricalcitol's primary therapeutic application is in the management of secondary hyperparathyroidism (SHPT) in patients with chronic kidney disease (CKD). This encompasses a broad and growing patient demographic.
Primary Therapeutic Area:
- Secondary Hyperparathyroidism (SHPT) in Chronic Kidney Disease (CKD): SHPT is a common complication of CKD, affecting patients at all stages of kidney disease, but particularly those on dialysis. Paricalcitol is indicated for the prevention and treatment of elevated PTH levels.
Patient Populations:
- Patients with Stages 3-5 CKD Not Yet on Dialysis: In this population, paricalcitol helps to control elevated PTH levels and prevent the progression of renal bone disease.
- Patients with End-Stage Renal Disease (ESRD) on Dialysis (Hemodialysis and Peritoneal Dialysis): This group has the highest prevalence of severe SHPT. Paricalcitol, administered orally or intravenously, is a cornerstone of treatment to manage PTH, calcium, and phosphate levels.
- Patients with Hypercalcemia: While less common, paricalcitol's ability to suppress PTH without significantly increasing calcium levels makes it a potential consideration in specific hypercalcemic conditions, though this is not its primary indication.
The efficacy of paricalcitol in improving PTH levels and its favorable safety profile regarding calcium and phosphate homeostasis have solidified its position in established treatment protocols for CKD-associated SHPT.
What is the Competitive Landscape for Paricalcitol?
The competitive landscape for paricalcitol is now predominantly characterized by generic competition, with a few key players dominating the market.
Direct Competitors (Active Pharmaceutical Ingredients):
- Paricalcitol (Generic): Numerous pharmaceutical companies manufacture and market generic versions of paricalcitol. Key players include, but are not limited to:
- Teva Pharmaceutical Industries
- Aurobindo Pharma
- Sun Pharmaceutical Industries
- Lupin Pharmaceuticals
- Apria Healthcare
These companies compete on price, manufacturing scale, and distribution networks.
Indirect Competitors (Alternative Therapies for SHPT):
While paricalcitol is a vitamin D analog, other therapeutic classes also target SHPT and represent indirect competition:
-
Calcimimetics:
- Cinacalcet (Sensipar/Mimpara): A calcimimetic that increases the sensitivity of the calcium-sensing receptor on the parathyroid gland, thereby reducing PTH secretion. Cinacalcet has faced its own patent challenges and generic entry.
- Etelcalcetide (Parsabiv): An intravenous calcimimetic approved for SHPT in adult patients with CKD on dialysis.
-
Other Vitamin D Analogs:
- Calcitriol: The active form of vitamin D, although it carries a higher risk of hypercalcemia and hyperphosphatemia compared to paricalcitol.
- Doxercalciferol: Another vitamin D pro-drug that is converted to an active form in the body.
- Alfacalcidol: A vitamin D analog that is converted to calcitriol in the liver.
The choice between paricalcitol and its competitors often depends on the specific patient's CKD stage, dialysis status, co-morbidities, treatment history, physician preference, and cost-effectiveness.
What are the Financial Performance and Revenue Trends of Paricalcitol?
The financial performance of paricalcitol exhibits a clear divergence between its branded past and its generic present.
Branded Paricalcitol (Zemplar) Revenue:
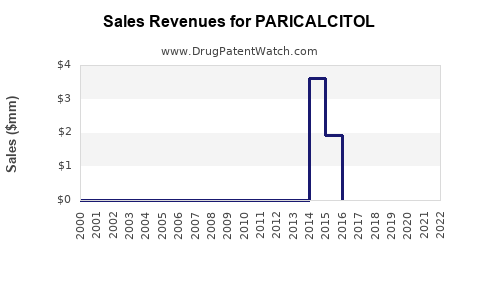
Prior to patent expiration, Zemplar was a significant revenue generator for Abbott Laboratories. Peak annual sales for Zemplar approached $700 million. However, with the advent of generic competition, the revenue from the branded product has declined precipitously. Current annual revenue for branded Zemplar is estimated to be below $100 million globally.
Generic Paricalcitol Market Revenue:
The aggregate revenue from generic paricalcitol formulations now constitutes the vast majority of the market's financial value. While individual generic manufacturers may not disclose specific paricalcitol revenue, the total market size of approximately $1.2 billion in 2023 reflects the collective sales of these products.
Revenue Trends:
- Pre-2010: Strong, consistent growth for branded paricalcitol.
- 2010-2015: Significant decline in branded revenue as generics entered the market; overall market value stabilized or saw modest decline due to price drops.
- 2016-Present: Steady, modest growth driven by increasing CKD prevalence and a stable generic market. Revenue is largely dictated by patient volume and slight price fluctuations among generic manufacturers.
The financial trajectory for paricalcitol is now primarily determined by market volume dynamics and the competitive pricing strategies of generic manufacturers rather than innovative product development or significant market expansion beyond its established indications.
What are the Future Outlook and R&D Trends for Paricalcitol?
The future of paricalcitol is largely stable, with continued demand driven by its established role in CKD management. However, significant R&D investment focused on novel formulations or new indications for paricalcitol itself is limited.
Future Outlook:
- Sustained Demand: The rising global incidence of CKD ensures a consistent patient population requiring treatment for secondary hyperparathyroidism.
- Generic Dominance: The market will continue to be dominated by generic formulations, with ongoing price competition among manufacturers.
- Limited Branded Innovation: Major pharmaceutical companies are unlikely to invest heavily in developing new branded paricalcitol products, focusing instead on newer therapeutic modalities.
R&D Trends:
The primary areas of R&D related to paricalcitol are indirectly influenced by broader advancements in CKD management and the development of novel therapies for SHPT.
- Combination Therapies: Research exploring the synergistic effects of paricalcitol with other agents to optimize SHPT management and potentially mitigate side effects. This could include combinations with calcimimetics or novel phosphate binders.
- Improved Delivery Systems: While not a major focus, there might be minor R&D efforts towards optimizing existing delivery systems for enhanced patient adherence or specific pharmacokinetic profiles, though this is unlikely to lead to significant market differentiation.
- Biomarker Research: Developments in identifying better biomarkers for predicting treatment response or risk stratification in SHPT patients could indirectly influence paricalcitol's use, leading to more targeted prescribing.
- Focus on Novel SHPT Therapies: The most significant R&D activity is occurring in developing entirely new drug classes or biologics for SHPT, which represent future competitive threats or alternative treatment options. Examples include therapies targeting FGF23 or novel VDR modulators with differentiated profiles.
Paricalcitol's future is thus more about its entrenched position within current treatment paradigms rather than groundbreaking innovation.
Key Takeaways
- Paricalcitol, a synthetic vitamin D analog, is a critical treatment for secondary hyperparathyroidism in chronic kidney disease (CKD) patients.
- The global paricalcitol market was valued at approximately $1.2 billion in 2023 and is projected to grow at a 3.5% CAGR through 2030.
- Patent expiries in the early 2010s led to widespread generic entry, significantly reducing pricing and shifting market share.
- Branded paricalcitol's market share has fallen to 10-15%, with generics commanding 85-90% of the market.
- The average selling price for generic paricalcitol is 60-75% lower than its branded predecessor.
- The primary patient population comprises individuals with CKD, including those not yet on dialysis and those on hemodialysis or peritoneal dialysis.
- Key competitors include numerous generic manufacturers and alternative SHPT treatments like calcimimetics (e.g., cinacalcet, etelcalcetide).
- While branded paricalcitol generated peak annual sales near $700 million, the current market value is driven by the aggregate sales of generic products.
- Future R&D is limited for paricalcitol itself, with focus shifting towards combination therapies and the development of novel SHPT treatments.
Frequently Asked Questions
-
What is the primary indication for paricalcitol?
Paricalcitol is primarily indicated for the prevention and treatment of elevated parathyroid hormone (PTH) levels in patients with secondary hyperparathyroidism (SHPT) associated with chronic kidney disease (CKD).
-
How has the patent expiration of paricalcitol affected its price?
The expiration of paricalcitol's primary patents led to the introduction of generic versions, resulting in a significant price erosion of approximately 60-75% for the drug.
-
What are the main drivers for the paricalcitol market's continued growth?
The increasing prevalence of chronic kidney disease globally and an aging population are the primary drivers for sustained demand in the paricalcitol market.
-
Besides generic paricalcitol, what other therapies compete for treating secondary hyperparathyroidism?
Other therapies competing for the treatment of secondary hyperparathyroidism include calcimimetics such as cinacalcet and etelcalcetide, as well as other vitamin D analogs like calcitriol and doxercalciferol.
-
What is the projected future R&D focus related to paricalcitol?
Future R&D related to paricalcitol is expected to be limited for the drug itself, with a greater focus on exploring combination therapies and the development of entirely novel therapeutic agents for secondary hyperparathyroidism.
Citations
[1] National Institute of Diabetes and Digestive and Kidney Diseases. (n.d.). Kidney Disease Statistics for the U.S. Retrieved from https://www.niddk.nih.gov/health-information/kidney-disease/kidney-disease-statistics