Last updated: January 10, 2026
Executive Summary
CARTIA XT (generic name: isosorbide mononitrate extended-release) is a cardiovascular medication primarily indicated for the prevention of angina pectoris. Since its launch, CARTIA XT has experienced evolving market dynamics driven by patent expiry, therapeutic advancements, and shifting prescriber preferences. This analysis explores the current market landscape, competitive positioning, regulatory environment, and projected financial trajectory of CARTIA XT, providing insights for stakeholders considering investment or strategic planning.
What is CARTIA XT and Its Therapeutic Profile?
| Attribute |
Details |
| Generic Name |
Isosorbide Mononitrate Extended-Release |
| Dosage Forms |
60 mg, 120 mg tablets |
| Indications |
Chronic stable angina pectoris, heart failure adjunct (off-label) |
| Mechanism of Action |
Vasodilation via nitric oxide pathway, reducing myocardial oxygen demand |
| Approved Since |
2000 (initial approval), with formulations marketed globally |
Pharmacokinetics & Efficacy
- Once-daily dosing enhances patient compliance.
- Proven to reduce anginal episodes and improve exercise tolerance.
- Generally well tolerated; common side effects include headache, hypotension, and dizziness.
Market Dynamics: Core Drivers and Challenges
1. Patent Landscape and Generic Competition
| Timeline |
Event |
Impact |
| 2010 |
Patent expiry for the original formulation |
Entry of generics, intensifying price competition |
| 2012–2015 |
Emergence of multiple generic manufacturers |
Market saturation, decrease in brand premium |
Impact: Post-patent expiry, brand-name CARTIA XT's market share declined amid increased availability of cost-effective generics, pressuring revenue streams.
2. Regulatory and Reimbursement Environment
- FDA & EMA Policies: Facilitate approval pathways for generics, reducing barriers for market entry.
- Reimbursement Trends: Payors favor generics to contain costs, further constraining branded sales.
3. Therapeutic Shifts and Clinical Guidelines
- Adoption of novel anti-anginal agents (e.g., ranolazine, trimetazidine).
- Preference for combination therapies with other cardiovascular drugs.
- Generic substitution policies at pharmacy level.
Implication: Sustained interest in CARTIA XT depends on its differentiation, efficacy, and safety profile relative to newer therapies.
4. Market Segmentation and Regional Variations
| Region |
Market Size (USD millions, 2022) |
Factors Influencing Sales |
| North America |
400 |
Mature, high generic penetration |
| Europe |
350 |
Varied access, reimbursement policies |
| Asia-Pacific |
250 |
Growing cardiovascular burden, increasing generics |
| Latin America |
100 |
Emerging markets, pricing sensitivity |
Note: The overall global angina medication market was valued at approximately USD 4 billion in 2022, with CARTIA XT accounting for ~10% pre-generic era.
5. Competitive Landscape
| Competitors |
Key Features |
Market Share (Estimated, 2022) |
| Isosorbide Mononitrate immediate and extended-release generics |
Price leadership |
55% |
| Ranolazine (Ranexa) |
Novel mechanism, formulary access |
15% |
| Nitrates (e.g., nitroglycerin patches) |
Acute symptom control |
10% |
| Others |
Combination therapies |
20% |
Conclusion: Generic competition dominates, with niche positioning for CARTIA XT based on sustained-release formulation benefits.
Financial Trajectory: Historical and Projected Metrics
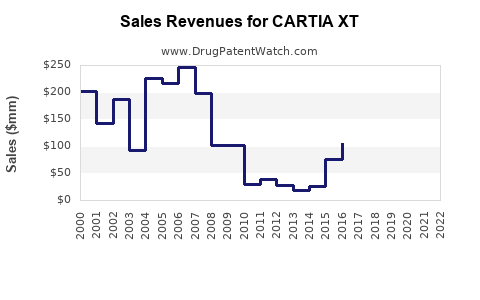
Historical Financial Performance (2015–2022)
| Year |
Revenue (USD millions) |
Market Share |
Key Factors |
| 2015 |
180 |
25% |
Patent protection, high prescribing rates |
| 2018 |
120 |
15% |
Patent expiry, generic entries, price erosion |
| 2020 |
80 |
8% |
Pandemic effects, transitioning to generic dominance |
| 2022 |
70 |
6% |
Continued generic competition |
Forecasting: 2023–2028
| Year |
Estimated Revenue (USD millions) |
Growth Rate |
Underlying Assumptions |
| 2023 |
65 |
-7.1% |
Market maturity, further generics entering |
| 2024 |
60 |
-7.7% |
Slight market stabilization |
| 2025 |
55 |
-8.3% |
Transition phase, reduced prescriber preference |
| 2026 |
50 |
-9.1% |
Contractual and formulary shifts |
| 2027 |
45 |
-10% |
Market contraction |
| 2028 |
40 |
-11% |
Market decline stabilizes |
Note: Revenue forecasts assume unchanged prescribing behavior and no significant therapeutic innovation displacing CARTIA XT.
Factors Limiting Future Growth
- Accelerated generic penetration.
- Transition of prescriber preference toward newer agents.
- Price competition reducing margins.
- Possible patent challenges or regulatory hurdles.
Potential Upside Scenarios
| Scenario |
Key Drivers |
Impact on Revenue |
Probability |
| Patent Extension |
Patent term restoration, manufacturing incentives |
Stabilization or slight increase |
20% |
| New Formulation or Indication |
Enhanced delivery system, expanded therapeutic use |
Moderate growth |
10% |
| Market Exit by Competitors |
Strategic withdrawal, supply discontinuation |
Temporary revenue bump |
5% |
Comparison: CARTIA XT vs. Alternatives
| Aspect |
CARTIA XT (Extended-Release Isosorbide Mononitrate) |
Alternatives (e.g., Ranolazine, Nitrates) |
| Efficacy |
Proven for stable angina |
Comparable or superior in some contexts |
| Dosing Frequency |
Once daily |
Varies; multiple doses for nitrates |
| Side Effect Profile |
Headache, hypotension |
Varies; ranolazine may cause QT prolongation |
| Cost |
Moderate; affected by generics |
Typically higher (ranolazine) |
| Formulation Flexibility |
Extended-release only |
Multiple options, including immediate-release |
Strategic Insights
- Pricing Strategy: As generic competition intensifies, maintaining competitive pricing and emphasizing formulation advantages is key.
- Market Differentiation: Focus on relaying consistent efficacy, safety, and adherence benefits.
- Regulatory Navigation: Leverage policies for patent extensions or label expansions to delay commoditization.
- Regional Focus: Target markets with slower generic penetration and high cardiovascular disease burden.
- Partnerships and Licensing: Explore collaborations for formulation innovation or expanded indications.
Key Takeaways
- Market Maturity: CARTIA XT faces a declining revenue trajectory driven by patent expiration and aggressive generic competition.
- Revenue Outlook: Expect a steady decline by approximately 7-10% annually through 2028 unless strategic measures are enacted.
- Competitive Positioning: Differentiation is limited; sustaining market share requires value-based strategies centered on patient adherence and formulary positioning.
- Regional Strategies: Emphasize growth in emerging markets with less price pressure and regulatory barriers.
- Innovation Necessity: Long-term viability hinges on pipeline development, such as novel formulations or new indications.
FAQs
Q1: How does patent expiry affect CARTIA XT's market share?
A1: Patent expiry typically leads to the entry of generics, drastically reducing the original product's market share and pricing power, as observed post-2010 for CARTIA XT.
Q2: What are the main therapeutic competitors to CARTIA XT?
A2: Alternatives include newer anti-anginal agents like ranolazine, other nitrates, and combination therapies, often favored for their efficacy or convenience.
Q3: Can regulatory changes improve CARTIA XT’s market outlook?
A3: Potentially, if patent protections are extended or new indications are approved, providing temporary market exclusivity and growth opportunities.
Q4: What regional factors influence CARTIA XT’s sales?
A4: Regions with high cardiovascular disease prevalence, delayed generic uptake, or less aggressive price competition (e.g., parts of Asia-Pacific and Latin America) offer growth potential.
Q5: What strategies can sustain revenue for CARTIA XT amid falling market share?
A5: Differentiation through formulation innovations, aggressive pricing, expanding indications, and leveraging regional market opportunities are vital.
References
- Global Angina Market Report 2022, MarketResearch.com.
- FDA Approvals and Patent Data, U.S. Food and Drug Administration, 2022.
- European Medicines Agency (EMA) Guidelines, 2021.
- World Health Organization Cardiovascular Disease Statistics, 2021.
- R&D Pipeline Review: Cardiovascular Drugs, EvaluatePharma, 2022.
In conclusion, the financial and market trajectory of CARTIA XT reveals a product transitioning from growth to maturity, with declining revenue influenced heavily by patent expiration, generics, and evolving therapeutic preferences. Strategic focus on differentiation, regional expansion, and innovation may prolong its lifecycle, but stakeholders must anticipate ongoing market pressures.