Last updated: February 19, 2026
Adapalene, a topical retinoid, demonstrates sustained market presence and a clear financial trajectory driven by its efficacy in treating acne vulgaris. Its established safety profile and over-the-counter (OTC) availability in many regions contribute to consistent prescription and sales volumes.
What is Adapalene's Chemical Classification and Mechanism of Action?
Adapalene is a synthetic naphthoic acid derivative classified as a third-generation topical retinoid. Its primary mechanism of action involves binding to specific retinoic acid receptor (RAR) subtypes, particularly RAR-beta and RAR-gamma, with relative selectivity over RAR-alpha. This selective binding modulates cellular processes critical in acne pathogenesis.
Adapalene normalizes follicular keratinization, preventing the formation of microcomedones, the initial lesions in acne development. It exhibits comedolytic activity, promoting the shedding of dead skin cells within hair follicles, thereby unblocking pores. Additionally, adapalene possesses anti-inflammatory properties, reducing the redness and swelling associated with inflammatory acne lesions such as papules and pustules. Unlike older retinoids, adapalene exhibits lower affinity for cellular retinoic acid binding proteins (CRABPs), which may contribute to its improved tolerability and reduced potential for skin irritation.
Who are the Key Manufacturers and Their Market Share?
The market for adapalene is characterized by a mix of originator products and a significant presence of generic manufacturers. Key players include:
- Galderma: The originator of adapalene, marketed as Differin®. Galderma maintains a strong market position, particularly with its prescription and subsequently OTC formulations.
- Generic Manufacturers: A substantial number of pharmaceutical companies produce generic adapalene products. These include, but are not limited to, manufacturers such as:
- Bausch Health Companies
- Valeant Pharmaceuticals International (now rebranded as Bausch Health)
- Teva Pharmaceutical Industries
- Sandoz (a division of Novartis)
- Sun Pharmaceutical Industries
- Lupin Pharmaceuticals
- Mylan N.V. (now Viatris)
The market share is fragmented due to the widespread availability of generics. Galderma, as the innovator, retains brand loyalty and market share, especially in premium OTC segments and continuing prescription markets. Generic manufacturers compete primarily on price, capturing significant volume. Precise market share percentages are dynamic and vary by region and distribution channel (prescription vs. OTC).
What is the Global Market Size and Projected Growth for Adapalene?
The global market size for adapalene is estimated to be in the range of USD 500 million to USD 700 million annually. This figure encompasses both prescription and over-the-counter (OTC) sales across major pharmaceutical markets.
Projected growth for the adapalene market is moderate, with an expected Compound Annual Growth Rate (CAGR) of 3% to 5% over the next five to seven years. This growth is primarily driven by:
- Increasing Acne Prevalence: Global incidence of acne vulgaris remains high, particularly among adolescents and young adults.
- OTC Accessibility: The shift of adapalene to OTC status in numerous key markets (e.g., United States, Canada, Australia) has broadened consumer access and driven unit sales.
- Combination Therapies: The development and marketing of adapalene in combination with other active ingredients (e.g., benzoyl peroxide) offer enhanced efficacy and expand treatment options.
- Emerging Markets: Growing healthcare infrastructure and increasing awareness of dermatological conditions in emerging economies are expected to contribute to market expansion.
However, growth is tempered by:
- Competition: The availability of a wide array of generic products and alternative acne treatments limits significant price appreciation.
- Evolving Treatment Paradigms: Newer therapies, including oral medications and advanced dermatological procedures, may divert some patient populations.
What are the Key Regulatory Approvals and Their Timeline?
The regulatory journey of adapalene has been marked by its progression from prescription-only to over-the-counter (OTC) status in several major markets.
- Initial Prescription Approval (U.S. FDA): Adapalene was first approved by the U.S. Food and Drug Administration (FDA) in 1996 as a prescription topical treatment for acne vulgaris, marketed as Differin® gel by Galderma Laboratories.
- European Union Approval: Adapalene received marketing authorization in the EU subsequent to its U.S. approval, also initially as a prescription product.
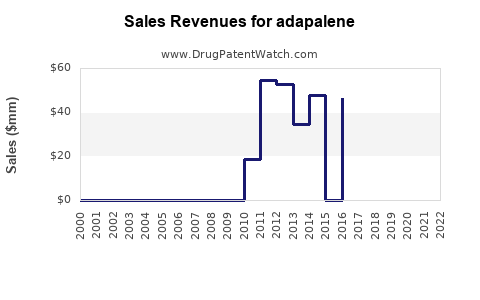
- OTC Reclassification (U.S. FDA): A significant milestone was the FDA's approval of Differin® Gel 0.1% for OTC use in July 2016. This decision was based on a thorough review of safety and efficacy data, supporting its suitability for self-treatment of mild to moderate acne.
- OTC Availability in Other Regions: Following the U.S. approval, other regulatory bodies have also permitted OTC access to adapalene, with timelines varying by country and region. This process typically involves submitting data demonstrating safety for unsupervised use. For instance, Health Canada and Australia’s Therapeutic Goods Administration (TGA) have also facilitated OTC access.
- European Union OTC Considerations: While adapalene is available in various formulations and concentrations across EU member states, its full OTC reclassification at a unified level mirrors the U.S. timeline and regulatory pathway, with progress being made through national marketing authorizations.
What are the Pricing Strategies and Revenue Streams?
Adapalene exhibits diverse pricing strategies influenced by its regulatory status, brand versus generic competition, and formulation.
- Prescription Formulations:
- Innovator Brand (Differin®): Commands a premium price due to brand recognition, established clinical trust, and marketing investment. Pricing is typically higher than generics.
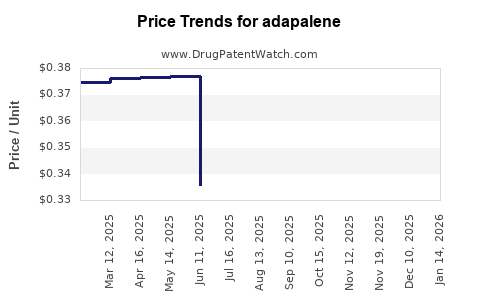
- Generic Prescription: Priced significantly lower than the innovator brand, driven by competition among multiple generic manufacturers. Reimbursement policies by private insurers and national health systems heavily influence these prices.
- Over-the-Counter (OTC) Formulations:
- Brand OTC (Differin® Gel 0.1%): Positioned as a premium OTC product, priced above generic OTC equivalents but below prescription-only formulations of the innovator brand. This tier targets consumers seeking a trusted, clinically proven brand for self-care.
- Generic OTC: The most price-competitive segment. Manufacturers focus on volume sales, offering adapalene at accessible price points to a broad consumer base.
- Formulation Variations: Different concentrations (e.g., 0.1%, 0.3%) and vehicle types (gel, cream, lotion) can influence pricing. Higher concentrations or specialized formulations may command slightly higher prices.
- Combination Products: Adapalene formulated with other active ingredients (e.g., benzoyl peroxide) are typically priced higher than monotherapy products, reflecting the combined therapeutic benefit and added formulation complexity.
- Revenue Streams:
- Direct Sales to Pharmacies and Retailers: Manufacturers sell products in bulk to distributors, pharmacies, and major retail chains.
- Wholesale Distribution: A significant portion of revenue is generated through sales to wholesale drug distributors who then supply pharmacies.
- Online Pharmacies and E-commerce: Increasing sales through direct-to-consumer online platforms and major e-commerce retailers.
- Licensing and Partnerships: In some instances, originator companies may license their technology or formulations to other entities for regional manufacturing or distribution, generating royalty revenue.
What are the Key Patent Expirations and Their Impact?
The patent landscape for adapalene has largely expired, significantly impacting market dynamics.
- Composition of Matter Patents: The original patents covering the chemical structure of adapalene have long since expired. The primary composition of matter patent for adapalene expired in 2009.
- Formulation and Method of Use Patents: While composition of matter patents have expired, secondary patents related to specific formulations (e.g., novel delivery systems, combinations) or methods of use may have existed for a period after the primary patent. However, these have also largely expired or are nearing expiration.
- Impact of Patent Expirations:
- Generic Entry and Price Erosion: The expiration of key patents opened the door for widespread generic competition. This has led to substantial price erosion for both prescription and OTC adapalene products.
- Increased Market Volume: Lower prices driven by generic competition have made adapalene more accessible to a larger patient population, potentially increasing overall unit sales volume.
- Shift in Manufacturer Strategy: Companies that previously held strong positions based on patent exclusivity now focus on brand building, marketing, combination products, and cost-efficient manufacturing to maintain market share.
- Innovation Focus on New Formulations or Combinations: With the core compound off-patent, innovation efforts from originators and research institutions tend to focus on novel delivery systems for improved tolerability, efficacy, or unique combination therapies that may be protectable by new intellectual property.
What are the Competitive Landscape and Alternatives?
The acne treatment market is highly competitive, with adapalene facing competition from both topical and systemic agents, as well as other therapeutic modalities.
Direct Topical Retinoid Competitors:
- Tretinoin: The first-generation topical retinoid. Available by prescription, it has a long history of use but can be associated with higher irritation potential.
- Tazarotene: A third-generation topical retinoid, more potent than adapalene but often with a less favorable tolerability profile. It is available by prescription.
Other Topical Acne Treatments:
- Benzoyl Peroxide: A widely available OTC and prescription antibacterial agent. Often used in combination with adapalene.
- Topical Antibiotics: (e.g., clindamycin, erythromycin) Primarily target Cutibacterium acnes bacteria and reduce inflammation. Often used in combination with other agents to prevent antibiotic resistance.
- Azelaic Acid: Has antibacterial, anti-inflammatory, and mild comedolytic effects. Available OTC and by prescription.
- Salicylic Acid: A beta-hydroxy acid (BHA) with comedolytic and mild anti-inflammatory properties. Widely available OTC.
- Topical Dapsone: An anti-inflammatory and antibacterial agent, often prescribed for inflammatory acne.
Systemic Acne Treatments:
- Oral Antibiotics: (e.g., doxycycline, minocycline) Used for moderate to severe inflammatory acne.
- Isotretinoin: A highly effective oral retinoid for severe, recalcitrant acne. Its use is associated with significant side effects and requires strict monitoring.
- Hormonal Therapies: (e.g., oral contraceptives, spironolactone) Can be effective for hormonal acne in women.
Other Modalities:
- Chemical Peels: Professional treatments that exfoliate the skin.
- Light and Laser Therapies: Used for reducing bacteria and inflammation.
Adapalene's competitive advantage lies in its balanced profile of efficacy, tolerability, and accessibility (especially OTC). While other retinoids may offer higher potency, adapalene's lower irritation potential makes it a preferred first-line option for many patients and dermatologists, particularly for long-term maintenance therapy. Its availability as an OTC product further strengthens its market position against prescription-only alternatives for mild to moderate acne.
What are the Key Growth Drivers and Restraints?
Growth Drivers:
- Rising Acne Prevalence Globally: The consistent and widespread occurrence of acne vulgaris across all age groups, particularly among adolescents and young adults, provides a large and sustained patient base.
- Increased OTC Availability and Accessibility: The reclassification of adapalene to OTC status in major markets has significantly broadened its consumer reach, driving self-treatment and unit sales volume. Consumers can now purchase effective acne treatment without a prescription.
- Combination Product Development: The strategic formulation of adapalene with other active ingredients, such as benzoyl peroxide, offers enhanced efficacy and multi-target treatment, appealing to patients with more complex acne conditions. These combination products often carry premium pricing.
- Growing Awareness of Dermatological Health: Increasing consumer awareness regarding skin health, the impact of acne on quality of life, and the availability of effective treatments fuels demand for products like adapalene.
- Expansion in Emerging Markets: As healthcare infrastructure improves and disposable incomes rise in emerging economies, access to and demand for dermatological treatments, including adapalene, is expected to increase.
- Favorable Tolerability Profile: Compared to older retinoids, adapalene generally exhibits better tolerability, leading to higher patient adherence to treatment regimens.
Restraints:
- Intense Generic Competition and Price Erosion: The widespread availability of low-cost generic adapalene products has significantly reduced pricing power for both branded and generic manufacturers, limiting revenue growth from price increases.
- Emergence of Novel Acne Therapies: Ongoing research and development in dermatology may introduce new therapeutic agents or technologies that could offer superior efficacy or novel mechanisms of action, potentially shifting treatment paradigms away from established options.
- Regulatory Hurdles for New Indications or Formulations: While adapalene itself is well-established, obtaining approval for new indications or significantly novel formulations can be a lengthy and costly process.
- Patient Adherence Challenges: Despite improved tolerability, some patients may still experience side effects (e.g., dryness, redness, peeling) that can impact adherence, especially with long-term use.
- Potential for Skin Irritation: While generally well-tolerated, adapalene can still cause irritation in a subset of users, requiring careful usage and potentially limiting its appeal for individuals with highly sensitive skin.
What is the Future Outlook and Potential Market Disruptions?
The future outlook for adapalene remains stable, characterized by steady demand and moderate growth. The market is unlikely to experience dramatic disruption in the short to medium term, but several factors could influence its trajectory.
- Continued Dominance in OTC Segment: Adapalene's established position as a leading OTC topical retinoid will likely continue. Its efficacy and safety profile for mild to moderate acne are well-documented and trusted by consumers.
- Growth in Combination Formulations: The trend towards combination therapies will likely persist, with adapalene being a key component. Innovations in fixed-dose combinations offering enhanced convenience and efficacy will drive sales.
- Geographic Expansion: Further expansion of OTC access in remaining regulated markets could unlock new growth opportunities and increase global penetration.
- Focus on Patient Support and Education: Manufacturers will likely invest in patient education programs to improve adherence and manage potential side effects, maximizing treatment outcomes and brand loyalty.
Potential Market Disruptions:
- Advanced Delivery Systems: Development of novel drug delivery systems (e.g., microencapsulation, sustained-release formulations) that further enhance adapalene's tolerability and efficacy could create a competitive advantage for innovator products or licensed generics.
- Biologic Treatments for Acne: While currently focused on severe or inflammatory conditions, the advancement of biologic therapies in dermatology could eventually extend to severe forms of acne, potentially impacting the market share of oral and topical retinoids for these specific patient groups.
- AI-Driven Dermatology Solutions: The integration of artificial intelligence in diagnosis and treatment recommendations could influence how acne therapies are prescribed and accessed, though it is unlikely to directly disrupt the fundamental efficacy of chemical compounds like adapalene.
- Direct-to-Consumer (DTC) Model Evolution: Changes in DTC healthcare models, including telehealth and direct prescription services, could alter distribution channels for both prescription and OTC adapalene, potentially increasing competition and impacting traditional pharmacy sales.
Key Takeaways
Adapalene maintains a strong and stable market position, driven by its efficacy, favorable tolerability, and increasing accessibility, particularly its OTC availability. The market is characterized by mature patent protection leading to robust generic competition and price erosion. Future growth is expected to be moderate, sustained by the high prevalence of acne and the development of combination therapies. Key competitors include other topical retinoids, benzoyl peroxide, and antibiotics. Potential disruptions are likely to arise from advancements in drug delivery systems and evolving healthcare access models rather than a complete replacement of the drug class.
FAQs
-
Is adapalene still considered a first-line treatment for acne?
Yes, adapalene remains a first-line topical treatment for mild to moderate acne vulgaris, especially for comedonal and inflammatory lesions, due to its efficacy and favorable tolerability profile.
-
What is the primary difference between prescription and OTC adapalene?
The primary difference is regulatory approval status. OTC adapalene is typically a lower concentration (e.g., 0.1% gel) deemed safe and effective for self-treatment, while higher concentrations or specific formulations may remain prescription-only.
-
Can adapalene be used with other acne treatments?
Adapalene is frequently used in combination with other acne treatments, such as benzoyl peroxide, topical antibiotics, or azelaic acid, to enhance efficacy. However, it is crucial to consult a healthcare professional to avoid potential irritation from combining multiple active ingredients.
-
What is the typical duration of treatment for adapalene to show results?
Visible improvement with adapalene typically occurs after 4 to 8 weeks of consistent use. Complete clearance or significant improvement may take up to 12 weeks or longer.
-
Are there any long-term safety concerns associated with the chronic use of adapalene?
Adapalene has a well-established safety profile for chronic use in acne management. Common side effects are typically localized skin reactions like dryness, redness, and peeling, which often decrease with continued use or dose adjustment. Systemic absorption is minimal, limiting the risk of systemic side effects.
Citations
[1] Food and Drug Administration. (2016, July 28). FDA approves Differin Gel 0.1% as first over-the-counter topical retinoid acne product. U.S. Food & Drug Administration. https://www.fda.gov/drugs/postmarket-drug-safety-information-for-patients-and-providers/differin-gel-01-first-over-counter-topical-retinoid-acne-product
[2] Galderma Laboratories L.P. (n.d.). Differin Gel. Retrieved from manufacturer's product information.
[3] National Center for Biotechnology Information. (n.d.). Adapalene. PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/Adapalene
[4] Search for relevant market research reports on topical acne treatments and retinoids. (Specific reports vary and are proprietary).
[5] Pharmaceutical patent databases (e.g., USPTO, Espacenet) for patent expiration timelines. (Specific patent numbers and expiry dates vary).