ENTRESTO Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Entresto, and what generic alternatives are available?
Entresto is a drug marketed by Novartis Pharms Corp and Novartis and is included in two NDAs. There are twelve patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and sixty-three patent family members in forty-four countries.
The generic ingredient in ENTRESTO is sacubitril; valsartan. There are eleven drug master file entries for this compound. Two suppliers are listed for this compound. Additional details are available on the sacubitril; valsartan profile page.
DrugPatentWatch® Generic Entry Outlook for Entresto
Entresto was eligible for patent challenges on July 7, 2019.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be May 9, 2036. This may change due to patent challenges or generic licensing.
There have been eleven patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
Summary for ENTRESTO
| International Patents: | 163 |
| US Patents: | 8 |
| Applicants: | 2 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 13 |
| Clinical Trials: | 40 |
| Patent Applications: | 22 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for ENTRESTO |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ENTRESTO |
| What excipients (inactive ingredients) are in ENTRESTO? | ENTRESTO excipients list |
| DailyMed Link: | ENTRESTO at DailyMed |
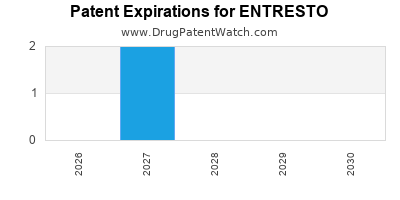
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ENTRESTO
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for ENTRESTO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Instituto Nacional de Cardiologia Ignacio Chavez | Phase 2/Phase 3 |
| Boehringer Ingelheim laboratory | Phase 2/Phase 3 |
| Brigham and Women's Hospital | Phase 2 |
Pharmacology for ENTRESTO
| Drug Class | Angiotensin 2 Receptor Blocker Neprilysin Inhibitor |
| Mechanism of Action | Angiotensin 2 Receptor Antagonists Neprilysin Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for ENTRESTO
Paragraph IV (Patent) Challenges for ENTRESTO
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ENTRESTO | Tablets | sacubitril; valsartan | 24 mg/26 mg, 49 mg/51 mg 97 mg/103 mg | 207620 | 18 | 2019-07-08 |
US Patents and Regulatory Information for ENTRESTO
ENTRESTO is protected by eight US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ENTRESTO is ⤷ Sign Up.
This potential generic entry date is based on patent ⤷ Sign Up.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting ENTRESTO
Sacubitril-valsartan dosage regimen for treating heart failure
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATING CHRONIC HEART FAILURE WITH REDUCED EJECTION FRACTION IN PATIENTS NOT TAKING AN ACE INHIBITOR OR AN ARB OR PREVIOUSLY TAKING LOW DOSES OF THESE AGENTS, BY TITRATING UP FROM HALF THE USUALLY RECOMMENDED STARTING DOSE
Inhibitors for treating diseases characterized by atrial enlargement or remodeling
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF HEART FAILURE WITH PRESERVED EJECTION FRACTION
Methods of treatment and pharmaceutical composition
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Methods of treatment and pharmaceutical composition
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Compounds containing S-N-valeryl-N-{[2'-(1H-tetrazole-5-yl)-biphenyl-4-yl]-methyl}-valine and (2R,4S)-5-biphenyl-4-yl-4-(3-carboxy-propionylamino)-2-methyl-pentanoic acid ethyl ester moieties and cations
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Compounds containing S-N-valeryl-N-{[2'-(1H-tetrazole-5-yl)-biphenyl-4-yl]-methyl)-valine and (2R,4S)-5-biphenyl-4-yl-4-(3-carboxy-propionylamino)-2-methyl-pentanoic acid ethyl ester moieties and cations
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Inhibitors for treating diseases characterized by atrial enlargement or remodeling
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF HEART FAILURE WITH PRESERVED EJECTION FRACTION
Inhibitors for treating diseases characterized by atrial enlargement or remodeling
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF HEART FAILURE WITH PRESERVED EJECTION FRACTION
FDA Regulatory Exclusivity protecting ENTRESTO
LABELING REVISIONS RELATED TO CLINICAL STUDIES
Exclusivity Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-002 | Apr 12, 2024 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-001 | Apr 12, 2024 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-002 | Apr 12, 2024 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-001 | Apr 12, 2024 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Novartis Pharms Corp | ENTRESTO | sacubitril; valsartan | TABLET;ORAL | 207620-001 | Jul 7, 2015 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Novartis Pharms Corp | ENTRESTO | sacubitril; valsartan | TABLET;ORAL | 207620-002 | Jul 7, 2015 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for ENTRESTO
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Novartis Europharm Limited | Neparvis | sacubitril, valsartan | EMEA/H/C/004343 Paediatric heart failureNeparvis is indicated in children and adolescents aged one year or older for treatment of symptomatic chronic heart failure with left ventricular systolic dysfunction (see section 5.1).Adult heart failureNeparvis is indicated in adult patients for treatment of symptomatic chronic heart failure with reduced ejection fraction (see section 5.1). |
Authorised | no | no | no | 2016-05-26 | |
| Novartis Europharm Limited | Entresto | sacubitril, valsartan | EMEA/H/C/004062 Paediatric heart failureEntresto is indicated in children and adolescents aged one year or older for treatment of symptomatic chronic heart failure with left ventricular systolic dysfunction.Adult heart failureEntresto is indicated in adult patients for treatment of symptomatic chronic heart failure with reduced ejection fraction. |
Authorised | no | no | no | 2015-11-19 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for ENTRESTO
When does loss-of-exclusivity occur for ENTRESTO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Croatia
Patent: 0230480
Estimated Expiration: ⤷ Sign Up
Cyprus
Patent: 26036
Estimated Expiration: ⤷ Sign Up
Denmark
Patent: 94283
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 94283
Estimated Expiration: ⤷ Sign Up
Patent: 12152
Estimated Expiration: ⤷ Sign Up
Finland
Patent: 94283
Estimated Expiration: ⤷ Sign Up
Hungary
Patent: 62195
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 76469
Estimated Expiration: ⤷ Sign Up
Patent: 18519266
Estimated Expiration: ⤷ Sign Up
Lithuania
Patent: 94283
Estimated Expiration: ⤷ Sign Up
Poland
Patent: 94283
Estimated Expiration: ⤷ Sign Up
Portugal
Patent: 94283
Estimated Expiration: ⤷ Sign Up
Serbia
Patent: 242
Estimated Expiration: ⤷ Sign Up
Slovenia
Patent: 94283
Estimated Expiration: ⤷ Sign Up
Spain
Patent: 45866
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ENTRESTO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Poland | 2887961 | ⤷ Sign Up | |
| South Korea | 102159601 | ⤷ Sign Up | |
| Brazil | 112015003067 | inibidores de nep para tratar doenças caracterizadas por alargamento ou remodelagem atrial | ⤷ Sign Up |
| European Patent Office | 1467728 | COMPOSITIONS PHARMACEUTIQUES A BASE DE VALSARTAN ET D'INHIBITEURS NEP (PHARMACEUTICAL COMPOSITIONS COMPRISING VALSARTAN AND NEP INHIBITORS) | ⤷ Sign Up |
| Canada | 2590511 | COMBINAISON PHARMACEUTIQUES D'UN ANTAGONISTE DE RECEPTEUR D'ANGIOTENSINE ET D'UN INHIBITEUR DE NEP (PHARMACEUTICAL COMBINATIONS OF AN ANGIOTENSIN RECEPTOR ANTAGONIST AND AN NEP INHIBITOR) | ⤷ Sign Up |
| China | 104602711 | Nep inhibitors for treating diseases characterized by atrial enlargement or remodeling | ⤷ Sign Up |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ENTRESTO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2340828 | PA2021502 | Lithuania | ⤷ Sign Up | PRODUCT NAME: SAKUBITRILAS/VALSARTANAS, KAIP SAKUBITRILO VALSARTANO NATRIO DRUSKOS KOMPLEKSAS, T.Y. (((S)-N-VALERIL-N-((2'-(1H-TETRAZOL-5-IL)-BIFENIL-4-IL)-METIL)-VALINAS)((2R,4S)-5-BIFENIL-4-IL-4-(3-KARBOKSI-PROPIONILAMINO)-2-METIL-PENTANO RUGSTIES ETILO ESTERIS)NA3; REGISTRATION NO/DATE: EU/1/15/1058 20151119 |
| 1467728 | SPC/GB16/025 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: SACUBITRIL/VALSARTAN, INCLUDING PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF; REGISTERED: UK EU/1/15/1058(NI) 20151123; UK PLGB 00101/1041 20151123; UK PLGB 00101/1042 20151123; UK PLGB 00101/1043 20151123 |
| 1948158 | 2016C/026 | Belgium | ⤷ Sign Up | PRODUCT NAME: SACUBITRIL/VALSARTAN, ALS SACUBITRIL VALSARTAN NATRIUMZOUT COMPLEX, I.E. TRINATRIUM (3-((1S,3R)-1-BIFENYL-4-YLMETHYL-3-ETHOXYCARBONIL-1-BUTYLCARBAMOYL)PROPIONAAT-(S)-3'-METHYL-2'-(PENTANOYL(2-(TETRAZOL-5-YLATE)BIFENYL-4'-YLMETHYL)AMINO)BUTYRAAT)HEMIPENTAHYDRAAT; AUTHORISATION NUMBER AND DATE: EU/1/15/1058 20151123 |
| 1467728 | 93074 | Luxembourg | ⤷ Sign Up | PRODUCT NAME: SACUBITRIL/VALSARTAN, Y COMPRIS LES SELS PHARMACEUTIQUEMENT ACCEPTABLES DE CELUI-CI; FIRST REGISTRATION DATE: 20151123 |
| 2340828 | CA 2021 00001 | Denmark | ⤷ Sign Up | PRODUCT NAME: SACUBITRIL/VALSARTAN, SOM SACUBITRILVALSARTAN-NATRIUMSALTKOMPLEKS, DVS. ...; REG. NO/DATE: EU/1/15/1058 20151123 |
| 2340828 | 2/2021 | Austria | ⤷ Sign Up | PRODUCT NAME: SACUBITRIL/VALSARTAN ALS SACUBITRIL-VALSARTAN-NATRIUMSALZ-KOMPLEX, D.H. (((S)-N-VALERYL-N-((2'-(1H-TETRAZOL-5-YL)BIPHENYL-4-YL)METHYL)-VALIN) ((2R,4S)-5-BIPHENYL-4-YL-4-(3-CARBOXY-PROPIONYLAMINO)-2-METHYL-PENTANSAEUREETHYLESTER))NA3 ? X H2O, WOBEI X FUER 0 BIS 3 STEHT; REGISTRATION NO/DATE: EU/1/15/1058 (MITTEILUNG) 20151123 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


