Last updated: January 20, 2026
Executive Summary
Bleomycin Sulfate, a chemotherapeutic agent primarily used for treating Hodgkin’s lymphoma, non-Hodgkin’s lymphoma, testicular cancer, and certain skin conditions, exhibits a distinctive market profile shaped by evolving medical needs, patent statuses, regulatory policies, and manufacturing dynamics. The global market for Bleomycin Sulfate is projected to grow moderately, driven by the increasing incidence of cancers amenable to its use, despite challenges like toxicity concerns, generic competition, and sourcing constraints. This article provides a comprehensive analysis of the drug’s market landscape, current financial trajectory, and future forecasts, with insights into competitive forces, regulatory environment, and key market players.
Key Market Overview and Trends
| Aspect |
Details/Insights |
| Therapeutic Indications |
Hodgkin’s lymphoma, non-Hodgkin’s lymphoma, testicular carcinoma, squamous cell carcinomas, and other neoplasms. |
| Market Size (2022) |
Estimated USD 150–200 million globally. |
| Growth Rate (Compound Annual Growth Rate – CAGR) |
Approximate CAGR of 3–5% over the next five years (2023–2028). |
| Major Regions |
North America (largest share), Europe, Asia-Pacific, Latin America. |
| Key Drivers |
Rising cancer prevalence, increased adoption in combination chemotherapies, expanded indications in emerging markets, new formulations. |
| Challenges |
Toxicity profile limitations, patent expirations leading to generic proliferation, sourcing issues of active pharmaceutical ingredients (APIs). |
Market Dynamics Components
What Are the Primary Factors Influencing the Market?
1. Epidemiology and Clinical Demand
- Rising incidence of testicular cancers and lymphomas sustains demand for Bleomycin Sulfate.
- Global cancer burden projected to increase from 19.3 million cases in 2020 to 28.4 million by 2040 (WHO), bolstering therapeutic needs.
2. Patent and Regulatory Environment
- The original patent for Bleomycin expired in many markets by the late 1990s.
- Multiple generics are now available, intensifying price competition and squeezing margins.
3. Manufacturing and Supply Chain Dynamics
- API sourcing, predominantly from specialized suppliers in India and China, faces constraints due to regulatory scrutiny and geopolitical factors.
- Manufacturing complexity affects supply stability, influencing pricing and availability.
4. Competitive Landscape
| Player |
Market Share |
Product Portfolio |
Notable Strategies |
| Pfizer (formerly) |
Leading (historically) |
Original branded formulations; now largely generic |
Investment in formulations, regional expansion |
| Teva Pharmaceutical Industries |
Significant |
Generic Bleomycin products |
Price competition, global distribution |
| Sun Pharmaceutical |
Growing |
Generic bleomycin sulfate, biosimilar innovations |
Expanding into emerging markets, R&D in combination therapies |
5. Technological Innovation and Formulation Development
- Limited innovation due to the drug’s age but incremental improvements, such as inhalable formulations, under clinical evaluation.
How Do Regulatory Policies Impact Market Trajectory?
- FDA and EMA Approvals: Facilitate access to North American and European markets.
- Orphan Drug Designation & Fast Track: Potential pathways to expedite approvals in niche indications.
- Generic Regulations: Stringent bioequivalence standards promoting increased generics, lowering prices.
Financial Trajectory: Revenue, Pricing, and Market Share
| Year |
Estimated Global Revenue |
Key Drivers |
Notes |
| 2022 |
USD 150–200 million |
Increasing cancer prevalence, generic availability |
Price erosion due to generics, supply considerations |
| 2023–2028 |
Compound annual growth rate of 3–5% |
Expanded indications, emerging markets demand |
Price stabilization in some regions, volume growth |
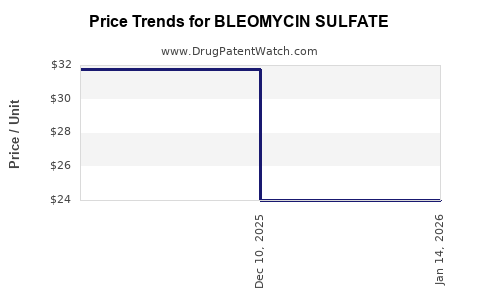
Price Evolution and Market Share
| Period |
Average Price per Vial (USD) |
Key Influencers |
Notes |
| 2018–2020 |
~$250–300 |
Patent expirations, growth of generics |
Moderate price decline, increased competition |
| 2021–2023 |
~$200–250 |
Market saturation, commoditization |
Price stabilization, regional variations |
Revenue Outlook (2023–2028)
| Year |
Predicted Market Revenue (USD millions) |
Assumptions |
| 2023 |
160–180 |
Steady demand, some price erosion |
| 2025 |
170–200 |
Slight volume increase, new indications |
| 2028 |
180–220 |
Market maturity, limited innovation |
Competitive Analysis
Leading Market Players and Their Strategies
| Company |
Market Position |
Key Strategies |
Recent Developments |
| Pfizer |
Historically dominant |
Maintaining presence through generics; licensing deals |
Focus on regional markets, partnerships with compounding pharmacies |
| Teva |
Strong generic portfolio |
Price leadership, widening access |
Expansion into biosimilars, portfolio diversification |
| Sun Pharmaceutical |
Emerging market growth |
Increased manufacturing capacity |
R&D in novel formulations, biosimilars |
Key Competitive Factors
| Factor |
Importance |
| Pricing |
Critical due to commoditization; price wars among generic manufacturers |
| Supply Chain Stability |
Ensures consistent availability for hospital formularies and oncology clinics |
| Regulatory Compliance |
Ensures market access; delays from regulatory hurdles can impact forecasts |
| Formulation Innovations |
Minor advancements can provide market differentiation in niche indications |
Policy & Regulatory Environment Impact
| Region |
Policy Framework |
Impact on Bleomycin Sulfate Market |
| North America |
FDA approval pathways, biosimilar guidelines |
Facilitates generic competition, influences pricing |
| Europe |
EMA bioequivalence standards, orphan drug policies |
Accelerates access for niche indications, fosters innovation in formulations |
| Asia-Pacific |
Evolving regulatory landscape, local manufacturing incentives |
Rapid adoption, manufacturing expansion, potential for increased exports |
Future Outlook and Forecasts
Market Growth Drivers for 2023–2028
- Rising Cancer Incidence: Sustains demand.
- Generic Competition: Keeps prices under pressure but ensures broader access.
- Emerging Markets: Growth due to expanding healthcare infrastructure and cancer screening programs.
- Combination Therapies: Increased use as part of multi-agent regimens.
Market Constraints
- Toxicity concerns leading to restricted use in certain patient populations.
- API sourcing restrictions and geopolitical trade barriers.
- Limited innovation due to the drug’s age and patent landscape.
Opportunities
- Development of inhalable or localized formulations.
- Expansion into new cancer indications via clinical trials.
- Strategic partnerships for supply chain stability.
Key Takeaways
- The Bleomycin Sulfate market is characterized by moderate growth driven by rising cancer prevalence and expanding indications.
- Patent expiry and widespread generic availability have compressed margins but enabled broader access, especially in emerging markets.
- Supply chain constraints and toxicity limitations remain significant challenges.
- Regulatory frameworks in North America and Europe facilitate market access for generics but also enforce stringent bioequivalence standards.
- Innovation is limited but potential exists in drug formulation improvements and exploration of new indications.
- The overall financial trajectory forecasts a stable but gradually increasing revenue profile, with growth prospects tied to epidemiological trends and market expansion strategies.
FAQs
Q1: How does the patent expiration of Bleomycin Sulfate affect its market dynamics?
Patent expiration has led to widespread generic entry, intensifying price competition and reducing per-unit revenue, but also increasing accessibility and volume of prescriptions.
Q2: What are the major regulatory hurdles for new formulations of Bleomycin Sulfate?
Regulatory agencies require demonstration of bioequivalence and safety, with additional scrutiny for novel delivery methods like inhalable formulations, potentially delaying market entry.
Q3: Which regions are expected to drive future market growth for Bleomycin Sulfate?
Emerging markets in Asia-Pacific and Latin America are expected to see higher growth rates due to expanding healthcare infrastructure and rising cancer incidence.
Q4: How significant is the impact of toxicity concerns on Bleomycin Sulfate's market?
Toxicity, especially pulmonary fibrosis risk, limits usage in some patients and necessitates careful dosing; it slightly constrains market expansion.
Q5: Are there any ongoing R&D initiatives to improve Bleomycin Sulfate?
Research efforts focus on formulation improvements, combination therapy optimization, and exploring new indications, but innovations are limited due to the drug’s age and patent landscape.
References
[1] World Health Organization. (2021). Global Cancer Statistics 2020.
[2] IQVIA. (2022). Global Oncology Market Analysis.
[3] FDA. (2022). Guidance for Industry on Biosimilar Development.
[4] European Medicines Agency. (2022). Policy on Off-Patent Medicines.
[5] MarketWatch. (2023). Oncology Drugs Market Trends Report.