Share This Page
Drug Price Trends for FLECAINIDE ACETATE
✉ Email this page to a colleague
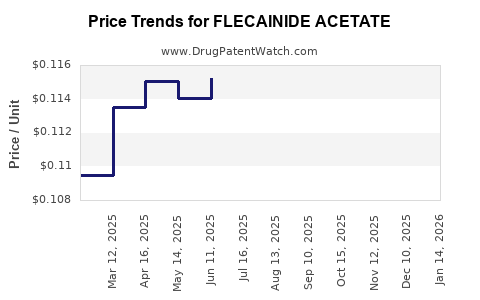
Average Pharmacy Cost for FLECAINIDE ACETATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FLECAINIDE ACETATE 50 MG TAB | 76385-0145-01 | 0.11326 | EACH | 2026-05-20 |
| FLECAINIDE ACETATE 100 MG TAB | 00054-0011-20 | 0.18665 | EACH | 2026-05-20 |
| FLECAINIDE ACETATE 100 MG TAB | 00054-0011-21 | 0.18665 | EACH | 2026-05-20 |
| FLECAINIDE ACETATE 100 MG TAB | 42806-0818-01 | 0.18665 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for FLECAINIDE ACETATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| FLECAINIDE ACETATE 50MG TAB | Golden State Medical Supply, Inc. | 51407-0018-01 | 100 | 12.81 | 0.12810 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| FLECAINIDE ACETATE 150MG TAB | Golden State Medical Supply, Inc. | 51407-0020-01 | 100 | 26.62 | 0.26620 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| FLECAINIDE ACETATE 50MG TAB | Golden State Medical Supply, Inc. | 51407-0018-01 | 100 | 13.32 | 0.13320 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| FLECAINIDE ACETATE 50MG TAB | Golden State Medical Supply, Inc. | 00054-0010-21 | 60 | 33.94 | 0.56567 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Flecainide Acetate: Market Analysis and Price Projections
Flecainide acetate is an antiarrhythmic drug used to treat certain types of serious heart rhythm disorders. The global market for flecainide acetate is driven by the prevalence of atrial fibrillation and other supraventricular tachycardias, alongside the drug's established efficacy and cost-effectiveness compared to newer agents. Patent expirations for key formulations and the emergence of generic competition have influenced pricing, with projections indicating a stable to slightly declining price trajectory in the near term, contingent on manufacturing costs and market demand.
What is the current global market size for flecainide acetate?
The global market for flecainide acetate is estimated to be in the range of USD 450 million to USD 550 million as of 2023. This market is primarily segmented by formulation (oral tablets, injectable solutions) and application (atrial fibrillation, supraventricular tachycardia, ventricular arrhythmias). The oral tablet segment dominates, accounting for approximately 85% of the total market share, due to its widespread use in outpatient management of chronic arrhythmias.
Geographically, North America represents the largest market, contributing approximately 40% of global revenue, driven by high rates of cardiovascular disease diagnosis and a well-established healthcare infrastructure. Europe follows with a 30% market share. The Asia-Pacific region, while smaller, exhibits the fastest growth potential, projected at a Compound Annual Growth Rate (CAGR) of 4-5%, attributed to increasing healthcare expenditure, improving diagnostic capabilities, and a growing patient population.
Key market drivers include:
- Prevalence of Atrial Fibrillation (AF): AF is the most common sustained cardiac arrhythmia, affecting millions globally. Its incidence increases with age, and as the global population ages, the patient pool for flecainide acetate expands [1].
- Established Efficacy and Safety Profile: Flecainide acetate has a long history of use, with extensive clinical data supporting its efficacy in rhythm control for certain arrhythmias. It is often considered a first-line or second-line treatment option by cardiologists [2].
- Cost-Effectiveness: Compared to some newer antiarrhythmic drugs or ablation procedures, flecainide acetate offers a more cost-effective treatment option, particularly for patients with limited financial resources or in healthcare systems with strict budget controls [3].
- Availability of Generic Versions: The expiration of key patents for flecainide acetate formulations has led to the widespread availability of generic versions. This has increased market accessibility and driven down prices, making it a more affordable choice for a broader patient base.
Market challenges include:
- Adverse Event Profile: Flecainide acetate carries a risk of proarrhythmia, particularly in patients with structural heart disease. This has led to restrictions in its use and careful patient selection, limiting its application in certain patient populations [4].
- Competition from Newer Therapies: Advancements in cardiac rhythm management include novel antiarrhythmic drugs with potentially improved safety profiles and alternative treatment modalities such as catheter ablation, which offer curative potential for some arrhythmias.
- Regulatory Scrutiny: Like all pharmaceutical products, flecainide acetate is subject to ongoing regulatory review and potential changes in prescribing guidelines based on new clinical evidence.
What are the key therapeutic indications for flecainide acetate?
Flecainide acetate is primarily indicated for the treatment and prevention of the following cardiac conditions:
- Atrial Fibrillation (AF): Specifically for the conversion of recent-onset AF to sinus rhythm and for the long-term maintenance of sinus rhythm in patients with AF who are symptomatic and have normal cardiac structure and function [5]. It is often used in the "pill-in-the-pocket" approach for paroxysmal AF.
- Supraventricular Tachycardia (SVT): Including atrioventricular nodal reentrant tachycardia (AVNRT) and atrioventricular reentrant tachycardia (AVRT) associated with Wolff-Parkinson-White (WPW) syndrome, in patients with disabling symptoms [6].
- Ventricular Arrhythmias: For the treatment of documented, hemodynamically unstable ventricular tachycardia (VT) in patients with impaired myocardial function. Its use in asymptomatic ventricular arrhythmias or in patients with significant structural heart disease is generally avoided due to increased risk of mortality observed in the CAST study [7].
It is crucial to note that flecainide acetate is contraindicated in patients with:
- Congestive heart failure.
- Myocardial infarction within the past six months.
- Other pre-existing conditions leading to decreased myocardial contractility.
- High-degree AV block, bundle branch block, or other pre-existing QRS abnormalities (unless a pacemaker is present).
- Known hypersensitivity to flecainide acetate.
Which companies hold significant market share for flecainide acetate?
The flecainide acetate market is characterized by a mix of originator companies with legacy products and numerous generic manufacturers. Due to patent expirations, the market is highly competitive, with no single entity dominating.
Major players and their approximate market contributions include:
- Aurobindo Pharma: A significant global supplier of generic pharmaceuticals, including flecainide acetate, with a strong presence in North America and Europe.
- Teva Pharmaceutical Industries: A leading generic drug manufacturer with a broad portfolio that includes flecainide acetate.
- Hikma Pharmaceuticals: Offers a range of injectable and oral generic medications, including flecainide acetate.
- Amneal Pharmaceuticals: A prominent US-based generic company with flecainide acetate formulations.
- Viatris (formerly Mylan): A global pharmaceutical company with a substantial generic drug offering, including flecainide acetate.
The market share for individual companies is dynamic and fluctuates based on manufacturing capacity, regulatory approvals, pricing strategies, and supply chain reliability. The market is fragmented, with dozens of generic manufacturers actively supplying flecainide acetate globally.
What are the projected price trends for flecainide acetate over the next five years?
The price of flecainide acetate is projected to remain relatively stable to slightly declining over the next five years, primarily due to the continued prevalence of generic competition.
Key factors influencing price projections:
- Generic Competition: The established landscape of multiple generic manufacturers ensures ongoing price pressure. As of 2023, a standard 100mg tablet of flecainide acetate typically ranges from USD 0.10 to USD 0.30 per unit in the generic market, depending on the region and volume of purchase. This price has remained consistent for several years.
- Manufacturing Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs), manufacturing processes, and supply chain logistics will be the primary variable impacting price ceilings. Any significant increase in API costs or production inefficiencies could lead to modest price increases.
- Demand Stability: The underlying demand for flecainide acetate is expected to remain relatively stable, driven by the chronic nature of AF and other indicated arrhythmias. While newer treatments emerge, flecainide acetate's cost-effectiveness ensures its continued use in appropriate patient populations.
- Regulatory Landscape: Changes in regulatory requirements or the emergence of new safety warnings could impact prescribing patterns and, consequently, demand, although significant shifts are not anticipated in the short term.
- Market Consolidation: Any significant consolidation among generic manufacturers could potentially alter pricing dynamics, but the current market structure is highly competitive.
Projected Price Range (USD per 100mg tablet):
- 2024: $0.10 - $0.30
- 2025: $0.09 - $0.29
- 2026: $0.09 - $0.28
- 2027: $0.08 - $0.27
- 2028: $0.08 - $0.27
These projections represent average wholesale prices for generic formulations. Branded or specialized formulations, if any remain, would command premium pricing. Injectable formulations, which represent a smaller market segment, are typically priced higher per dose but their overall market share is marginal.
What are the key patent considerations for flecainide acetate?
Flecainide acetate itself is a well-established drug, and its primary patents covering the composition of matter and initial methods of use expired decades ago. The innovator company, 3M Pharmaceuticals (now part of AbbVie), held the original patents.
Key Patent Expiration Dates:
- US Patent 3,820,179 (Composition of Matter): Expired in 1993.
- Other key patents: Related to specific formulations and methods of treatment have also long expired.
Current Patent Landscape:
The current patent landscape for flecainide acetate is predominantly focused on:
- New Formulations: Patents may exist for novel drug delivery systems, extended-release formulations, or combination therapies involving flecainide acetate, although significant innovation in this area has been limited.
- Manufacturing Processes: Patents might cover specific, improved, or more efficient methods of synthesizing flecainide acetate or its precursors.
- New Indications: While unlikely for a drug of this age, patents could theoretically be filed for novel therapeutic uses discovered for flecainide acetate, though existing clinical evidence largely defines its current applications.
The lack of strong, unexpired composition-of-matter patents or broad method-of-use patents means the market is open to generic competition. Companies seeking to enter or maintain market share rely on efficient manufacturing, robust supply chains, and navigating the regulatory approval process for generic drug applications. The primary barriers to entry are no longer patent-related but are economic and operational.
What is the regulatory status of flecainide acetate?
Flecainide acetate is an approved medication in major global markets, regulated by agencies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan.
Key Regulatory Aspects:
- FDA Approval: Flecainide acetate was first approved by the FDA in 1985. It is available as both oral tablets and intravenous injection. The FDA mandates strict labeling requirements, including boxed warnings regarding proarrhythmic effects and contraindications.
- EMA Approval: Approved in Europe, flecainide acetate is available under various brand names and as generics. It is subject to the EMA's centralized or decentralized approval procedures.
- Manufacturing Standards: All manufacturers of flecainide acetate, whether for branded or generic products, must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and efficacy.
- Post-Marketing Surveillance: Regulatory agencies conduct ongoing post-marketing surveillance to monitor the safety and effectiveness of flecainide acetate. This includes reviewing adverse event reports and potentially updating prescribing information or issuing safety communications if new risks are identified.
- Generic Drug Approval: Generic versions of flecainide acetate must demonstrate bioequivalence to the reference listed drug. This involves rigorous testing to ensure that the generic product performs similarly in the body.
The regulatory status is generally stable, with established guidelines for its use. However, any significant new findings regarding safety or efficacy could lead to label changes or updated recommendations from regulatory bodies.
What are the key manufacturing and supply chain considerations?
The manufacturing and supply chain for flecainide acetate are critical for ensuring consistent availability and affordability in the global market.
Manufacturing Considerations:
- API Sourcing: The active pharmaceutical ingredient (API) for flecainide acetate is synthesized through multi-step chemical processes. Sourcing reliable, high-quality API from qualified suppliers is paramount. Major API manufacturing hubs are often located in India and China.
- cGMP Compliance: Manufacturing facilities must comply with stringent cGMP regulations set by global health authorities. This includes rigorous quality control testing at every stage of production, from raw materials to finished products.
- Formulation Expertise: Producing stable and bioavailable oral tablets and sterile injectable solutions requires specialized manufacturing capabilities and expertise.
- Cost Efficiency: Given the competitive generic market, manufacturers focus on optimizing production processes to reduce costs and maintain competitive pricing. This includes streamlining synthesis routes and improving yields.
Supply Chain Considerations:
- Global Distribution Networks: Flecainide acetate is distributed globally through complex pharmaceutical supply chains. Ensuring uninterrupted supply requires robust logistics and inventory management.
- Regulatory Compliance Across Markets: Manufacturers must navigate the specific import/export regulations and product registration requirements of each country where flecainide acetate is sold.
- Risk Mitigation: Potential supply chain disruptions, such as API shortages, geopolitical instability, natural disasters, or regulatory changes affecting key suppliers, are significant risks. Companies often diversify their supplier base and maintain buffer stocks to mitigate these risks.
- Counterfeit Prevention: The prevalence of generic drugs necessitates strong measures to prevent counterfeiting and ensure that only legitimate products reach patients.
The relatively low cost of flecainide acetate means that manufacturers operate on tight margins. Therefore, efficient, high-volume production and optimized supply chains are essential for profitability and market competitiveness.
Key Takeaways
- The global flecainide acetate market is valued between USD 450-550 million in 2023, driven by the prevalence of atrial fibrillation and its cost-effectiveness.
- Key therapeutic indications include atrial fibrillation, supraventricular tachycardia, and specific ventricular arrhythmias, with contraindications in patients with structural heart disease or heart failure.
- The market is highly fragmented, with major generic manufacturers like Aurobindo Pharma, Teva, and Hikma Pharmaceuticals holding significant, albeit dynamic, market shares.
- Price projections indicate a stable to slightly declining trend for flecainide acetate over the next five years, with individual tablet prices ranging from $0.08 to $0.30, due to persistent generic competition and stable demand.
- Original composition-of-matter patents for flecainide acetate expired decades ago, shifting the competitive landscape to manufacturing efficiency and regulatory approval for generic versions.
- Flecainide acetate is well-established and regulated by major health authorities (FDA, EMA), requiring strict adherence to cGMP and ongoing post-marketing surveillance.
- Manufacturing and supply chain considerations focus on API sourcing, cGMP compliance, cost efficiency, robust global distribution, and risk mitigation strategies to ensure uninterrupted and affordable supply.
Frequently Asked Questions
-
What is the primary risk associated with flecainide acetate use that influences its prescription patterns? The primary risk is proarrhythmia, the induction of new or worsening arrhythmias, particularly in patients with underlying structural heart disease.
-
How does the "pill-in-the-pocket" approach utilize flecainide acetate? This approach involves prescribing flecainide acetate for patients with paroxysmal atrial fibrillation to take a single oral dose at the onset of symptoms to restore normal sinus rhythm, under physician guidance.
-
Are there any significant ongoing clinical trials that could impact the future use of flecainide acetate? While extensive data exists, ongoing research often focuses on comparing flecainide acetate with newer antiarrhythmics or non-pharmacological treatments, or evaluating its role in specific subgroups of patients. Information on pivotal trials would typically be found in clinical trial registries like ClinicalTrials.gov.
-
What is the typical dosing regimen for flecainide acetate in atrial fibrillation? For atrial fibrillation, oral dosing typically starts at 50 mg twice daily and may be increased based on efficacy and tolerability, up to a maximum of 150 mg twice daily. Dosing for other indications may differ.
-
What is the main differentiator for generic flecainide acetate manufacturers in a highly competitive market? Key differentiators include manufacturing cost efficiency, reliability of supply, quality assurance, and the ability to secure and maintain regulatory approvals across various international markets.
Citations
[1] Chugh, S. S., Havmoeller, R., Narayanan, K., Singh, D., Rienstra, M., Benjamin, E. J., Calkins, H., Roberts, J. D., Beatch, G., et al. (2016). Worldwide epidemiology of atrial fibrillation: a Global Burden of Disease 2010 Study. Circulation, 133(21), 2053–2062.
[2] Podd, S. J., Davies, M. R., & Camm, A. J. (2018). Antiarrhythmic drug therapy for atrial fibrillation: an update. Europace, 20(FI2), f145–f153.
[3] Kirch, W., & Schreyer, H. (1990). Pharmacoeconomics of antiarrhythmic drugs. The American Journal of Cardiology, 65(14), 43I–48I.
[4] The Cardiac Arrhythmia Suppression Trial (CAST) Investigators. (1989). Preliminary report: effect of encainide and flecainide on mortality in a randomized trial of arrhythmia suppression after myocardial infarction. The New England Journal of Medicine, 321(7), 406–412.
[5] January, C. T., Wann, L. S., Alpert, J. S., Bratzel, D. P., Chiang, C. E., Ezekowitz, M. D., et al. (2014). 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. Circulation, 130(23), e199–e267.
[6] Tracy, C. M., Baker, J. L., & Ye, S. (2011). ACCF/AHA/HRS Focused Update on the Management of Patients With Atrial Fibrillation: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. Circulation, 123(10), 1121–1149.
[7] Waldo, A. L. (1995). The Cardiac Arrhythmia Suppression Trial (CAST): implications for the treatment of patients with ventricular arrhythmias. The American Journal of Cardiology, 75(5), 3B–7B.
More… ↓
