CELECOXIB Drug Patent Profile
✉ Email this page to a colleague
When do Celecoxib patents expire, and when can generic versions of Celecoxib launch?
Celecoxib is a drug marketed by Alembic, Amneal Pharms, Apotex, Aurobindo Pharma, Cadila Pharms Ltd, Cipla, Cspc Ouyi, Jubilant Generics, Lupin Ltd, Macleods Pharms Ltd, Micro Labs, Nanjing, Pangea, Pharmobedient, Qingdao Baheal Pharm, Sciegen Pharms, Strides Pharma, Teva, Tianjin Tianyao, Torrent, Umedica, Unichem, Watson Labs Inc, Yabao Pharm, Yiling, and Zydus Pharms. and is included in twenty-six NDAs.
The generic ingredient in CELECOXIB is celecoxib. There are twenty-six drug master file entries for this compound. Sixty-five suppliers are listed for this compound. Additional details are available on the celecoxib profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Celecoxib
A generic version of CELECOXIB was approved as celecoxib by TEVA on May 30th, 2014.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for CELECOXIB?
- What are the global sales for CELECOXIB?
- What is Average Wholesale Price for CELECOXIB?
Summary for CELECOXIB
| US Patents: | 0 |
| Applicants: | 26 |
| NDAs: | 26 |
| Finished Product Suppliers / Packagers: | 62 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 606 |
| Patent Applications: | 4,728 |
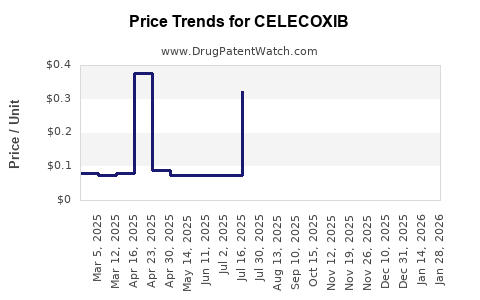
| Drug Prices: | Drug price information for CELECOXIB |
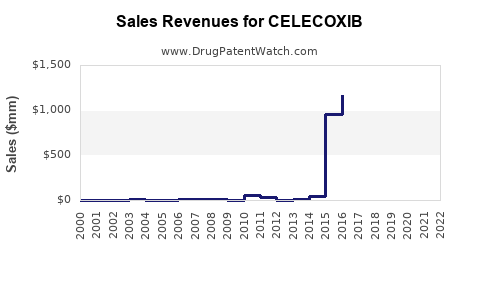
| Drug Sales Revenues: | Drug sales revenues for CELECOXIB |
| What excipients (inactive ingredients) are in CELECOXIB? | CELECOXIB excipients list |
| DailyMed Link: | CELECOXIB at DailyMed |
Recent Clinical Trials for CELECOXIB
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of California, Los Angeles | PHASE4 |
| Myopharm Limited | PHASE2 |
| Beijing Tiantan Hospital | NA |
Pharmacology for CELECOXIB
| Drug Class | Nonsteroidal Anti-inflammatory Drug |
| Mechanism of Action | Cyclooxygenase Inhibitors |
Medical Subject Heading (MeSH) Categories for CELECOXIB
Anatomical Therapeutic Chemical (ATC) Classes for CELECOXIB
US Patents and Regulatory Information for CELECOXIB
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Torrent | CELECOXIB | celecoxib | CAPSULE;ORAL | 207677-004 | Dec 23, 2015 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Micro Labs | CELECOXIB | celecoxib | CAPSULE;ORAL | 204776-003 | Apr 30, 2018 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Lupin Ltd | CELECOXIB | celecoxib | CAPSULE;ORAL | 202240-002 | Jun 9, 2015 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Yabao Pharm | CELECOXIB | celecoxib | CAPSULE;ORAL | 212564-002 | Apr 10, 2023 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Pharmobedient | CELECOXIB | celecoxib | CAPSULE;ORAL | 078857-004 | Feb 11, 2015 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Strides Pharma | CELECOXIB | celecoxib | CAPSULE;ORAL | 213127-003 | Apr 29, 2025 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for CELECOXIB
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Pfizer Limited | Onsenal | celecoxib | EMEA/H/C/000466Onsenal is indicated for the reduction of the number of adenomatous intestinal polyps in familial adenomatous polyposis (FAP), as an adjunct to surgery and further endoscopic surveillance (see section 4.4).The effect of Onsenal-induced reduction of polyp burden on the risk of intestinal cancer has not been demonstrated (see sections 4.4 and 5.1) | Withdrawn | no | no | no | 2003-10-17 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
Market Dynamics and Financial Trajectory for CELECOXIB
More… ↓