Last updated: February 13, 2026
What Are the Market Dynamics for Isradipine?
Isradipine is a dihydropyridine calcium channel blocker primarily used to treat hypertension. The drug’s market is constrained by several factors: generic competition, limited indication scope, and regulatory shifts.
Current Market Landscape
- Market Presence: Dominated by generic forms, leading to low retail prices.
- Competition: Several suppliers offer generic formulations, eroding brand value.
- Indications: Approved for hypertension and angina, but lacks expansion into broader cardiovascular or cerebrovascular risk reduction.
Key Drivers
- Prevalence of Hypertension: Over 1.3 billion people suffer from high blood pressure globally [1]. This high prevalence sustains demand for antihypertensive medications.
- Generic Penetration: Extensive generic availability reduces revenue opportunities for branded versions.
- Prescribing Trends: Preference shifts toward newer drugs with better side-effect profiles (e.g., amlodipine).
Barriers to Growth
- Limited Innovation: No recent advancements or new formulations.
- Market Saturation: Existing generics saturate the market; minimal room for price increases.
- Regulatory Focus: Emphasis on expanding indications or demonstrating superior efficacy is limited.
Future Market Opportunities
- New Formulations: Extended-release or combination formulations could gain traction.
- Regional Markets: Growth potential exists in emerging markets with expanding healthcare infrastructure.
- Patent Status: No recent patents restrict generics; patent expiry four decades ago (1980s).
What Is the Financial Trajectory of Isradipine?
Financial prospects for isradipine are subdued due to pricing pressure, high generic competition, and minimal innovation pipeline.
Revenue Trends
- Estimated global sales hover around $50 million annually, declining since patent expiration.
- Leading generic manufacturers account for the majority of sales.
Cost Considerations
- Manufacturing Costs: Low, due to established production processes.
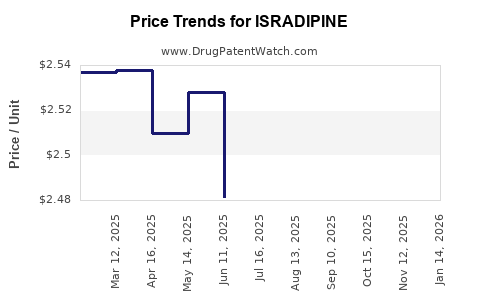
- Pricing: Declining, driven by aggressive generics pricing; average retail price per prescription approximates $10.
- Margins: Gross margins are near 60-70% for branded formulations but are minimal for generics due to competitive pricing.
Investment Outlook
- Market Stability: Limited growth prospects.
- Entry Opportunities: Challenging due to high market saturation and low innovation incentives.
- Potential Gains: Focused primarily on niche markets or regional expansion.
Mergers and Acquisitions
- Recent consolidation in the generic antihypertensive market attempts to optimize manufacturing and distribution but has not significantly changed the financial outlook for isradipine.
How Does Isradipine Compare to Other Calcium Channel Blockers?
| Drug Name |
Indications |
Patent Status |
Annual Sales |
Main Competitors |
| Amlodipine |
Hypertension, angina |
Patent expired (2010) |
~$1.2 billion |
Multiple generics |
| Nifedipine |
Hypertension, angina |
Patent expired (early 2000s) |
~$600 million |
Multiple generics |
| Isradipine |
Hypertension |
Patent expired (1980s) |
~$50 million |
Several generics |
The longevity of patents for other calcium channel blockers has led to mature markets with high generic penetration. Isradipine's older patent status and narrower indication scope compare unfavorably.
What Is the Regulatory Environment Impact?
- No recent regulatory barriers specific to isradipine.
- No approved new indications or formulations to extend patent exclusivity.
- Generic approvals dominate the market, reducing the likelihood of regulatory hurdles improving its financial trajectory.
Closing Summary
Isradipine remains a low-growth, low-margin antihypertensive agent. Its market is characterized by widespread generic availability, limited scope for differentiation, and minimal innovation. Significant revenue is unlikely without reformulation or indication expansion. Regional growth opportunities exist, but overall market dynamics favor generic competition over brand-driven growth.
Key Takeaways
- Isradipine’s global sales are declining due to saturation in the generic market.
- Patent expirations in the 1980s have resulted in abundant generic competition.
- Opportunities for revenue growth are limited to niche applications or emerging markets.
- Industry innovation focus has pivoted toward newer drugs with improved safety profiles.
- Investment in isradipine’s future appears risk-averse given current market conditions.
FAQs
1. What is the main driver for isradipine sales?
The primary driver is the high prevalence of hypertension worldwide, sustaining demand for generic calcium channel blockers.
2. Are there any recent developments to extend isradipine’s market?
No, there have been no recent formulations or regulatory submissions to expand indications or improve drug profiles.
3. How does patent expiration affect isradipine’s market?
Patent expiration in the 1980s allowed multiple generic manufacturers to enter, leading to significant price erosion and reduced profitability.
4. What role do regional markets play in isradipine’s future?
Emerging markets with expanding healthcare infrastructure may offer growth opportunities, but price sensitivity could limit revenue potential.
5. Can isradipine become a premium or specialty drug?
Unlikely, as the drug’s patent status, competition, and therapeutic profile do not support premium pricing or repositioning.
Sources:
- World Health Organization. "High blood pressure." 2021.