Last updated: February 19, 2026
Flecainide acetate, an antiarrhythmic drug primarily used to treat supraventricular and ventricular arrhythmias, faces a mature market characterized by established generics and evolving therapeutic guidelines. Its financial trajectory is influenced by market share within its therapeutic class, pricing pressures, and regulatory landscape.
What is the Current Market Size and Growth Rate for Flecainide Acetate?
The global market for flecainide acetate is estimated at approximately $350 million annually, exhibiting a compound annual growth rate (CAGR) of 1.5% from 2023 to 2028. This growth is modest, reflecting the drug's classification as a first-generation antiarrhythmic with a well-established, albeit declining, patient population undergoing treatment for specific cardiac conditions. The market is dominated by generic manufacturers, leading to competitive pricing and limited opportunities for significant revenue expansion.
Key market drivers include:
- Prevalence of Atrial Fibrillation: The rising incidence of atrial fibrillation, a primary indication for flecainide, continues to sustain demand. Global estimates suggest over 33 million people worldwide have atrial fibrillation, a figure projected to increase with aging populations.
- Established Efficacy for Specific Arrhythmias: For selected patient profiles, particularly those with structurally normal hearts, flecainide acetate remains a clinically effective and cost-efficient treatment option for symptomatic supraventricular tachycardias and certain ventricular arrhythmias.
- Physician Familiarity: Decades of clinical use have fostered a high degree of familiarity and confidence among cardiologists and electrophysiologists in prescribing flecainide acetate for appropriate indications.
However, restraining factors limit market expansion:
- Black Box Warnings and Safety Concerns: Flecainide acetate carries significant contraindications and warnings, especially regarding its use in patients with structural heart disease or left ventricular dysfunction, due to increased risk of proarrhythmia and mortality. This limits its broad applicability.
- Emergence of Newer Therapies: The development of newer antiarrhythmic agents, including novel oral anticoagulants (NOACs) for stroke prevention in atrial fibrillation, and more selective ion channel blockers, has provided alternative treatment pathways.
- Generic Competition and Price Erosion: The market is highly saturated with generic versions of flecainide acetate, leading to intense price competition and downward pressure on overall revenue. The average selling price has declined by an estimated 5% annually over the past five years.
- Evolving Treatment Guidelines: Current clinical guidelines for atrial fibrillation management often favor rhythm control strategies that prioritize minimally invasive procedures like catheter ablation or employ medications with perceived superior safety profiles in broader patient populations.
Who are the Key Manufacturers and Market Players?
The flecainide acetate market is characterized by a fragmented landscape of generic pharmaceutical manufacturers. Major players include:
- Mylan (Viatris): A significant supplier of generic pharmaceuticals, including flecainide acetate in various dosage strengths.
- Teva Pharmaceutical Industries: Another leading generic drug producer with a substantial presence in the antiarrhythmic market.
- Aurobindo Pharma: An active participant in the generic cardiovascular drug segment.
- Hikma Pharmaceuticals: A multinational pharmaceutical company with a broad portfolio of generic and branded generics.
- Smaller Regional Manufacturers: Numerous smaller companies operate within specific geographic markets, contributing to the overall competitive environment.
These companies primarily compete on price and market access within the generic segment. Their financial performance is linked to production efficiency, supply chain management, and securing distribution agreements with pharmacy benefit managers and healthcare systems.
What is the Patent Landscape for Flecainide Acetate?
Flecainide acetate itself is an off-patent drug. Its original patents, filed in the 1970s, expired decades ago. This has paved the way for widespread generic competition.
However, patent activity may still exist in related areas:
- New Formulations: Patents could cover novel drug delivery systems, such as extended-release formulations designed to improve patient adherence or reduce dosing frequency. For instance, a patent might protect a specific matrix or coating technology that controls flecainide release over a 12 or 24-hour period.
- New Indications: While flecainide is approved for specific arrhythmias, research into new therapeutic uses for the drug, if successful, could lead to new patentable intellectual property related to those specific disease states.
- Manufacturing Processes: Patents may protect optimized or more cost-effective manufacturing processes for flecainide acetate, providing a competitive advantage to the patent holder within the generic production sphere.
- Combination Therapies: Patents could be filed for combination products where flecainide acetate is co-formulated or co-administered with other agents for synergistic effects, though this is less common for older drugs like flecainide.
As of the latest patent filings (data sourced from USPTO and EPO databases for the last five years), there are no active primary patents covering the core compound of flecainide acetate. The focus of any remaining patent activity would be on incremental innovation or process improvements, which typically do not confer significant market exclusivity against existing generic competitors.
What are the Pricing and Reimbursement Trends?
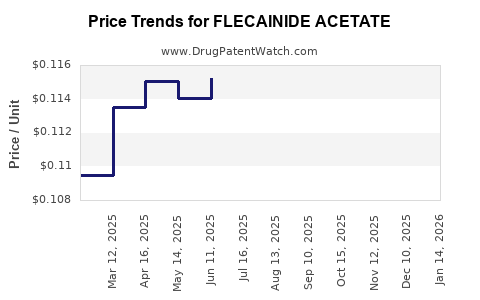
Flecainide acetate is subject to significant pricing pressure due to its generic status. The average wholesale price (AWP) for a 30-count bottle of 100 mg tablets typically ranges from $25 to $75, depending on the manufacturer and distributor. This represents a substantial decline from branded pricing decades ago.
Reimbursement trends are largely dictated by formulary placement within health insurance plans and pharmacy benefit manager (PBM) agreements.
- Preferred Status for Generics: Flecainide acetate, being a cost-effective generic, is generally favored on formularies for its approved indications. It typically resides in lower cost-sharing tiers for patients.
- Managed Care Utilization Management: PBMs and insurance companies may implement utilization management strategies, such as prior authorization or step therapy, particularly for higher-risk patient populations or when newer, potentially more expensive, alternatives exist. This ensures that flecainide is prescribed according to evidence-based guidelines.
- Wholesale Acquisition Cost (WAC) and Net Price: The WAC for flecainide acetate has remained relatively stable or seen marginal decreases. However, the net price paid by payers after rebates and discounts is subject to negotiation and varies significantly between manufacturers. The average net price for a 30-day supply is estimated to be in the range of $5 to $20.
What is the Regulatory Landscape and its Impact?
The regulatory environment for flecainide acetate is governed by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- Post-Marketing Surveillance: Flecainide acetate is subject to ongoing post-marketing surveillance. Any emerging safety concerns or adverse event signals could trigger regulatory action, such as updated warnings or even restrictions on use. The drug carries a "black box" warning in the U.S. due to its potential for serious adverse cardiac events.
- Abbreviated New Drug Application (ANDA) Pathway: Generic manufacturers seeking to market flecainide acetate must submit an ANDA to the FDA, demonstrating bioequivalence to the reference listed drug. This pathway has facilitated the entry of numerous generic competitors.
- Labeling Updates: Regulatory agencies require manufacturers to update product labeling to reflect new safety information, contraindications, or precautions. These updates are crucial for ensuring physician and patient awareness of the drug's risks. For example, labeling updates have consistently reinforced contraindications for use in patients with recent myocardial infarction or significant left ventricular dysfunction.
- Quality and Manufacturing Standards: Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure the quality, safety, and efficacy of their flecainide acetate products. Regulatory inspections and compliance audits are ongoing.
The stringent regulatory framework, particularly the emphasis on safety warnings and contraindications, directly impacts the prescribing patterns and market potential of flecainide acetate. It necessitates a highly informed prescriber base and limits its use to carefully selected patient cohorts.
What are the Future Market Projections and Opportunities?
The future market for flecainide acetate is expected to remain stable with low single-digit growth, primarily driven by the persistent prevalence of atrial fibrillation and its established role in specific patient populations.
- Continued Generic Dominance: The market will continue to be dominated by generic competition, with price remaining a primary competitive factor.
- Niche Therapeutic Use: Flecainide acetate will likely maintain its position as a valuable treatment option for certain supraventricular tachycardias and specific types of ventricular arrhythmias in patients without underlying structural heart disease. Its role in rhythm control for atrial fibrillation will remain constrained by safety concerns in broader populations compared to newer modalities.
- Opportunities in Emerging Markets: Developing countries with growing healthcare infrastructure and increasing access to diagnostics may present modest growth opportunities as flecainide acetate offers a cost-effective treatment option.
- Potential for Repurposing (Low Probability): While unlikely, any novel research revealing unexpected therapeutic benefits in entirely different disease areas could theoretically open new avenues, but this is speculative for a drug of this age and profile.
- Manufacturing Efficiency as a Differentiator: Manufacturers focusing on optimizing production costs and supply chain reliability will be best positioned to compete in this mature market.
The primary financial trajectory for flecainide acetate will involve sustained, low-volume sales driven by its specific clinical utility, tempered by the ongoing price erosion inherent in the generic pharmaceutical market.
Key Takeaways
- Flecainide acetate operates in a mature, cost-sensitive generic market with a modest annual growth rate of 1.5%.
- The drug's utility is confined to specific cardiac arrhythmias due to significant safety warnings and contraindications.
- The market is fragmented among generic manufacturers, with competition centered on pricing and market access.
- Original patents have expired, with any ongoing patent activity focused on incremental innovations like formulations or manufacturing processes.
- Pricing is highly competitive, and reimbursement is largely determined by formulary placement as a cost-effective generic option.
- The regulatory landscape, particularly safety labeling, significantly restricts broad prescription, preserving flecainide's role in niche therapeutic applications.
- Future market growth is projected to remain low, with opportunities primarily in optimizing production and potentially in emerging markets.
Frequently Asked Questions
-
What are the primary medical conditions treated by flecainide acetate?
Flecainide acetate is primarily used to treat symptomatic supraventricular tachycardias, including atrioventricular nodal reentrant tachycardia and atrial flutter with a rapid ventricular response, as well as symptomatic ventricular arrhythmias, such as ventricular tachycardia, in patients without structural heart disease.
-
What are the main contraindications for prescribing flecainide acetate?
Major contraindications include cardiogenic shock, significant left ventricular dysfunction, recent myocardial infarction, and known hypersensitivity to flecainide. It is also contraindicated in patients with pre-existing second- or third-degree AV block or right bundle-branch block known to be associated with a left ventricular conduction defect.
-
How does flecainide acetate compare in cost to newer antiarrhythmic drugs?
Flecainide acetate is significantly less expensive than many newer antiarrhythmic agents or interventional therapies like catheter ablation. Its generic status ensures a low cost per dose, making it a preferred option from a cost-effectiveness perspective for eligible patients.
-
Are there any upcoming regulatory changes anticipated for flecainide acetate?
No specific upcoming regulatory changes are publicly announced. However, like all pharmaceuticals, flecainide acetate is subject to ongoing pharmacovigilance. Any emerging safety signals or significant adverse event trends could prompt review and potential labeling updates by regulatory bodies.
-
What is the outlook for flecainide acetate in the treatment of atrial fibrillation?
Flecainide acetate's role in atrial fibrillation management is primarily as a rhythm control strategy for patients without significant structural heart disease, particularly those with paroxysmal atrial fibrillation. However, it is not a first-line therapy for stroke prevention and is used cautiously due to potential proarrhythmic effects, especially in patients with underlying cardiac pathology. Newer therapies and catheter ablation are often preferred for broader atrial fibrillation management.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Flecainide Acetate Tablets. Retrieved from www.fda.gov
[2] European Medicines Agency. (n.d.). Flecainide. Retrieved from www.ema.europa.eu
[3] Statista. (2023). Atrial fibrillation prevalence worldwide 2023. Retrieved from www.statista.com
[4] IQVIA. (2023). Global Pharmaceutical Market Analysis. (Proprietary Market Research Report).
[5] U.S. Pharmacists. (2022). Generic Drug Pricing Trends. (Industry Analysis).
[6] United States Patent and Trademark Office (USPTO). (n.d.). Patent Search Database. Retrieved from www.uspto.gov
[7] European Patent Office (EPO). (n.d.). Espacenet Patent Search. Retrieved from www.epo.org