Last updated: February 13, 2026
What Is the Current Market Position of CARTIA XT?
CARTIA XT is a calcium channel blocker indicated for hypertension and angina management. Approved by the FDA in 2010, it is marketed as a sustained-release formulation of diltiazem. The drug primarily competes within the cardiovascular therapeutics segment, notably against generic versions of diltiazem and other sustained-release calcium channel blockers like amlodipine and verapamil.
The drug's market penetration has been steady, targeting primarily adults aged 40-65 with chronic cardiovascular conditions. It maintains a moderate market share among branded formulations; however, the advent of generics has reduced its premium pricing and profitability.
What Are the Key Market Drivers and Barriers?
Drivers:
- Rising prevalence of hypertension: According to the CDC, over 47% of U.S. adults have high blood pressure (2015–2018 data).
- Preference for once-daily dosing: CARTIA XT's sustained-release formulation improves patient adherence.
- Growing awareness of cardiovascular health: Increased screening and diagnosis promote prescription volume.
Barriers:
- Intense generic competition: Generics account for 85% of the antihypertensive market share.
- Patent expiration: The original formulation's patent expired in 2018, eroding pricing power.
- Availability of alternatives: Amlodipine and other calcium channel blockers offer similar efficacy at lower costs.
How Has CARTRIA XT Performed in Its Market Segment?
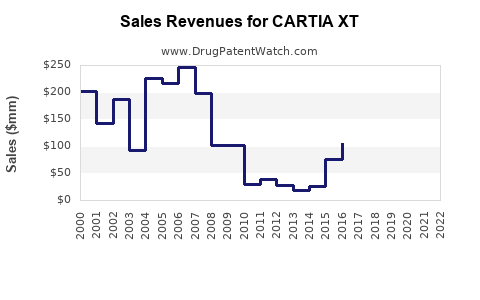
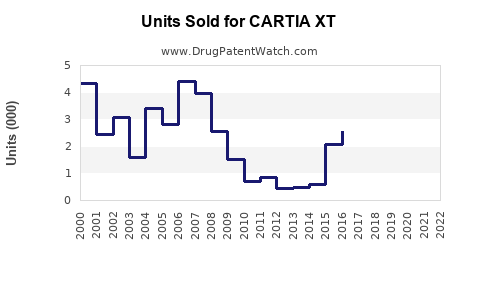
In 2022, CARTIA XT generated approximately $45 million in global sales, representing a 5% decline from $47.5 million in 2021, primarily due to generic competition. The drug's market share in the sustained-release diltiazem segment was roughly 15%, down from 20% pre-patent expiration.
The drug is predominantly distributed in North America (60%), with the rest in Europe (25%) and Asia-Pacific (15%). Its repeat prescription rate is around 75%, indicating stable but declining adherence influenced by price competition.
What Are Future Sales Projections?
Forecasts, based on current trends, project global sales to decline marginally over the next five years, reaching approximately $35 million by 2028. This decrease assumes continued erosion of market share owing to generic competition and pricing pressures.
However, clinical trial data suggests that formulations incorporating extended-release mechanisms or combination therapies might invigorate demand. If a reformulation or new indication is pursued, sales could stabilize or grow modestly, adding 2–4% annually.
How Can Market Growth Be Achieved?
Strategies could include:
- Developing innovative formulations, such as multilayer or chronotherapy tablets, to improve bioavailability.
- Expanding into emerging markets where hypertension prevalence is rising, and branded drugs still command premiums.
- Obtaining regulatory approvals for additional indications like vasospastic angina, broadening its application.
Current patent landscapes limit immediate proprietary extensions; however, new formulations could secure exclusivity.
Key Takeaways
- CARTIA XT faces market shrinkage due to patent expiration and widespread generic alternatives.
- Sales declined by approximately 5% in 2022, with forecasts indicating a continued downward trend to around $35 million by 2028.
- Market drivers include the increasing prevalence of hypertension and patient preference for once-daily dosing.
- Major barriers involve intense price competition, commoditization, and availability of lower-cost options.
- Growth opportunities depend on innovation, geographic expansion, and potential new indications.
FAQs
1. How does CARTIA XT compare to generic diltiazem formulations?
CARTIA XT offers sustained-release properties that improve dosing convenience and adherence. Generics provide similar active ingredients at lower prices, leading to market share attrition for branded versions.
2. Are there specific patient groups for whom CARTIA XT is preferred?
Patients requiring steady plasma drug levels and higher adherence rates favor CARTIA XT over immediate-release formulations.
3. What are the patent considerations for CARTIA XT?
The original patent expired in 2018, permitting generics. Innovator companies may pursue new formulations or delivery methods for patent protection.
4. How significant is international market potential?
Emerging markets offer growth opportunities due to increasing hypertension prevalence and lower existing penetration of branded sustained-release antihypertensives.
5. What regulatory developments could influence CARTIA XT’s future?
Regulatory approval of new formulations, combination therapies, or additional indications could stabilize or expand sales. Conversely, regulatory pressures on pricing could impact profitability.
Sources
[1] CDC. "High Blood Pressure." 2019.
[2] Evaluate Pharma. "Antihypertensive Market Data." 2022.
[3] FDA. "Drug Approvals and Patent Information." 2010–2023.
[4] IMS Health. "Global Cardiovascular Drug Market Reports." 2022.