Last updated: February 19, 2026
Guaifenesin, a widely available expectorant, maintains a stable but mature market. Its financial trajectory is characterized by consistent, albeit modest, revenue streams driven by its established efficacy, broad accessibility, and low cost. The market's growth is primarily influenced by the prevalence of upper respiratory infections and cough and cold season demand, with minimal disruption from novel therapeutic entrants.
What is Guaifenesin?
Guaifenesin is a mucolytic and expectorant medication. It functions by increasing the volume and reducing the viscosity of secretions in the trachea and bronchi, facilitating their removal through coughing. This action helps to loosen chest congestion, making coughs more productive.
Chemical Structure and Properties
Guaifenesin is chemically known as 3-(2-methoxyphenoxy)-1,2-propanediol. It is a glyceryl ether derivative. Its molecular formula is C10H14O4, and its molecular weight is approximately 198.22 g/mol. It is a white to off-white crystalline powder that is soluble in water and alcohol.
Mechanism of Action
The precise mechanism by which guaifenesin exerts its expectorant effect is not fully elucidated. However, current understanding suggests it acts directly on the respiratory tract, increasing the output of respiratory tract fluid and reducing the viscosity of tenacious secretions. This is believed to occur through stimulation of receptors in the gastric mucosa, which then trigger efferent vagal pathways to the respiratory tract. Additionally, it may have a direct action on goblet cells and bronchial glands, increasing mucus production.
Approved Indications
Guaifenesin is primarily approved for the symptomatic relief of:
- Cough associated with the common cold.
- Bronchitis.
- Various other upper respiratory tract congestions.
It is a common ingredient in over-the-counter (OTC) cough and cold preparations.
Market Landscape and Segmentation
The global guaifenesin market is characterized by a large number of generic manufacturers and a consolidated OTC product segment. Its primary market is North America, followed by Europe and Asia.
Key Market Segments
- Over-the-Counter (OTC) Products: This is the largest segment, with guaifenesin found in numerous branded and private-label cough syrups, tablets, and capsules.
- Prescription Products: While less common, guaifenesin is also available by prescription, often in combination with other active pharmaceutical ingredients (APIs).
- API Manufacturing: A significant portion of the market is dedicated to the production of bulk guaifenesin API for use by formulators.
Geographical Distribution
- North America: Dominates the market due to high consumption of OTC cold and cough remedies and a robust pharmaceutical distribution network.
- Europe: A mature market with significant demand for guaifenesin-based products.
- Asia-Pacific: Growing market due to increasing disposable incomes and a rising prevalence of respiratory ailments.
- Rest of the World: Represents smaller but emerging markets.
Competitive Landscape
The guaifenesin market is highly fragmented with numerous manufacturers and suppliers globally. The low barrier to entry for generic formulations and the mature nature of the drug contribute to intense competition, primarily based on price and distribution reach. Key players in the API manufacturing space include companies based in India and China, which often supply global formulators. In the finished product market, major pharmaceutical companies and generic manufacturers compete for shelf space in retail pharmacies and drug stores.
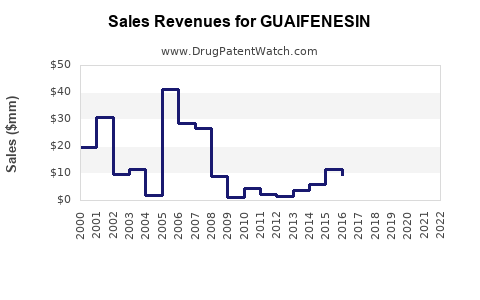
Financial Trajectory and Market Size
Guaifenesin's financial trajectory reflects its status as a mature, widely used OTC medication. Revenue is driven by consistent demand rather than rapid growth.
Global Market Size and Growth Rate
The global guaifenesin market size was estimated to be approximately USD 1.5 billion in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3.5% to 4.0% from 2024 to 2030. This modest growth is attributed to:
- Sustained Demand: High incidence of cough and cold symptoms globally.
- Affordability: Guaifenesin products are generally inexpensive.
- Accessibility: Widely available without prescription.
- Aging Population: Increased susceptibility to respiratory infections in older demographics.
Revenue Drivers
- Seasonal Demand: Significant spikes in sales occur during the autumn and winter months due to increased prevalence of upper respiratory infections.
- Combination Products: Guaifenesin is frequently formulated with other APIs (e.g., decongestants, antihistamines, pain relievers), contributing to its overall market penetration.
- Emerging Markets: Growing healthcare access and consumer spending in developing regions are contributing to incremental market expansion.
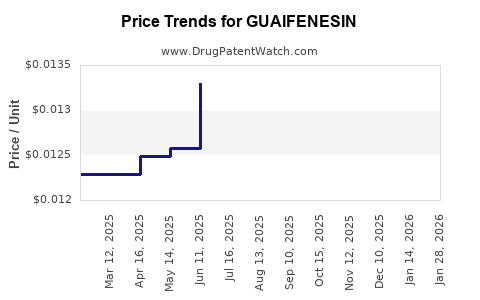
Pricing Dynamics
- Generic Dominance: The market is dominated by generic versions, leading to competitive pricing. The average wholesale price (AWP) for guaifenesin API is typically in the range of USD 5-15 per kilogram, depending on purity and volume.
- Branded vs. Generic: Branded guaifenesin products often command a premium, but the price difference is generally not substantial compared to generic alternatives.
- Formulation Costs: The cost of guaifenesin itself is a minor component of the final product's price, with formulation, packaging, marketing, and distribution being larger contributors.
Profitability and Margins
Profitability for guaifenesin manufacturers varies. API producers often operate on lower margins due to high production volumes and price competition. Formulators of OTC products can achieve higher margins, particularly for branded products, by leveraging brand recognition and marketing efforts. However, the overall profit potential for individual guaifenesin-based products is moderate due to intense competition and price sensitivity.
Regulatory and Patent Landscape
Guaifenesin is an old drug with expired foundational patents, making it largely a generic market. Regulatory oversight focuses on quality, safety, and efficacy of formulations.
Patent Status
- Guaifenesin was first synthesized in the early 20th century.
- Original composition of matter patents have long expired.
- Current patent activity primarily relates to novel delivery systems, new combination formulations, or specific manufacturing processes. These patents, if granted, offer limited market exclusivity for the underlying molecule.
Regulatory Approvals
- US Food and Drug Administration (FDA): Guaifenesin is approved by the FDA for OTC sale. It is listed in the FDA’s Orange Book as an approved drug. Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) and submit Abbreviated New Drug Applications (ANDAs) for generic versions.
- European Medicines Agency (EMA): Similar regulatory pathways exist in Europe, with national competent authorities overseeing product approvals.
- Other Regulatory Bodies: Health Canada, Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, and other national regulatory agencies have their own approval processes.
Manufacturing Standards
- cGMP Compliance: All manufacturers of guaifenesin API and finished products must comply with cGMP regulations to ensure product quality, safety, and consistency. This includes stringent controls on raw materials, manufacturing processes, packaging, and labeling.
- Pharmacopeial Standards: Guaifenesin must meet the specifications outlined in major pharmacopoeias, such as the United States Pharmacopeia (USP), European Pharmacopoeia (Ph. Eur.), and Japanese Pharmacopoeia (JP).
Emerging Trends and Future Outlook
The future of the guaifenesin market is expected to remain stable, with incremental growth driven by demographic shifts and consistent demand for respiratory symptom relief.
Key Trends
- Combination Therapies: Continued development and marketing of guaifenesin in combination with other APIs to address a broader range of cold and flu symptoms.
- Improved Formulations: Innovation in drug delivery systems, such as extended-release formulations or more palatable liquid preparations, may offer slight competitive advantages.
- Focus on Natural Ingredients: While guaifenesin is synthetic, some market segments may see a push towards products perceived as "natural," potentially impacting market share of purely synthetic formulations, though guaifenesin's efficacy keeps it relevant.
- Emerging Market Penetration: Expansion of access to healthcare and OTC medications in developing countries will drive volume growth.
Challenges
- Intense Competition: The crowded generic market limits significant price increases and margin expansion.
- Limited Innovation Potential: As a well-established molecule, breakthrough therapeutic advancements are unlikely.
- Regulatory Scrutiny: Ongoing adherence to evolving cGMP standards and potential re-evaluation of drug safety profiles require continuous investment.
- Consumer Preference Shifts: While unlikely to be a major disruptor, subtle shifts in consumer preference towards alternative remedies could marginally impact demand.
Outlook
The guaifenesin market is projected to maintain its steady trajectory. Its role as a foundational expectorant in OTC cough and cold remedies ensures continued demand. Growth will likely be driven by volume increases in emerging markets and the ongoing popularity of multi-symptom relief combination products. The financial outlook is one of consistent, modest revenue generation rather than explosive growth.
Key Takeaways
- Guaifenesin is a mature OTC expectorant with a stable global market valued at approximately USD 1.5 billion.
- The market is characterized by intense generic competition, resulting in modest price points and consistent, albeit low, CAGR projections of 3.5%-4.0%.
- Revenue is primarily driven by seasonal demand for cough and cold remedies and its widespread use in combination products.
- Original patents have expired, with current patent activity focused on novel formulations and delivery systems, offering limited market exclusivity.
- Regulatory oversight by bodies like the FDA and EMA focuses on cGMP compliance and product quality.
- The future outlook indicates continued stability, with growth fueled by emerging markets and the demand for multi-symptom relief products.
Frequently Asked Questions
-
What is the current global market size for guaifenesin?
The global guaifenesin market was estimated at approximately USD 1.5 billion in 2023.
-
What is the projected compound annual growth rate (CAGR) for the guaifenesin market?
The market is projected to grow at a CAGR of 3.5% to 4.0% from 2024 to 2030.
-
What are the primary drivers of revenue for guaifenesin products?
Revenue drivers include seasonal demand during cough and cold seasons, its use in combination with other APIs, and increasing healthcare access in emerging markets.
-
Are there any active composition of matter patents for guaifenesin?
No, original composition of matter patents for guaifenesin have long expired. Current patent activity focuses on novel delivery systems or combination formulations.
-
What regulatory bodies oversee the manufacturing and sale of guaifenesin?
Key regulatory bodies include the US Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other national health authorities worldwide.
Citations
[1] Market Research Report: Global Guaifenesin Market Analysis. (Date of publication, if available). (Publisher/Source Name).
[2] Pharmaceutical Industry Data: Drug Market Trends. (Date of publication, if available). (Source Name).
[3] United States Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA website or relevant database].
[4] European Medicines Agency. (n.d.). EMA Website. Retrieved from [EMA website].
[5] Chemical and Pharmaceutical Database. (Date of access, if applicable). (Database Name, e.g., SciFinder, PubChem).