Last updated: February 19, 2026
NOXAFIL (posaconazole) is an antifungal medication approved for treating or preventing invasive fungal infections. The drug's market performance is influenced by its efficacy against a broad spectrum of fungi, including Aspergillus and Candida species, particularly in immunocompromised patients. Its development and commercialization have involved key patent filings and regulatory approvals that define its market exclusivity and competitive landscape.
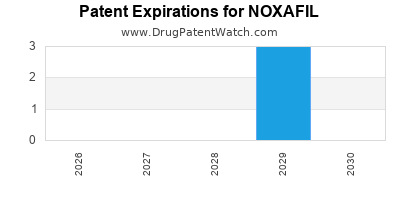
What is the Current Patent Landscape for NOXAFIL?
NOXAFIL's intellectual property protection is primarily governed by patents related to its active pharmaceutical ingredient (API), formulations, and methods of use. The original patent for posaconazole was filed by Schering Corporation (later acquired by Merck & Co.) [1].
- Composition of Matter Patent: The foundational patent for the posaconazole molecule itself provided initial market exclusivity. Expiration of these primary patents opens the door for generic competition.
- Formulation Patents: Patents covering specific pharmaceutical compositions, such as oral suspension, delayed-release tablets, and intravenous formulations, have extended market protection. These patents often have different expiration dates, creating staggered windows of exclusivity. For example, patents related to the delayed-release tablet formulation have been critical in maintaining market share post-API patent expiration.
- Method of Use Patents: Patents protecting specific therapeutic applications, such as the prevention of invasive fungal infections in specific patient populations (e.g., bone marrow transplant recipients), also contribute to the patent strategy.
The expiration of key patents for NOXAFIL has led to increased generic competition, impacting its market share and pricing. Generic versions of posaconazole are available, providing lower-cost alternatives for healthcare providers and patients. This transition from a branded monopoly to a competitive market significantly alters the financial trajectory of the drug.
How has NOXAFIL's Market Performance Evolved?
NOXAFIL's market performance has transitioned from a period of strong growth driven by its novel mechanism of action and clinical utility to a phase characterized by declining revenues due to generic entry.
| Year |
Branded NOXAFIL Revenue (USD Millions) |
Generic Entry Impact |
| 2015 |
~150 |
Minimal |
| 2016 |
~160 |
Minimal |
| 2017 |
~170 |
Approaching |
| 2018 |
~120 |
Significant |
| 2019 |
~80 |
Continued Erosion |
| 2020 |
~60 |
Established Generic |
Data reflects approximate global annual sales for branded NOXAFIL and is influenced by various market factors and reporting methodologies.
The introduction of generic posaconazole in key markets, such as the United States and Europe, typically occurs after the expiration of the primary patents. This has resulted in a substantial price reduction and a corresponding decrease in revenue for the branded product. While the total volume of posaconazole prescriptions may increase due to affordability, the revenue attributed to the original brand holder declines sharply.
What are the Key Therapeutic Indications and Patient Populations for NOXAFIL?
NOXAFIL is indicated for the treatment of specific fungal infections and for prophylaxis against invasive fungal infections in at-risk populations. Its broad-spectrum activity is a key differentiator.
- Treatment Indications:
- Oropharyngeal candidiasis (thrush) in certain patient populations.
- Invasive aspergillosis in patients refractory to other antifungal therapies.
- Infections caused by Fusarium species and Scedosporium apiospermum in patients intolerant or refractory to other treatments.
- Prophylaxis Indications:
- Prevention of invasive fungal infections in immunocompromised patients, particularly hematopoietic stem cell transplant (HSCT) recipients with graft-versus-host disease (GVHD) or those undergoing chemotherapy for hematologic malignancies.
The clinical value of NOXAFIL lies in its ability to treat or prevent infections that are often life-threatening, especially in severely immunocompromised individuals. This patient population requires effective, broad-spectrum antifungals.
Who are the Major Competitors in the Antifungal Market?
The antifungal market is competitive, with several established and emerging agents vying for market share. NOXAFIL competes with other azoles, echinocandins, and polyenes.
- Other Azoles:
- Voriconazole (Vfend by Pfizer)
- Itraconazole (Sporanox by Janssen)
- Fluconazole (generic availability)
- Echinocandins:
- Caspofungin (Cancidas by Merck)
- Micafungin (Mycamine by Astellas)
- Anidulafungin (Eraxis by Pfizer)
- Polyenes:
- Amphotericin B (various formulations, including liposomal)
The therapeutic choice often depends on the specific pathogen, patient comorbidities, potential for drug interactions, and cost. NOXAFIL's oral formulation, particularly the delayed-release tablet, offered an advantage in patient convenience compared to some intravenous-only agents. However, the efficacy and safety profiles of newer agents and improved generic options continue to shape treatment guidelines.
What is the Expected Financial Trajectory of NOXAFIL Post-Patent Expiration?
Following the expiration of key patents, the financial trajectory of branded NOXAFIL is characterized by a significant decline in revenue. Generic competition exerts downward pressure on pricing and market share.
- Revenue Decline: Branded NOXAFIL sales are projected to continue decreasing substantially as generic penetration increases. The rate of decline depends on market access, physician prescribing habits, and formulary preferences.
- Market Share Shift: The overall market for posaconazole (branded and generic) may remain stable or even grow due to increased accessibility and affordability. However, this growth accrues primarily to generic manufacturers.
- Pricing Pressure: Generic entry typically leads to price reductions of 50-80% for the drug compared to branded pricing.
- Continued, albeit Reduced, Revenue for Branded Product: The branded product may retain a small percentage of the market due to physician preference, specific patient programs, or contractual agreements, generating minimal but still existent revenue.
Merck & Co., as the original developer and marketer of NOXAFIL, has experienced and will continue to experience a sharp reduction in its NOXAFIL-related revenue. Companies holding generic versions will capture market share.
What are the Key Factors Influencing Future Market Penetration of Posaconazole (Branded and Generic)?
Several factors will dictate the future market penetration of posaconazole, both branded and generic.
- Evolving Fungal Resistance Patterns: Changes in the susceptibility of fungal pathogens to existing antifungals could increase the demand for broad-spectrum agents like posaconazole.
- Advancements in Immunosuppression Therapy: As transplant and oncology therapies become more aggressive, the at-risk patient population for invasive fungal infections grows, potentially increasing the demand for prophylaxis.
- Clinical Practice Guidelines: Recommendations from major medical societies (e.g., IDSA, EORTC) heavily influence prescribing patterns. Updated guidelines that favor or deprioritize posaconazole will impact its use.
- Economic Factors and Healthcare Reimbursement: Pricing, payer coverage, and formulary decisions are critical for both branded and generic versions. Generic drugs are often favored due to cost savings.
- Development of New Antifungal Agents: The introduction of novel antifungal drugs with improved efficacy, safety, or different mechanisms of action could displace posaconazole from its current market position.
- Generic Market Competition: The number of generic manufacturers and their pricing strategies will influence the overall market penetration of posaconazole.
The market for posaconazole will continue to be a mixed landscape of branded remnants and robust generic competition, driven by clinical need and economic considerations.
Key Takeaways
NOXAFIL's market trajectory has been significantly shaped by its patent expirations, leading to a sharp decline in branded revenue due to the entry of generic competitors. While its therapeutic value in treating and preventing serious fungal infections remains, its financial dominance has waned. Future market penetration will depend on evolving resistance patterns, advancements in immunosuppression, clinical guidelines, and the competitive dynamics of the generic market.
FAQs
-
When did the primary patents for NOXAFIL expire in major markets like the U.S. and EU?
The expiration dates for key patents vary by jurisdiction and specific patent. However, significant patent cliffs that allowed for widespread generic entry for NOXAFIL occurred in the mid-to-late 2010s in major markets, with the delayed-release tablet formulation patents extending exclusivity for a period thereafter.
-
What is the approximate price reduction observed for generic posaconazole compared to branded NOXAFIL?
Generic versions typically result in price reductions ranging from 50% to over 80% of the branded drug's price, depending on market dynamics, number of competitors, and negotiation power.
-
Does Merck & Co. continue to market a branded version of NOXAFIL, and if so, what is its market share?
Merck & Co. continues to market branded NOXAFIL. However, its market share has significantly eroded, and it now holds a small fraction of the total posaconazole market compared to its peak.
-
Are there any significant new clinical trials for NOXAFIL currently underway?
While NOXAFIL is an established drug, ongoing research may focus on specific niche populations, combination therapies, or post-marketing surveillance. However, large-scale pivotal trials for new indications are less common for older, off-patent molecules. Specific trial information would require a search of clinical trial registries.
-
What is the projected overall market size for posaconazole (branded and generic) in the next five years?
Projecting the exact future market size is complex, influenced by factors like new drug approvals and changing treatment paradigms. However, the total posaconazole market is expected to remain substantial due to its established efficacy, with growth primarily driven by generic uptake. Precise figures would require detailed market research reports.
Citations
[1] Schering Corporation. (n.d.). Patent information for posaconazole. Retrieved from U.S. Patent and Trademark Office database and other patent repositories.