Last updated: January 9, 2026
Summary
Epinephrine, also known as adrenaline, is a critical drug in emergency medicine, primarily used for anaphylactic shock, cardiac arrest, and local vasoconstriction during anesthesia. Its global market is driven by rising allergy prevalence, advancements in delivery systems, and expanding indications. Despite intense patent expirations and generic competition, the drug’s essential status ensures sustained demand, supported by regulatory developments and corporate strategies. This analysis explores epinephrine’s market forces, financial projections, competitive landscape, regulatory influences, and future growth trajectories.
What Are the Core Market Drivers for Epinephrine?
1. Rising Incidence of Anaphylaxis and Allergic Diseases
- The global prevalence of food, insect sting, and drug allergies is increasing, propelling demand for epinephrine auto-injectors.
- Statistics:
- US adults: ~7.6% report food allergies (FAO report, 2021).
- Pediatric populations: up to 8% experience food allergies (CDC, 2020).
- Impact: Growth in acute allergy emergencies elevates emergency medication needs, especially in North America and Europe.
2. Expanded Indications and Approvals
- While traditionally used for anaphylaxis and cardiac arrest, recent off-label research and approvals explore expanding uses (e.g., trauma, bronchodilation).
- Novel formulations (e.g., autoinjectors, inhalers) improve portability and administration speed.
3. Regulatory Initiatives and Guidelines
- The FDA and EMA promote auto-injector availability, with inclusion in emergency kits.
- FDA’s guidance (2018): Emphasizes improving access to epinephrine in schools and public spaces.
- Global Impact: Policy support elevates demand and investment.
4. Scientific and Technological Advancements
- Innovations in delivery systems (auto-injectors, pre-filled syringes, novel formulations) boost market appeal.
- Market Penetration: Auto-injectors constitute ~70% of global sales, reflecting consumer preference for ease of use.
What Are the Key Market Segments?
| Segment |
Characteristics |
Market Share (Estimated) |
Growth Drivers |
| Pharmaceutical Manufacturers |
Large pharma, generics, biosimilar entrants |
55% |
Patent expirations, cost offers |
| Auto-injectors & Devices |
Epinephrine auto-injectors (EpiPen, Auvi-Q) |
70% |
Convenience, safety, user preference |
| Injectable Formulations |
Vials for hospital use |
30% |
Emergency supply, hospital settings |
| Emerging Delivery Systems |
Inhalers, nasal sprays |
Niche |
Innovation potential |
Note: Market share estimations subject to change with regulatory shifts and manufacturing trends.
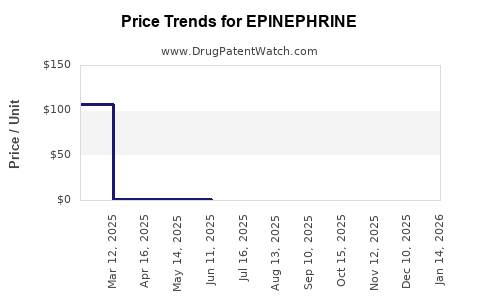
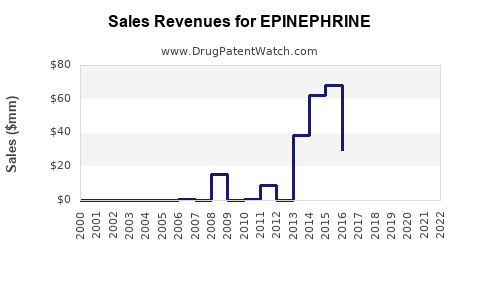
How Do Patent Expirations and Generic Competition Influence the Market?
Patent Landscape and Market Erosion
- EpiPen (by Mylan, now part of Viatris) held dominant market share but faced patent expirations:
- US patent lost in 2017.
- Increased generic entry led to price erosion.
- Impact: Price drops by up to 80% post-generic entry, impacting revenues for originating manufacturers.
Market Share Redistribution
| Year |
Patent Status |
Key Events |
Market Share Impact |
| 2016-2018 |
Patent expiration for EpiPen |
Launch of generics in US |
Revenue decline for original patent holder |
| 2019+ |
Multiple generics available |
Increased price competition |
Market consolidation, upward price trend for brands with differentiation |
Strategies to Counteract Patent Losses
- Development of new formulations (e.g., alternative delivery systems).
- Expanding indications and geographical markets.
- Acquiring or licensing new patents or trademarks targeting improved safety or convenience.
What Are the Key Regulatory and Policy Influences?
Global Regulatory Environment
| Region |
Regulatory Body |
Recent Initiatives |
Effect on Market |
| USA |
FDA (Food and Drug Administration) |
REMS (Risk Evaluation and Mitigation Strategies), auto-injector guidelines |
Increased safety protocols, new approvals |
| EU |
EMA (European Medicines Agency) |
EMA's review of auto-injector safety |
Facilitates approval of novel devices |
| Asia-Pacific |
Local agencies (e.g., China NMPA) |
Market entry support |
Growing access in emerging markets |
Governmental Policies
- Enhanced access programs (e.g., School Access Law, US National Allergy Strategy) increase the distribution of epinephrine.
- Subsidies and insurance coverage improve affordability, boosting demand.
What Is the Competitive Landscape?
Major Players and Their Market Shares
| Company |
Key Products |
Market Share |
Strategic Moves |
| Mylan/Viatris |
EpiPen, brand auto-injectors |
~65% (pre-2017) |
Diversification, price reductions |
| Sanofi |
Auvi-Q |
Niche player |
Focus on innovative delivery systems |
| Pfizer |
Adrenalin (generic auto-injector) |
Growing |
Generics market entry |
| Teva, Kelstar |
Generic epinephrine auto-injectors |
Increasing |
Cost competition |
Emerging Competitors and Innovation Trends
- Startup firms introducing smarter auto-injector designs.
- Developments in inhalable epinephrine for easier administration.
- Biosimilar versions reducing costs.
What Are Future Market Growth Projections?
Market Size and Forecasts (2022–2027)
| Year |
Estimated Market Size ($ Millions) |
CAGR (%) |
Description |
| 2022 |
$1,200 |
— |
Baseline |
| 2027 |
$1,800 |
8% |
Driven by rising allergy prevalence, device innovation |
Key Drivers of Growth
- Increasing allergy prevalence leading to higher demand.
- Device innovation, making administration safer and user-friendly.
- Regulatory support expanding auto-injectors into new indications.
- Market expansion into emerging economies with rising healthcare infrastructure.
Challenges and Risks
- Patent cliffs creating price pressures.
- Stringent regulations delaying or complicating new product approvals.
- Competitive commoditization with generics further reducing margins.
- Supply chain vulnerabilities, especially for auto-injector components.
How Do Technological Innovations Shape Market Trajectory?
Smart Auto-injectors and Digital Features
- Bluetooth-enabled devices for dose tracking.
- Enhanced safety features (e.g., needle lock, dose confirmation).
- Potential for integration with digital health records to improve patient compliance.
Alternative Delivery Methods
- Inhalation: Research into inhalable epinephrine formulations.
- Nasal sprays: Under development, offering needle-free administration.
Impact on Market Dynamics
- These innovations could command premium pricing.
- Enhance market penetration and patient adherence.
- Potentially open new indications and applications.
FAQs
Q1: How significant are patent expirations for epinephrine auto-injectors on the market?
A: Very. The expiration of key patents (notably EpiPen in 2017) led to a surge of generic competitors, significantly reducing prices and changing market share dynamics.
Q2: What regional factors influence epinephrine demand?
A: Developed countries with higher allergy prevalence and better healthcare infrastructure drive demand. Emerging markets are expanding due to increased awareness and policy support.
Q3: Are there promising innovative delivery systems for epinephrine?
A: Yes. Smart auto-injectors, nasal sprays, and inhalation formulations are under development, aiming to enhance safety, ease of use, and expand indications.
Q4: How do regulatory policies affect epinephrine’s market trajectory?
A: Policies promoting auto-injector availability, safety standards, and emergency access programs support market growth, especially in North America and Europe.
Q5: What is the outlook for epinephrine market revenues over the next five years?
A: The market is projected to grow at a CAGR of approximately 8%, reaching $1.8 billion by 2027, driven by innovation, expanding indications, and global health initiatives.
Key Takeaways
- Market Growth: Currently valued around $1.2 billion globally, with an estimated CAGR of 8% (2022–2027).
- Drivers: Rising allergy incidences, technological innovations, regulatory support, and expanded indications.
- Challenges: Patent expirations, price erosion, intense competition from generics, regulatory hurdles.
- Innovation Opportunities: Smart auto-injectors, needle-free formulations, inhalables.
- Strategic Focus: Investment in R&D, diversification of delivery systems, geographic expansion, and navigating regulatory landscapes.
References
- FAO (Food and Agriculture Organization). (2021). "Global Food Allergy Statistics."
- CDC (Centers for Disease Control and Prevention). (2020). "Food Allergies in US Children."
- FDA (2018). "Guidance on Auto-Injectors for Anaphylaxis."
- MarketsandMarkets. (2022). "Epinephrine Market Forecast to 2027."
- European Medicines Agency. (2021). "Regulatory Updates on Auto-Injectors."
Disclaimer: This article is for informational purposes and does not constitute financial advice.