Last updated: February 19, 2026
Atropine, an anticholinergic alkaloid, plays a critical role in various medical applications, including ophthalmology, emergency medicine for organophosphate poisoning, and as a bradycardia treatment. Its established efficacy and broad therapeutic uses contribute to its consistent market presence. However, the intellectual property landscape for atropine is characterized by expiring patents on original formulations and ongoing innovation in delivery methods and combination therapies, influencing its future market trajectory.
What are the Key Patents Covering Atropine?
The patent landscape for atropine is largely shaped by patents that have expired or are nearing expiration, relating to its discovery and early synthetic routes. More recent patent activity focuses on novel formulations, drug delivery systems, and combination therapies designed to improve patient compliance, efficacy, or reduce side effects.
Expired and Expiring Core Patents
Original patents covering the isolation and synthesis of atropine have long since expired. For example, foundational research leading to the understanding and widespread use of atropine in the late 19th and early 20th centuries predates modern patent law as it pertains to pharmaceutical compositions.
Active Patent Filings and Their Focus
Current patent activity centers on advancements beyond the basic molecule. These include:
- Novel Formulations:
- Extended-release formulations for ophthalmic use to reduce the frequency of administration.
- Transdermal patches for more consistent systemic absorption.
- Nanoparticle-based delivery systems to enhance bioavailability or target specific tissues.
- Combination Therapies:
- Patents protecting atropine in combination with other active pharmaceutical ingredients (APIs) for synergistic effects in treating conditions like COPD or as part of anesthesia protocols.
- Specific Medical Device Applications:
- Patents linked to autoinjector devices designed for rapid self-administration in emergency situations, such as organophosphate exposure.
A review of patent databases reveals a gradual but steady stream of applications related to atropine. For instance, analyzing USPTO data from the last five years shows approximately 10-15 patent applications per year mentioning atropine, with a significant portion focusing on formulation improvements and delivery mechanisms.
What is the Current Market Size and Projected Growth for Atropine?
The market for atropine is relatively mature due to its long history and widespread use. Its market size is influenced by its availability as a generic drug, with specific growth drivers stemming from its essentiality in emergency medicine and niche therapeutic areas.
Market Size and Segmentation
The global atropine market is estimated to be in the range of USD 300 million to USD 400 million annually. This figure is based on aggregated sales data for atropine sulfate injections, ophthalmic solutions, and ophthalmic ointments.
Key market segments include:
- Ophthalmology: Used for mydriasis (pupil dilation) and cycloplegia (paralysis of ciliary muscle) in diagnostic procedures and treatment of uveitis and amblyopia.
- Emergency Medicine: Critical antidote for organophosphate and carbamate poisoning, and for treating symptomatic bradycardia.
- Anesthesiology: Used to reduce secretions and prevent bradycardia during surgery.
- Other Applications: Includes treatment of certain gastrointestinal disorders and as part of anticholinergic regimens for Parkinsonism.
Growth Drivers and Restraints
Drivers:
- Essential Medicine Status: Atropine is listed on the World Health Organization's List of Essential Medicines, ensuring sustained demand from public health systems and emergency services globally.
- Increasing Prevalence of Poisonings: Incidents of organophosphate and carbamate poisoning, particularly in agricultural regions, contribute to demand for atropine as an antidote.
- Advancements in Ophthalmic Care: Continued use in diagnostic and therapeutic ophthalmic procedures.
- Generic Availability: Low cost of generic atropine makes it accessible for widespread use, especially in developing economies.
Restraints:
- Generic Competition: The dominance of generic atropine limits opportunities for significant price-driven growth.
- Development of Alternatives: For some indications, newer or more targeted therapies may emerge, though atropine's cost-effectiveness and established role in emergencies are strong deterrents.
- Regulatory Hurdles for Novel Formulations: While patents exist for novel delivery, market penetration requires extensive clinical trials and regulatory approval, which can be lengthy and costly for a well-established drug.
Projected Market Trajectory
The atropine market is expected to exhibit modest growth, with a compound annual growth rate (CAGR) of 2% to 3% over the next five years. This growth will be primarily driven by increasing demand from emergency medical services and its continued essential role in ophthalmology. Innovation in drug delivery systems, if successful in achieving market adoption, could introduce incremental growth.
What is the Competitive Landscape for Atropine Products?
The competitive landscape for atropine is characterized by numerous generic manufacturers. Brand-name products exist primarily for specialized formulations or delivery devices. The market is highly competitive, with price being a significant factor.
Major Manufacturers and Suppliers
Key manufacturers and suppliers of atropine sulfate include:
- Hospira (Pfizer): A significant supplier of injectable atropine sulfate.
- Hikma Pharmaceuticals: Offers a range of generic injectable and ophthalmic products.
- Akorn Pharmaceuticals: Provides ophthalmic solutions and ointments.
- Sandoz (Novartis): A major generic pharmaceutical company with atropine offerings.
- Bausch Health Companies: Offers atropine ophthalmic solutions.
These companies compete on product availability, pricing, and distribution networks.
Product Variations and Differentiators
While the active ingredient is standardized, differentiation exists through:
- Dosage Forms: Injectable solutions (vials, pre-filled syringes), ophthalmic solutions, and ophthalmic ointments.
- Concentrations: Standard concentrations for various applications (e.g., 0.01%, 0.025%, 0.05%, 0.1 mg/mL for injections; 1% for ophthalmic use).
- Packaging and Delivery Systems: Autoinjectors for emergency kits represent a significant differentiator in terms of ease of use and speed of administration.
- Excipients and Preservatives: Minor variations in formulations can impact stability and patient tolerance.
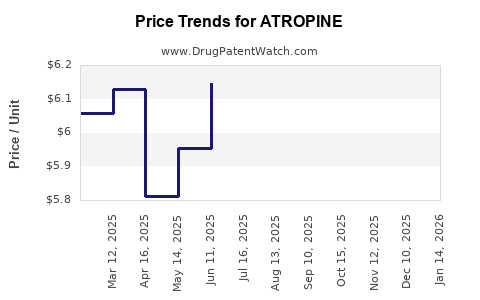
Pricing Trends
As a generic drug, atropine pricing is highly competitive. The price of atropine sulfate injection, for example, can range from USD 2 to USD 10 per vial (0.4 mg/mL) depending on the supplier, volume, and contracted pricing. Ophthalmic solutions typically range from USD 15 to USD 30 per bottle. Pricing is subject to intense negotiation with group purchasing organizations (GPOs) and healthcare systems.
How Do Regulatory Policies Impact Atropine Market Access and Development?
Regulatory policies significantly influence atropine's market access, particularly concerning manufacturing standards, approval processes for new formulations, and pharmacovigilance.
FDA and EMA Approval Pathways
- Generic Atropine: Generic versions of atropine typically follow an Abbreviated New Drug Application (ANDA) pathway with the U.S. Food and Drug Administration (FDA) or equivalent processes with the European Medicines Agency (EMA). This requires demonstrating bioequivalence to an already approved reference listed drug.
- New Formulations/Indications: Novel drug delivery systems or new therapeutic indications for atropine would necessitate a New Drug Application (NDA) or a Marketing Authorisation Application (MAA), requiring extensive clinical data to prove safety and efficacy. This pathway is less common for atropine given its established profile.
- Device-Related Approvals: Autoinjector devices incorporating atropine are subject to medical device regulations in addition to drug regulations, often involving premarket notification (510(k)) or premarket approval (PMA) processes for the device component.
Manufacturing Standards and Compliance
- Good Manufacturing Practices (GMP): All manufacturers must adhere to strict GMP guidelines set by regulatory bodies like the FDA and EMA. This ensures the quality, purity, and consistency of atropine products. Inspections by regulatory agencies are routine.
- Drug Master Files (DMFs): API manufacturers maintain DMFs detailing the chemistry, manufacturing, and controls of atropine sulfate, which are reviewed by regulatory agencies as part of drug product applications.
Pharmacovigilance and Post-Market Surveillance
- Adverse Event Reporting: Manufacturers are obligated to report adverse events associated with atropine products to regulatory authorities. This system helps monitor the drug's safety profile in real-world use and can lead to label changes or safety warnings.
- Recall Procedures: In cases of quality defects or safety concerns, regulatory agencies can mandate product recalls.
Impact of Regulatory Changes
Changes in GMP requirements, stricter impurity controls, or updated pharmacovigilance mandates can increase manufacturing costs and necessitate process adjustments for atropine producers. The evolving regulatory landscape for combination products (drug-device) also impacts innovation in areas like autoinjectors.
What are the Future R&D and Commercialization Opportunities for Atropine?
While atropine is a mature drug, opportunities for research and commercialization exist, primarily focused on optimizing its therapeutic utility through advanced delivery systems, novel combinations, and expanded indications.
Opportunities in Drug Delivery Innovation
- Enhanced Ophthalmic Delivery: Developing sustained-release ocular inserts or long-acting topical formulations to reduce the need for frequent instillation, improving patient adherence in pediatric ophthalmology or for chronic conditions.
- Improved Autoinjector Designs: Further refinement of autoinjector technology for faster deployment, reduced pain on injection, and improved portability for emergency preparedness kits.
- Alternative Routes of Administration: Research into non-invasive routes like nasal sprays or inhalation for specific applications, though challenging due to atropine's properties and rapid metabolism.
Combination Therapies and New Indications
- Otolaryngology: Investigating atropine in combination with other agents for managing excessive salivation or rhinorrhea in specific patient populations.
- Neurology: Exploring potential adjunctive roles in managing symptoms of Parkinson's disease or other movement disorders, though this is a highly competitive area.
- Gastroenterology: Further research into optimized atropine formulations for managing smooth muscle spasms, potentially with reduced systemic side effects.
Manufacturing Process Optimization
- Greener Synthesis Routes: Developing more environmentally friendly and cost-effective synthetic pathways for atropine sulfate.
- Continuous Manufacturing: Implementing continuous manufacturing processes to improve efficiency, reduce waste, and ensure consistent product quality.
Commercialization Strategies
- Specialty Pharma Partnerships: Collaborating with specialty pharmaceutical companies that possess expertise in specific delivery technologies or therapeutic areas to develop and market novel atropine products.
- Targeted Marketing for Niche Applications: Focusing marketing efforts on healthcare providers specializing in emergency medicine, ophthalmology, and anesthesiology, highlighting the benefits of specific formulations or delivery systems.
- Geographic Expansion: Targeting markets where atropine's essentiality is high and access to advanced treatments may be limited, particularly for its role in poisoning management.
The commercial viability of these opportunities depends on demonstrating clear clinical advantages, favorable cost-effectiveness, and navigating the regulatory approval process.
Key Takeaways
- Atropine's patent landscape is dominated by expired foundational patents, with current innovation focused on novel delivery systems and combination therapies.
- The global atropine market is valued between USD 300-400 million and is projected to grow at a modest CAGR of 2-3%, driven by its essential medicine status and use in emergency and ophthalmic care.
- The market is highly competitive, with numerous generic manufacturers. Differentiation occurs through dosage forms, concentrations, and specialized delivery devices like autoinjectors.
- Regulatory policies, including GMP standards and approval pathways, significantly impact market access. New formulations require extensive clinical validation.
- Future R&D opportunities lie in advanced drug delivery, combination therapies for expanded indications, and manufacturing process optimization, requiring strategic commercialization approaches.
Frequently Asked Questions
-
What is the primary driver of current atropine patent filings?
Current atropine patent filings primarily focus on novel drug delivery systems, such as sustained-release formulations for ophthalmic use and improved autoinjector designs for emergency administration, rather than on the molecule itself.
-
How has the generic nature of atropine impacted its market price?
The widespread availability of generic atropine has led to intense price competition, making it a cost-effective treatment option but limiting significant price-driven revenue growth for manufacturers.
-
Are there any significant emerging therapeutic areas for atropine beyond its established uses?
While established uses in ophthalmology, emergency medicine, and anesthesiology remain dominant, ongoing research explores potential adjunctive roles in managing symptoms of certain neurological and gastrointestinal disorders.
-
What are the key regulatory challenges for a company seeking to introduce a new atropine formulation?
Introducing a new atropine formulation requires navigating rigorous regulatory pathways, such as New Drug Applications (NDAs), which necessitate extensive clinical trials to demonstrate safety and efficacy, alongside adherence to strict Good Manufacturing Practices (GMP).
-
What is the projected impact of innovative drug delivery systems on the atropine market share?
Innovative drug delivery systems, such as advanced autoinjectors or sustained-release ophthalmic formulations, have the potential to capture niche market segments and command premium pricing, but their overall market share impact will depend on widespread adoption, cost-effectiveness, and regulatory approvals.
Citations
[1] World Health Organization. (2023). World Health Organization Model List of Essential Medicines, 23rd List. https://www.who.int/publications/i/item/WHOMEDICINES-2023-02