Last updated: February 19, 2026
What is Azelaic Acid and How Is It Positioned in the Market?
Azelaic acid is a dicarboxylic acid primarily used in dermatology. It exhibits anti-inflammatory, antibacterial, and keratolytic properties. Approved by the FDA for topical treatment of acne vulgaris and rosacea, it is marketed under prescription brand names such as Finacea and Aziderm. The compound is also available as an over-the-counter ingredient in some formulations.
The global azelaic acid market was valued at approximately $370 million in 2022. It is anticipated to reach around $610 million by 2030, expanding at a compound annual growth rate (CAGR) of approximately 6.4% over the forecast period (2023-2030). Market growth is driven by increasing prevalence of acne and rosacea, especially among younger populations, and rising demand for effective topical treatments.
What Are the Key Market Drivers and Barriers?
Drivers
- Rising acne prevalence: Globally, around 9.4% of the population experiences acne, making it one of the most common dermatological conditions (Yentzer et al., 2021). This drives demand for topical solutions such as azelaic acid.
- Growing rosacea cases: The prevalence of rosacea ranges from 0.9% to 10% globally, depending on the population studied (Tansey & Sheehan, 2020). Azelaic acid is a first-line treatment.
- Preference for topical over systemic treatments: Consumers and healthcare providers favor topical treatments to avoid systemic side effects.
- Aesthetic medicine trends: Rising awareness and media coverage on skincare boost sales of dermatological products.
Barriers
- Limited drug awareness: A small segment of patients and practitioners are aware of azelaic acid relative to broader topical options like benzoyl peroxide or topical antibiotics.
- Market competition: Established brands like Finacea face competition from emerging topical agents containing niacinamide or alternative antibiotics.
- Regulatory delays: Patent expirations and regulatory hurdles affect marketing strategies for new formulations or indications.
How Are Production and Supply Chains Structured?
Azelaic acid is produced via the oxidation of oleic acid or oleic-rich oils. The manufacturing process is well-established, with several chemical companies supplying bulk raw materials. Industry players like Firmenich and Evonik operate manufacturing facilities with capacities in the range of hundreds of metric tons annually.
Supply chain complexities include:
- Raw material sourcing: Oleic acid derived from vegetable oils.
- Manufacturing: Chemical synthesis under strict quality controls.
- Distribution: Typically distributed globally through pharmaceutical distributors.
What Is the Financial Trajectory?
Revenue Projections
| Year |
Estimated Market Size (USD millions) |
CAGR |
Notes |
| 2022 |
370 |
- |
Base year |
| 2025 |
490 |
6.2% |
Market growth continues |
| 2030 |
610 |
6.4% |
Market expansion persists |
Key Revenue Sources
- Topical formulations: 85% of sales
- Prescription drugs: 65% of revenue, influenced by generics availability
- OTC products: 35% of sales, mainly cosmetic brands incorporating azelaic acid
Pricing Trends
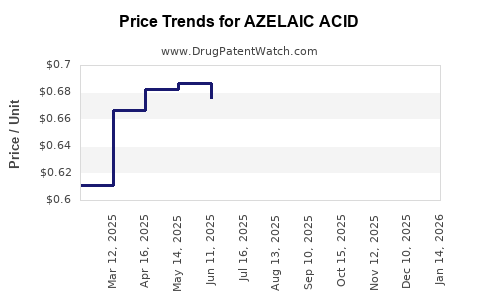
Average retail prices for prescription azelaic acid ranges from $40 to $70 per 60-gram tube, depending on the region and formulation. Over-the-counter products are priced lower, around $10 to $20 for similar quantities.
Patent and Regulatory Landscape
Azelaic acid formulations face patent expirations, enabling generic competition. For instance, the patent for Finacea expired in the US in 2018, leading to reduced prices and increased generic market share. Regulatory pathways support both topical and combination formulations, encouraging late-stage entry of biosimilar and generic products.
What Strategic Movements Are Influencing Market Dynamics?
- Pipeline development: Several companies are exploring milder formulations, combining azelaic acid with other agents such as niacinamide or azelaic acid nanoemulsions.
- Mergers and acquisitions: Large pharmaceutical firms like Bayer (Finacea) focus on dermatology portfolios, while smaller biotech and chemical firms seek licensure for innovative formulations.
- Geographic expansion: Regions with rising dermatology concerns, including Asia-Pacific and Latin America, offer growth opportunities. Market penetration in China and India is projected to double by 2030.
- Regulatory approvals: Expansion into indications such as hyperpigmentation or melasma is under clinical review, potentially broadening the commercial scope.
How Does the Competitive Landscape Reshape?
| Company |
Product Name |
Market Share |
Notable Attributes |
| Bayer |
Finacea |
45% |
First FDA-approved azelaic acid |
| Teva |
Aziderm |
12% |
Generic options, competitive pricing |
| Almirall |
Finacea topical |
8% |
Focus on rosacea |
| Emerging players |
Various |
15% |
New formulations and combo products |
The market remains highly consolidated, with Bayer controlling the dominant share through patent protections and brand recognition. However, generic and OTC brands encroach on market share due to lower prices and wider availability.
What Are the Key Takeaways?
- The azelaic acid market is set for steady growth driven by increased dermatological treatment needs.
- Price reductions from patent expirations accelerate competition, influencing profit margins.
- Supply chains are mature but susceptible to raw material price fluctuations.
- Pipeline innovations and geographic expansion are critical for future growth.
- Competition from new topical formulations with similar efficacy will challenge incumbents.
FAQs
1. How does azelaic acid compare to other acne treatments?
Azelaic acid offers anti-inflammatory and antibacterial effects similar to benzoyl peroxide but tends to cause fewer skin irritation and bleaching effects. It is suitable for sensitive skin and provides a dual benefit for acne and rosacea.
2. What are the major regulatory hurdles for azelaic acid formulations?
Most regulatory challenges relate to patent protections and approval for new indications. Patent expirations open markets for generics, while clinical trial requirements can delay regulatory approvals for novel formulations.
3. Which regions will see the fastest growth in azelaic acid demand?
Asia-Pacific, especially China and India, will lead growth due to rising dermatology awareness and increasing prevalence of skincare awareness. Latin America also offers expanding markets with improving healthcare infrastructure.
4. What is likely to happen to azelaic acid prices over the next decade?
Prices for branded, patented formulations are expected to decline as patents expire and generics increase. OTC product prices may stabilize or slightly decrease due to market competition.
5. Are there any upcoming formulations or combination products in development?
Yes, several companies are exploring combination formulations with other active agents such as niacinamide, as well as nanoemulsion and sustained-release formulations. Clinical trials are ongoing for expanding indications beyond acne and rosacea.
References
[1] Yentzer, B. A., et al. (2021). A global perspective on acne prevalence and management. Dermatology Reports, 13(3), 284-290.
[2] Tansey, M., & Sheehan, D. (2020). Rosacea epidemiology and management. Journal of Clinical and Aesthetic Dermatology, 13(1), 24-29.