HEPARIN Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Heparin, and when can generic versions of Heparin launch?
Heparin is a drug marketed by Hospira, Intl Medication, Luitpold, Parke Davis, Smith And Nephew, Solopak, Abraxis Pharm, Aspen Global Inc, B Braun Medical, Be Pharms, Casi Pharms Inc, Chamberlin Parenterl, Dell Labs, Dr Reddys, Emerge Bioscience, Epic Pharma Llc, Fresenius Kabi Usa, Gland, Hikma, Hospira Inc, Lilly, Pfizer, Pharm Spec, Pharmacia And Upjohn, Pharmobedient, Plano Pharms, Sagent Pharms, Shenzhen Techdow, Watson Labs Inc, Baxter Hlthcare, Mcgaw, B Braun, and Pharma Serve Ny. and is included in eighty-four NDAs.
The generic ingredient in HEPARIN is heparin sodium. There are seventy-seven drug master file entries for this compound. Twenty-five suppliers are listed for this compound. Additional details are available on the heparin sodium profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Heparin
A generic version of HEPARIN was approved as heparin sodium by HOSPIRA on April 28th, 1983.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for HEPARIN?
- What are the global sales for HEPARIN?
- What is Average Wholesale Price for HEPARIN?
Summary for HEPARIN
| US Patents: | 0 |
| Applicants: | 33 |
| NDAs: | 84 |
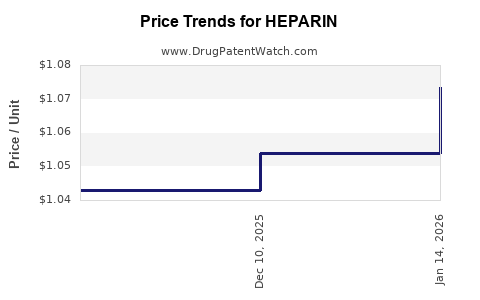
| Drug Prices: | Drug price information for HEPARIN |
| DailyMed Link: | HEPARIN at DailyMed |
US Patents and Regulatory Information for HEPARIN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gland | HEPARIN SODIUM | heparin sodium | INJECTABLE;INJECTION | 205323-001 | Feb 6, 2017 | AP | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Pfizer | HEPARIN SODIUM | heparin sodium | INJECTABLE;INJECTION | 201370-002 | Jul 21, 2011 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Dr Reddys | HEPARIN SODIUM | heparin sodium | INJECTABLE;INJECTION | 017064-015 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Chamberlin Parenterl | HEPARIN SODIUM | heparin sodium | INJECTABLE;INJECTION | 017130-003 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
DrugChatter Q&A for HEPARIN
How is the demand for heparin expected to change?
What are the emerging applications of heparin in the market?
What factors contribute to the current market value of heparin?
Are there any recent developments in heparin production techniques?