Last updated: February 19, 2026
This report analyzes the market dynamics and financial trajectory of ABSORICA LD, a prescription medication. It examines patent exclusivity, market competition, pricing, and sales performance to forecast its future financial performance.
What is ABSORICA LD's Current Patent Status and Exclusivity Landscape?
ABSOLICA LD (Isotretinoin) is a brand-name prescription medication used for the treatment of severe recalcitrant nodular acne. The active pharmaceutical ingredient, isotretinoin, has a long history of use and has been off-patent for many years as a generic compound. However, specific formulations and delivery systems can be protected by patents.
According to publicly available patent databases, the original patents protecting the broad use and composition of isotretinoin have expired. For example, U.S. Patent No. 4,272,422, which covers the use of isotretinoin in treating keratinization disorders, expired in 2002.
However, ABSORICA LD is a specific formulation developed by Bausch Health Companies Inc. (formerly Valeant Pharmaceuticals International, Inc.). The "LD" in ABSORICA LD stands for "Lipid Dispersion," indicating a proprietary formulation designed to improve the bioavailability and tolerability of isotretinoin. This formulation is protected by specific patents that extend its market exclusivity beyond the expiration of the original compound patents.
Key patents associated with ABSORICA LD's formulation and its improved pharmacokinetic profile include:
- U.S. Patent No. 8,703,804: This patent, titled "Isotretinoin compositions with improved bioavailability," was granted on April 22, 2014, and has an expected expiration date of April 22, 2031. It covers specific lipid-based formulations of isotretinoin.
- U.S. Patent No. 8,895,088: Granted on November 25, 2014, and expiring on April 22, 2031, this patent is related to the formulation and method of manufacturing the lipid-dispersion isotretinoin.
These formulation patents are critical for maintaining Bausch Health's market exclusivity for ABSORICA LD, preventing generic competitors from marketing an identical formulation until patent expiration. The expiration of U.S. Patent No. 8,703,804 in April 2031 represents a significant future event that will open the market to potential generic competition for the specific ABSORICA LD formulation.
What is the Competitive Landscape for Severe Nodular Acne Treatments?
The market for severe recalcitrant nodular acne treatments is multifaceted, encompassing oral retinoids, antibiotics, hormonal therapies, and topical agents. ABSORICA LD competes within this complex ecosystem.
Direct Competition (Oral Isotretinoin):
- Generic Isotretinoin: The most significant competitive threat to ABSORICA LD's pricing power comes from generic versions of isotretinoin. While these generics do not replicate the specific Lipid Dispersion formulation, they offer the same active ingredient at a substantially lower cost. Market penetration of generics is a primary driver of price erosion for branded drugs.
- Other Branded Isotretinoin Formulations: While ABSORICA LD's LD formulation aims for improved outcomes, other branded isotretinoin products may exist, though less prominent.
Indirect Competition (Alternative Therapies):
- Oral Antibiotics: Doxycycline and minocycline are commonly prescribed for inflammatory acne, though their use in severe nodular acne is typically limited due to concerns about antibiotic resistance and efficacy compared to isotretinoin.
- Hormonal Therapies: For female patients, oral contraceptives and spironolactone can be effective in managing acne by targeting hormonal imbalances.
- Topical Treatments: While not primary treatments for severe nodular acne, topical retinoids (e.g., tretinoin, adapalene), benzoyl peroxide, and topical antibiotics can be used as adjunctive therapies or for milder forms of acne.
- Newer Agents: Emerging treatments, including biologics and other novel oral agents, may eventually enter the severe acne space, although they are not currently considered direct competitors to isotretinoin for the core indication of severe recalcitrant nodular acne.
The competitive advantage of ABSORICA LD lies in its proprietary formulation, which Bausch Health markets as providing improved tolerability and bioavailability compared to standard generic isotretinoin. This differentiation is key to justifying its premium pricing.
What is ABSORICA LD's Pricing Strategy and Market Penetration?
ABSOLICA LD is positioned as a premium branded product within the isotretinoin market. Its pricing reflects the perceived benefits of its proprietary Lipid Dispersion formulation and the market exclusivity granted by its patents.
Pricing:
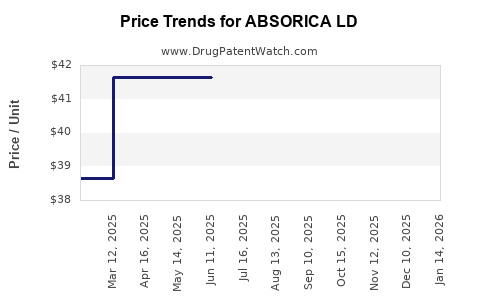
- Brand vs. Generic Pricing: ABSORICA LD commands a significantly higher price point than generic isotretinoin products. The average wholesale price (AWP) for ABSORICA LD is considerably higher, often several hundred dollars per month's supply, depending on dosage. Generic isotretinoin can be obtained for a fraction of this cost, often under $100 per month.
- Manufacturer's Suggested Retail Price (MSRP) and Actual Selling Price: While MSRP provides a benchmark, actual selling prices are subject to negotiations with pharmacy benefit managers (PBMs), insurance providers, and discounts offered through patient assistance programs.
- Patient Assistance Programs: Bausch Health offers patient assistance programs for ABSORICA LD, which can reduce out-of-pocket costs for eligible patients. These programs are standard practice for branded specialty medications to improve access and adherence, but they also impact net revenue.
Market Penetration:
- Target Patient Population: ABSORICA LD targets patients with severe recalcitrant nodular acne who have not responded to other treatments. This is a well-defined but niche patient population.
- Prescription Volume: While precise real-time prescription data is proprietary, market analysis indicates that ABSORICA LD has achieved substantial market share within the branded isotretinoin segment. However, its overall volume is constrained by the availability of lower-cost generic alternatives.
- Factors Influencing Penetration:
- Physician Prescribing Habits: Dermatologists familiar with and confident in the efficacy and tolerability of ABSORICA LD are key drivers of its adoption.
- Insurance Coverage and Formulary Placement: Favorable formulary placement with co-pays that do not create undue barriers for patients is critical. High co-pays or prior authorization requirements can limit market penetration.
- Patient Adherence and Tolerability: The marketing claims of improved tolerability are designed to enhance adherence, thereby supporting sustained use and market penetration.
The pricing strategy is to capture value from the improved formulation, while market penetration is driven by clinical perception, physician adoption, and payer landscape. The gap between ABSORICA LD's price and generic isotretinoin's price is a constant challenge for market share expansion beyond its current patient base.
What is ABSORICA LD's Sales Performance and Revenue Generation?
ABSOLICA LD is a significant contributor to Bausch Health's Pharmaceuticals segment. Analyzing its sales performance provides insights into its market acceptance and financial impact.
Historical Sales Trends:
Publicly available financial reports from Bausch Health Companies Inc. provide revenue figures for their dermatology products. While specific revenue for ABSORICA LD is often aggregated with other dermatology brands, segment performance indicates its importance.
- 2022 Revenue: Bausch Health reported total net sales of $8.1 billion in 2022. The Dermatology segment, which includes ABSORICA LD, is a key revenue driver. Specific figures for ABSORICA LD are not always disaggregated in quarterly or annual reports, but indications from segment performance are positive.
- 2023 Projections and Performance: In 2023, Bausch Health's dermatology business continued to be a core area of focus. The company has highlighted the strength of its prescription dermatology portfolio. For example, in their Q3 2023 earnings, Bausch Health reported net sales of $2.11 billion, with the prescription business contributing significantly. The company noted that growth in its dermatology segment was driven by its key products, which would include ABSORICA LD.
Factors Influencing Sales:
- Market Share in Indication: ABSORICA LD holds a notable share within the branded isotretinoin market. Its sales performance is directly linked to its ability to differentiate from generics and secure prescriber confidence.
- Competition from Generics: The availability of low-cost generic isotretinoin limits the total addressable market for branded products and exerts downward pressure on overall pricing.
- Formulation Benefits: Bausch Health emphasizes the Lipid Dispersion formulation as leading to better patient outcomes and tolerability. Positive clinical experience and physician advocacy are critical sales drivers.
- Marketing and Promotion: Investment in marketing and sales force detailing to dermatologists is essential for maintaining and growing ABSORICA LD's sales.
- Regulatory Environment and Risk Management: As with all isotretinoin products, ABSORICA LD is subject to strict regulatory oversight, including risk management programs (e.g., iPLEDGE in the U.S. historically, though iPLEDGE was discontinued and replaced by REMS programs for isotretinoin). Compliance and effective risk management are crucial for continued market access and sales.
Revenue Projections:
Forecasting ABSORICA LD's future revenue involves considering the remaining patent exclusivity, potential for generic entry upon patent expiration, and the competitive landscape.
- Pre-Patent Expiration (Until April 2031): During this period, ABSORICA LD is expected to maintain its premium pricing and capture a significant portion of the branded isotretinoin market. Sales are projected to remain robust, with modest growth or stability, assuming no major competitive disruptions or shifts in treatment paradigms.
- Post-Patent Expiration (After April 2031): Upon the expiration of the key formulation patents (U.S. Patent No. 8,703,804 in April 2031), the market is expected to open to generic versions of the Lipid Dispersion formulation. This will likely lead to a significant decline in ABSORICA LD's revenue, similar to the trajectory of other branded drugs facing generic competition. The extent of the decline will depend on how quickly generic manufacturers can launch and how aggressively they price their products.
The sales trajectory of ABSORICA LD is characterized by strong performance during its patent-protected period, with a significant anticipated shift upon patent expiration.
What are the Financial Implications and Future Outlook for ABSORICA LD?
The financial trajectory of ABSORICA LD is primarily dictated by its patent exclusivity, market competition, and pricing power. The drug represents a valuable asset for Bausch Health, particularly during its period of patent protection.
Current Financial Contribution:
- Revenue Driver: ABSORICA LD is a key revenue generator within Bausch Health's dermatology division. Its ability to command a premium price over generic isotretinoin contributes meaningfully to the company's profitability.
- Profitability: The higher gross margins typically associated with branded pharmaceuticals, especially those with proprietary formulations, mean that ABSORICA LD likely has strong profitability per unit sold compared to generic alternatives.
Future Outlook and Financial Projections:
The future financial outlook for ABSORICA LD is bifurcated based on its patent life.
1. Pre-Patent Expiration (Current to April 2031):
- Stable to Modest Growth: Expect continued strong sales performance. The market for severe acne is established, and ABSORICA LD's differentiated formulation provides a competitive advantage against generics.
- Continued Premium Pricing: Bausch Health will leverage its remaining patent protection to maintain pricing power.
- Investment in Marketing: Ongoing investment in marketing and sales force support will be crucial to defend its market share and maximize revenue before patent expiration.
- Potential for Slight Erosion: While patents are in place, gradual market share erosion to generics can occur due to price sensitivity among certain patient segments or payer pressures, though this is mitigated by formulation patents.
2. Post-Patent Expiration (April 2031 onwards):
- Significant Revenue Decline: Upon the expiration of U.S. Patent No. 8,703,804 in April 2031, generic competition for the Lipid Dispersion formulation will emerge. This will lead to a sharp and substantial decrease in ABSORICA LD's sales and profitability, a phenomenon known as "patent cliff."
- Price Compression: Generic entry will force a drastic reduction in the price of the formulation.
- Loss of Market Share: ABSORICA LD will face intense competition from multiple generic manufacturers, leading to a significant loss of market share.
- Bausch Health's Strategy: Bausch Health will need to manage this transition by focusing on other growth drivers within its portfolio, potentially exploring lifecycle management strategies (though less common for established generics), or shifting resources. The company's financial planning would account for this anticipated decline.
Key Financial Considerations for Investors and R&D:
- Risk Assessment: The primary financial risk associated with ABSORICA LD is the impending patent expiration. Investors must factor in the revenue decline post-2031.
- Valuation: The current valuation of ABSORICA LD within Bausch Health should reflect its strong performance during its patent-protected life. Future valuation will be significantly impacted by generic entry.
- R&D Investment: Bausch Health's ongoing R&D strategy will be critical. Investment in next-generation acne treatments or formulations that can achieve similar market positioning as ABSORICA LD did would be a logical diversification.
The financial trajectory of ABSORICA LD is characterized by a lucrative but finite period of market exclusivity, followed by a predictable and significant decline due to generic competition.
Key Takeaways
- ABSOLICA LD's market exclusivity is primarily protected by formulation patents, with U.S. Patent No. 8,703,804 expiring in April 2031.
- The drug competes with generic isotretinoin and other acne therapies, differentiating itself through a proprietary Lipid Dispersion formulation claimed to improve bioavailability and tolerability.
- ABSOLICA LD is priced as a premium branded product, significantly higher than generic isotretinoin, with sales performance driven by physician adoption and formulary access.
- The drug is a substantial revenue contributor to Bausch Health's dermatology segment.
- Post-April 2031, ABSORICA LD is projected to experience a significant revenue decline due to the anticipated entry of generic competitors for its specific formulation.
Frequently Asked Questions
-
What is the primary mechanism of action for ABSORICA LD?
ABSOLICA LD contains isotretinoin, a retinoid that works by reducing the size and oil production of sebaceous glands, preventing clogged pores, and decreasing inflammation, thereby treating severe recalcitrant nodular acne.
-
When is the key patent for ABSORICA LD's formulation scheduled to expire?
The key U.S. patent (U.S. Patent No. 8,703,804) protecting ABSORICA LD's Lipid Dispersion formulation is scheduled to expire on April 22, 2031.
-
How does ABSORICA LD differ from generic isotretinoin products?
ABSOLICA LD utilizes a proprietary Lipid Dispersion formulation designed to enhance isotretinoin's bioavailability and potentially improve tolerability compared to standard generic formulations.
-
What is the expected impact on ABSORICA LD's sales following patent expiration?
Upon patent expiration, generic versions of the ABSORICA LD formulation are expected to enter the market, leading to significant price compression and a substantial decline in ABSORICA LD's sales and market share.
-
Are there any significant risk management programs associated with isotretinoin products like ABSORICA LD?
Yes, isotretinoin products are subject to stringent risk management programs due to potential serious side effects, such as birth defects. While the specific iPLEDGE program was discontinued, REMS (Risk Evaluation and Mitigation Strategies) programs remain in place for isotretinoin to ensure safe prescribing and dispensing.
Citations
[1] Bausch Health Companies Inc. (n.d.). Investor Relations. Retrieved from [Company Investor Relations Website - specific report access varies]
[2] Google Patents. (n.d.). U.S. Patent 4,272,422. Retrieved from patents.google.com
[3] Google Patents. (n.d.). U.S. Patent 8,703,804. Retrieved from patents.google.com
[4] Google Patents. (n.d.). U.S. Patent 8,895,088. Retrieved from patents.google.com