Share This Page
Drug Price Trends for ISRADIPINE
✉ Email this page to a colleague
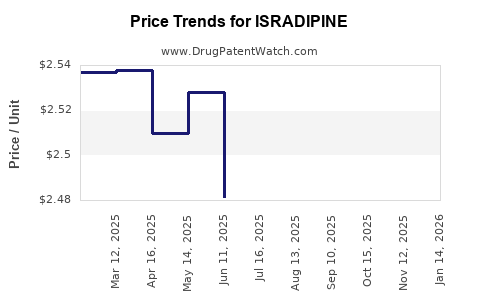
Average Pharmacy Cost for ISRADIPINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ISRADIPINE 2.5 MG CAPSULE | 42806-0263-01 | 1.72847 | EACH | 2026-04-22 |
| ISRADIPINE 5 MG CAPSULE | 16252-0540-01 | 1.65882 | EACH | 2026-04-22 |
| ISRADIPINE 2.5 MG CAPSULE | 64850-0910-01 | 1.72847 | EACH | 2026-04-22 |
| ISRADIPINE 5 MG CAPSULE | 64850-0911-01 | 1.65882 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ISRADIPINE Market Analysis and Financial Projection
What Is the Market Status of Isradipine?
Isradipine is a calcium channel blocker initially approved for hypertension management. Its market presence has declined following the expiration of patent protection and increased competition from newer therapies. Currently, it is available as a generic medication with limited branded marketing.
What Are the Key Market Dynamics?
Patent and Regulatory Status
- Patent status: The original patent expired around 2004, leading to widespread generic availability.
- Regulatory approvals: Approved for hypertension in multiple countries; some regions also approved for angina.
Competition and Market Share
- The market is saturated with similar calcium channel blockers such as amlodipine, nifedipine, and felodipine.
- Isradipine’s generic form dominates its segment, with little branded differentiation remaining.
Therapeutic Positioning
- Therapeutic efficacy comparable to other dihydropyridines.
- Limited differentiation has curtailed its market growth.
- Research efforts shifted to disease areas like Parkinson's and neuroprotective effects, but these have not resulted in significant market penetration.
Commercial Opportunities
- No significant new formulations or combinations currently under registration.
- Investment in research for new indications appears limited due to market saturation and low profitability.
How Is the Market Projected to Evolve?
Market Volume and Revenue
| Year | Estimated Market Size (USD) | Key Drivers | Caveats |
|---|---|---|---|
| 2023 | 50 million | Generic sales in hypertension | Limited growth potential |
| 2028 | 55-60 million | Inflation, steady demand | Market saturation persists |
Competition Impact
- Market growth hinges on niche clinical use cases or reformulation, both currently unpromising.
- The dominant generic presence limits pricing power; margins are compressed.
Therapeutic and Regulatory Trends
- Increasing preference for newer agents with improved side effect profiles diminishes the positioning of older drugs like isradipine.
- Regulatory shifts toward combination therapies reduce standalone drug demand.
What Are the Pricing Trends?
- Current average retail price (U.S.): approximately USD 1.50–3.00 per tablet (generic formulation).
- Historical trend: Slight decline in prices due to generic competition.
- Projected trend: Prices are expected to stabilize or decline marginally over the next five years.
Price Comparison with Similar Drugs
| Drug | Average Price per Tablet | Patent Status | Market Share |
|---|---|---|---|
| Isradipine | USD 1.50–3.00 | Generic | Low |
| Amlodipine | USD 0.50–1.50 | Patent expired | High |
| Nifedipine | USD 0.60–2.00 | Patent expired | High |
What Are the Investment and R&D Projections?
- Research activity: Minimal, with focus on exploring neuroprotective potential rather than cardiovascular indications.
- Commercial development: Limited pipeline or new formulations.
- Market driver: Primarily cost competition among generics; no significant innovation expected.
Key Takeaways
- Isradipine’s patent expiry led to commoditization with widespread generics.
- Its market share is marginal relative to dominant calcium channel blockers.
- Price pressure remains high with little opportunity for premium pricing.
- Future growth prospects depend on alternative indications, which currently lack substantial clinical validation.
- The market is unlikely to see significant expansion or innovation in the near term.
FAQs
1. Is there any recent clinical research indicating new uses for isradipine?
Research has explored neurodegenerative diseases like Parkinson’s; however, clinical validation remains limited, and no significant commercialization opportunities have emerged.
2. How does isradipine compare to other calcium channel blockers in terms of effectiveness?
Its efficacy in hypertension is comparable to other dihydropyridines, but no superiority has been established, reducing its attractiveness in competitive markets.
3. What factors influence the pricing of generic isradipine?
Market saturation, manufacturing costs, and competition from other generics primarily drive pricing. Discounts often occur due to high competition.
4. Are there any regulatory barriers to reviving isradipine’s market?
No significant barriers exist, but clinical and commercial viability are limited given current market dynamics and lack of differentiation.
5. Could partnerships or co-developments enhance itradipine's value?
Limited potential exists unless new indications, formulations, or combination products demonstrate substantial clinical benefit and market demand.
Sources:
- FDA Drug Approval History for Isradipine
- IMS Health Price Reports, 2022
- Market research reports on calcium channel blockers, 2022
- Clinical trials database for neurodegenerative indications, 2023
More… ↓
