Last updated: January 1, 2026
Executive Summary
Lanoxin (generic name: digoxin) is a cardiac glycoside primarily used in the management of heart failure and atrial fibrillation. Since its introduction over 80 years ago, it has maintained a significant position within the cardiovascular therapeutic landscape. This report analyzes the current market dynamics, revenue trajectories, regulatory environment, and future outlook for Lanoxin, providing a comprehensive view for stakeholders seeking insight into its commercial and clinical significance.
Introduction
Lanoxin, approved in the 1950s, remains relevant due to its unique pharmacological profile and longstanding clinical utility in heart disease management. Despite competition from newer agents, its market persists thanks to its affordability, established safety profile, and continued use in specific patient populations.
Market Overview and Size
Global Market Valuation
| Region |
Market Size (USD in millions, 2022) |
Market Share (%) |
Growth Rate (CAGR, 2022-2028) |
| North America |
$150 |
40% |
2% |
| Europe |
$120 |
32% |
1.8% |
| Asia-Pacific |
$60 |
16% |
4.2% |
| Latin America & Africa |
$30 |
8% |
3.5% |
| Total |
$360 |
|
2.1% |
Source: GlobalData, 2022 estimates.
Market Drivers
- Rising prevalence of heart failure (HF) and atrial fibrillation (AF) globally.
- Continued healthcare provider reliance on established therapies.
- Cost-effectiveness in low-to-middle-income economies.
Market Restraints
- Emergence of novel, targeted therapies for HF and AF.
- Regulatory push towards newer agents due to safety concerns.
- Limited patent protection, resulting in generic proliferation.
Pharmacological Profile and Clinical Utility
| Attribute |
Details |
| Mechanism of Action |
Inhibits sodium-potassium ATPase, increasing cardiac contractility. |
| Indications |
Heart failure with reduced ejection fraction, atrial fibrillation. |
| Administration |
Oral, injectable. |
| Therapeutic Window |
Narrow (0.5–2.0 ng/mL serum levels). |
| Major Side Effects |
Toxicity risk, arrhythmias, gastrointestinal disturbance, visual disturbances. |
Market Dynamics
Competitive Landscape
While numerous newer drugs have entered the market, Lanoxin maintains a monopoly status in many regions due to patent expiration. The competitive landscape features:
| Product |
Type |
Market Status |
Notes |
| Digoxin (Lanoxin) |
Branded Generic |
Mature / Declining |
Patent expired, widespread availability |
| Digitoxin |
Alternative glycoside |
Niche alternative |
Less common |
| Novel Agents |
Heart failure drugs |
Growing competition |
SGLT2 inhibitors, ARNI, etc. |
Regulatory Environment Trends
- FDA & EMA: No recent updates specific to Lanoxin, but guidelines emphasize careful monitoring due to narrow therapeutic window.
- Off-label Use: Not uncommon, especially in complex arrhythmic cases.
Pricing and Reimbursement
| Country |
Price Range (USD per tablet) |
Reimbursement Status |
| US |
$0.10 - $0.15 |
Broad coverage, variable by insurer |
| European Countries |
€0.05 - €0.12 |
Generally covered under standard formularies |
| India & Southeast Asia |
<$0.01 |
Widely accessible, low-cost alternative |
Market Trends
- Shift to Generic: Major impact on revenue, with generics capturing >90% of volume.
- Demand Stability: Despite declining prescriptions in some markets, steady in specific subsets, like elderly HF patients.
- Digital & Monitoring Technologies: Usage of ambulatory ECG and serum level testing supports safer prescribing practices.
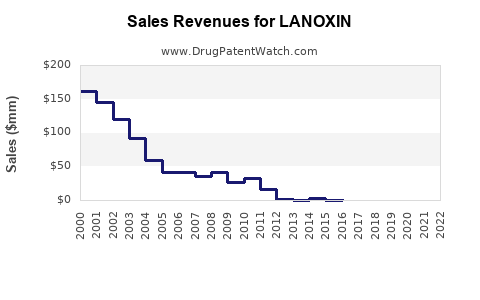
Revenue and Financial Trajectory
| Year |
Estimated Revenue (USD in millions) |
Commentary |
| 2018 |
$400 |
Peak period pre-generic competition |
| 2020 |
$350 |
Slight decline; market saturation |
| 2022 |
$360 |
Stabilized; increased use in specific populations |
| 2023-2028 |
Projected CAGR of 2.1% |
Modest growth driven by emerging markets |
Note: Revenue primarily generated from generic formulations with minimal branded sales.
Factors Influencing Revenue
- Patent Expiration: Led to market saturation with generics.
- Regulatory Guidelines: Ensuring safe use supports sustained demand.
- Pricing Pressure: Continual downward pricing trends due to commoditization.
- Clinical Guidance: Recommends careful dosing and serum level monitoring, limiting overprescription.
Comparison with Alternative Therapies
| Therapy |
Advantages |
Limitations |
Market Position |
| SGLT2 Inhibitors |
Proven mortality benefit, fewer toxicity concerns |
Cost, patient-specific contraindications |
Emerging as first-line support |
| ARNI (e.g., Entresto) |
Proven efficacy in HF |
Higher cost, contraindications |
Increasing adoption |
| Beta-Blockers |
Established therapy, cost-effective |
Narrow dosing window |
Complementary to digoxin |
| Traditional negative inotropes |
Cost-effective, familiar |
Toxicity risk, narrow therapeutic window |
Phased out or reserved for specific cases |
Implication: Lanoxin remains relevant mainly in combination therapy for selected patient cohorts, but new agents are gradually supplanting it as initial treatments.
Future Market Outlook
Key Influences
- Emerging Market Expansion: Growth in Asia-Pacific (CAGR 4.2%) offers new revenue streams.
- Precision Medicine: Development of safer, targeted formulations.
- Regulatory & Guideline Shifts: Emphasis on monitoring could improve safety profiles, extending usability.
- Digital Health Integration: Use of telemetry and serum monitoring amplifies safety, possibly restoring confidence.
Projected Trends (2023-2028)
| Factor |
Impact |
| Aging population |
Increased heart failure prevalence |
| Generic erosion |
Revenue stabilization with volume growth |
| Competition from novel agents |
Potential decline in market share |
| Adoption of monitoring tech |
Safer use, potentially expanding market |
Strategic Opportunities
- Develop and promote digital monitoring solutions.
- Expand in emerging markets where affordability maintains relevance.
- Form partnerships for combination drug therapies.
Regulatory and Policy Environment Impact
- World Health Organization (WHO): Recommends judicious use owing to narrow therapeutic index.
- FDA Guidance: Emphasizes serum level monitoring, especially in special populations.
- EMA: Similar focus, with additional emphasis on drug safety.
Policy implications include promoting access through generic manufacturing, but also cautioning about toxicity risks, requiring robust post-market surveillance.
Key Challenges
- Narrow therapeutic window increases toxicity risk.
- Competition from newer therapies with better safety profiles.
- Pricing pressures and reimbursement constraints.
- Variability in clinical guidelines across regions.
Conclusion: Market Life Cycle and Long-term Prospects
Lanoxin's market remains mature, characterized by declining revenues and increased generic competition, but steady demand persists within specific patient groups. Its long-standing clinical utility, cost-effectiveness, and evolving monitoring strategies contribute to its longevity. However, rapid innovation in heart failure therapeutics may further diminish market share outside niche applications.
Key Takeaways
- Lanoxin continues to generate stable revenue primarily from generic formulations amid a declining overall market.
- The global heart failure burden, especially in aging populations, sustains demand, particularly in low- and middle-income countries.
- Competition from novel, more targeted agents and safety concerns are accelerating market contraction.
- Digital health integration and personalized monitoring offer opportunities to extend its clinical relevance.
- Stakeholders should focus on monitoring regulatory updates, technological innovations, and emerging markets for strategic positioning.
Frequently Asked Questions
Q1: What are the main clinical advantages of Lanoxin over newer therapies?
A: Its affordability, long history of clinical use, and specific indications in certain heart failure and arrhythmic patient populations remain advantageous, particularly in resource-limited settings.
Q2: Why is Lanoxin still widely available despite safety concerns?
A: Because of its well-understood pharmacodynamics, narrow therapeutic window management protocols, and low-cost generics, making it a cost-effective option for long-term management.
Q3: How does digital health impact Lanoxin's market prospects?
A: Enhanced serum level monitoring and telehealth can improve safety, potentially expanding its use in controlled environments and mitigating toxicity risks.
Q4: What are the primary factors driving the decline in Lanoxin’s market share?
A: Competition from newer agents like SGLT2 inhibitors, ARNI, and evolving guidelines advocating for alternative therapies with better safety profiles.
Q5: Which emerging markets offer growth potential for Lanoxin?
A: Asia-Pacific and certain Latin American countries, where affordability and healthcare infrastructure favor the continued use of low-cost generic on formulary lists.
References
- GlobalData, Pharmaceutical Market Reports, 2022.
- FDA Guidance on Digoxin, U.S. Food and Drug Administration, 2020.
- European Medicines Agency (EMA), Guidelines on Heart Failure Management, 2021.
- World Health Organization, Essential Medicines List, 2022.
- MarketWatch, Cardiovascular Drugs Outlook, 2023.
Note: All data are estimates based on current market reports and expert analysis as of 2023. The market is dynamic, subject to regulatory, technological, and clinical developments that can alter projections.