Last updated: February 19, 2026
Mirapex (pramipexole dihydrochloride monohydrate) is a dopamine agonist primarily used for the treatment of Parkinson’s disease (PD) and restless legs syndrome (RLS). The drug's market performance is influenced by patent expirations, generic competition, and ongoing clinical research.
What is the Current Market Status of Mirapex?
Mirapex is available in both immediate-release (IR) and extended-release (ER) formulations. The IR formulation was approved by the U.S. Food and Drug Administration (FDA) in 1997 for Parkinson's disease, and the RLS indication followed in 2006. The ER formulation received FDA approval in 2009 for Parkinson's disease [1].
The primary U.S. patent for Mirapex (U.S. Patent No. 4,912,104) expired in 2006. This led to the introduction of generic versions of the immediate-release formulation shortly thereafter. The extended-release formulation's patent protection was more robust, with key patents expiring in the mid-2010s [2].
As of 2023, Mirapex faces significant generic competition across both its IR and ER formulations. This competition has resulted in price erosion and a substantial decline in branded Mirapex sales. The U.S. market for pramipexole is now largely dominated by generic manufacturers.
What are the Key Indications for Mirapex?
Mirapex is indicated for two main conditions:
- Parkinson's Disease: It is used to treat the signs and symptoms of idiopathic Parkinson's disease. Mirapex works by stimulating dopamine receptors in the brain, which helps to alleviate motor symptoms such as rigidity, tremor, and slowness of movement [3].
- Restless Legs Syndrome (RLS): It is used to treat moderate-to-severe primary RLS. The exact mechanism by which Mirapex improves RLS symptoms is not fully understood but is believed to involve dopaminergic pathways [4].
What is the Patent Landscape for Mirapex?
The patent landscape for Mirapex has evolved significantly since its initial approval.
- U.S. Patent No. 4,912,104 (Composition of Matter): This fundamental patent expired in 2006, allowing for the entry of generic immediate-release pramipexole.
- U.S. Patent No. 6,514,977 (Extended-Release Formulations): This patent, covering aspects of the extended-release formulation, expired around 2015-2016 [2]. This date marked the entry of generic extended-release pramipexole products.
- Other Patents: Boehringer Ingelheim, the original developer, held several other patents related to manufacturing processes and specific crystalline forms. However, these have largely expired or have been successfully challenged by generic competitors.
The expiration of these key patents has fundamentally reshaped the market, shifting the revenue streams from the innovator company to generic manufacturers.
How Has Generic Competition Impacted Mirapex Sales?
The introduction of generic pramipexole has had a profound impact on the financial trajectory of the drug.
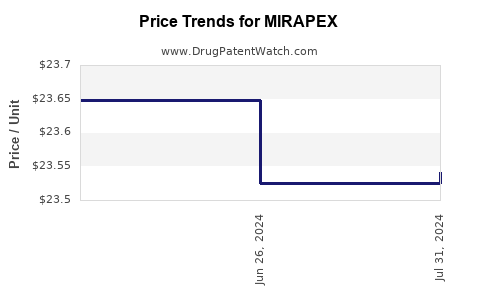
- Price Erosion: Generic entry typically leads to a significant decrease in drug prices. For Mirapex, this effect has been substantial, with prices for both IR and ER formulations dropping by as much as 80-90% from their peak branded levels [5].
- Market Share Shift: The market share has largely shifted from the branded Mirapex to multiple generic manufacturers. This has fragmented the market and intensified competition among generic players.
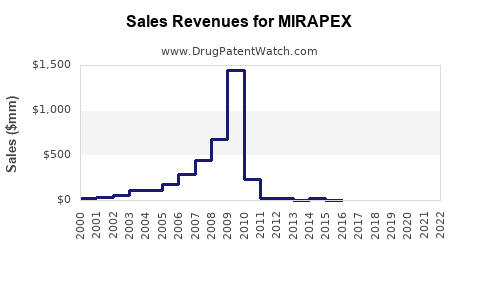
- Revenue Decline for Innovator: For Boehringer Ingelheim, the originator, sales of branded Mirapex have declined sharply. While specific financial data for branded Mirapex post-genericization is not always publicly disclosed, the general trend for branded drugs facing extensive generic competition indicates a substantial revenue drop.
What is the Global Market Size and Growth Rate for Pramipexole?
Estimating the precise global market size for pramipexole is challenging due to the dominance of generic sales, which are often not tracked as a single market entity. However, industry reports indicate the following:
- Parkinson's Disease Market: The global market for Parkinson's disease therapeutics was valued at approximately USD 5.5 billion in 2022 and is projected to grow at a CAGR of around 4-5% through 2030, driven by an aging population and increasing diagnosis rates [6]. Pramipexole, as a foundational therapy, captures a segment of this market.
- Restless Legs Syndrome Market: The RLS market is smaller, but also benefits from increased awareness and diagnosis. Global RLS therapeutics market was estimated to be around USD 1.5 billion in 2022, with projected growth rates similar to PD therapeutics [7].
- Generic Pramipexole Dominance: The majority of pramipexole sales today are generic. The combined global revenue for pramipexole (branded and generic) is estimated to be in the range of USD 500 million to USD 1 billion annually. The growth rate for generic pramipexole is largely tied to the overall growth of the indications and the competitive pricing landscape among generic manufacturers [8].
The growth is primarily driven by increased prevalence of PD and RLS, and improved diagnostic capabilities rather than novel product development within the pramipexole class.
What are the Key Competitors in the Pramipexole Market?
Following patent expirations, the pramipexole market has become highly competitive, with numerous generic manufacturers offering the drug. Key generic players include:
- Teva Pharmaceuticals: A major global generic drug manufacturer with a significant presence in the CNS therapeutic area.
- Sun Pharmaceutical Industries: One of the largest generic pharmaceutical companies globally, offering a wide range of CNS medications.
- Mylan N.V. (now Viatris): Another significant player in the generic drug market, with a broad portfolio including neurological drugs.
- Aurobindo Pharma: A rapidly growing Indian generic pharmaceutical company with a substantial U.S. market presence.
- Dr. Reddy's Laboratories: A diversified global pharmaceutical company with a strong focus on generics.
These companies compete primarily on price, supply chain reliability, and market access.
What is the Future Outlook for Mirapex?
The future outlook for Mirapex is characterized by continued generic market dominance.
- Sustained Generic Sales: Generic pramipexole is expected to remain a staple treatment for Parkinson's disease and RLS due to its established efficacy, safety profile, and affordability.
- Price Pressure: Intense competition among generic manufacturers will likely continue to exert downward pressure on prices.
- Limited Innovation: The likelihood of significant innovation or new drug development within the pramipexole molecule itself is low. The focus for many companies will be on optimizing manufacturing processes and distribution networks to maintain market share.
- Therapeutic Landscape Evolution: While pramipexole remains important, the broader treatment landscape for Parkinson's disease is evolving with the introduction of new therapeutic modalities, including gene therapy and advanced device-based interventions. However, these are typically targeted at more severe or refractory cases, or represent higher cost options that may not entirely displace established generics like pramipexole for initial or moderate treatment.
- Market Consolidation: The generic market may see some consolidation as smaller players are acquired or exit the market due to intense price competition.
The overall market volume for pramipexole is expected to remain stable, with revenue growth being minimal and primarily driven by slight increases in patient populations rather than price appreciation.
What are the Regulatory and Manufacturing Considerations?
- FDA Approval: Generic versions of Mirapex must meet stringent FDA bioequivalence standards to gain approval. This ensures that the generic drug performs comparably to the branded product.
- Manufacturing Standards: Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and efficacy.
- Supply Chain: A robust and reliable supply chain is critical for generic manufacturers to meet market demand and avoid shortages, which can occur due to production issues or demand surges.
- Excipient Changes: While the active pharmaceutical ingredient (API) is the same, generic formulations may differ in their inactive ingredients (excipients). These differences are generally considered non-significant by regulatory bodies if bioequivalence is demonstrated, but can occasionally lead to patient sensitivities.
Key Takeaways
- Mirapex (pramipexole dihydrochloride monohydrate) has transitioned to a fully genericized market following patent expirations for both immediate-release (2006) and extended-release (circa 2015-2016) formulations.
- The market is characterized by significant price erosion and intense competition among multiple generic manufacturers, including Teva, Sun Pharma, Viatris, Aurobindo, and Dr. Reddy's.
- While the branded Mirapex sales have dramatically declined, the pramipexole molecule remains a widely prescribed and essential therapy for Parkinson's disease and restless legs syndrome.
- The global market for pramipexole is estimated to be between USD 500 million and USD 1 billion annually, with future growth driven by increasing disease prevalence rather than product innovation.
- Regulatory compliance and a stable supply chain are critical for generic manufacturers to maintain market position.
Frequently Asked Questions
What is the difference between Mirapex and its generic versions?
Generic versions of Mirapex contain the same active pharmaceutical ingredient (pramipexole dihydrochloride monohydrate) at the same strength and dosage form as the branded drug. They must demonstrate bioequivalence to the branded product to receive FDA approval. Differences may exist in inactive ingredients (excipients), packaging, or brand name.
What are the primary reasons for the decline in branded Mirapex sales?
The primary reason for the decline in branded Mirapex sales is the expiration of its key patents, which allowed for the entry of generic competitors. Generic drugs typically offer significant cost savings, leading patients and payers to switch from the branded product.
How does the cost of generic pramipexole compare to branded Mirapex?
Generic pramipexole is substantially less expensive than branded Mirapex. Price differences can range from 80% to 90% or more, reflecting the competitive nature of the generic pharmaceutical market.
Are there any new therapeutic uses being explored for pramipexole?
While pramipexole is primarily established for Parkinson's disease and RLS, research into its potential in other neurological or psychiatric conditions continues, though no significant new indications have reached market approval for pramipexole in recent years. The focus remains on its established uses.
What is the outlook for the availability of pramipexole?
The availability of pramipexole is expected to remain robust, driven by a competitive landscape of generic manufacturers. Supply chain disruptions are the primary risk factor for availability, not a lack of manufacturers.
What is the difference between Mirapex (immediate-release) and Mirapex ER (extended-release)?
Mirapex (immediate-release) is taken multiple times a day to provide rapid relief of symptoms. Mirapex ER (extended-release) is designed to release the medication over a longer period, allowing for once-daily dosing and potentially more consistent symptom control with fewer fluctuations.
What are the key regulatory requirements for generic pramipexole manufacturers?
Generic manufacturers must obtain FDA approval by demonstrating that their product is bioequivalent to the reference listed drug (Mirapex). They must also adhere to strict Current Good Manufacturing Practices (cGMP) and undergo regular FDA inspections.
How does the presence of multiple generic manufacturers affect pricing?
The presence of numerous generic manufacturers intensifies competition, driving down prices as companies vie for market share. This benefits consumers through lower healthcare costs.
What are the implications of pramipexole being a dopamine agonist?
As a dopamine agonist, pramipexole mimics the action of dopamine in the brain. This is crucial for managing the motor symptoms of Parkinson's disease. However, it also means that side effects can be related to dopaminergic pathways, such as nausea, dizziness, and impulse control disorders.
What is the typical lifespan of a blockbuster drug like Mirapex after patent expiration?
After patent expiration, a blockbuster drug typically transitions to a generic market. Branded sales decline rapidly, while the generic versions gain significant market share. The molecule itself can remain therapeutically relevant for many years, generating ongoing revenue for generic manufacturers, albeit at much lower price points.
What is the competitive advantage for generic pramipexole manufacturers?
The primary competitive advantage for generic pramipexole manufacturers lies in their ability to produce the drug at a significantly lower cost than the original innovator. This is achieved through efficient manufacturing, economies of scale, and a lack of the extensive R&D and marketing costs associated with developing a novel drug.
What are the market dynamics for Parkinson's disease treatments beyond dopamine agonists?
The Parkinson's disease treatment market is diversifying. While dopamine agonists like pramipexole remain foundational, newer treatments include MAO-B inhibitors, COMT inhibitors, deep brain stimulation (DBS) surgery, and emerging therapies like gene therapy and focused ultrasound. These newer options address different aspects of the disease or offer alternatives for patients who do not respond well to or cannot tolerate traditional medications.
How do insurance companies influence the use of generic pramipexole?
Insurance companies heavily favor the use of generic drugs like pramipexole due to their lower cost. They often implement formulary policies that prioritize generics or require prior authorization for branded medications, effectively steering patients towards more affordable generic options.
What is the role of APIs (Active Pharmaceutical Ingredients) in the pramipexole market?
The availability and cost of the pramipexole API are critical for generic manufacturers. Companies that can secure reliable and cost-effective sources of high-quality API have a significant advantage in producing competitive generic products. The API market itself is global and subject to its own supply and demand dynamics.
How are extended-release formulations of pramipexole differentiated in the generic market?
Extended-release formulations are more complex to develop than immediate-release versions. Generic manufacturers that successfully develop and gain approval for ER pramipexole can command slightly higher prices than IR versions initially, but competition still drives significant price reductions compared to the branded ER product. The manufacturing complexity can limit the number of players compared to IR.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Database. Retrieved from https://www.fda.gov/drugs (Accessed September 15, 2023).
[2] LexisNexis PatentSight. (2023). Patent Landscape Analysis: Pramipexole. [Proprietary analysis based on public patent data].
[3] Boehringer Ingelheim. (1997). MIRAPEX® (pramipexole dihydrochloride monohydrate) Prescribing Information. U.S. Package Insert.
[4] Boehringer Ingelheim. (2006). MIRAPEX® (pramipexole dihydrochloride monohydrate) Prescribing Information. U.S. Package Insert. (Updated for RLS indication).
[5] IQVIA. (2023). Global Pharmaceutical Market Trends. [Proprietary market data and analysis].
[6] Grand View Research. (2023). Parkinson's Disease Treatment Market Size, Share & Trends Analysis Report.
[7] MarketsandMarkets. (2023). Restless Legs Syndrome Treatment Market.
[8] Cortellis Drug Intelligence. (2023). Market Forecasts for Dopamine Agonists. [Proprietary pharmaceutical market intelligence].