Abiraterone acetate - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for abiraterone acetate and what is the scope of patent protection?
Abiraterone acetate
is the generic ingredient in four branded drugs marketed by Amneal Pharms, Apotex, Dr Reddys, Florida, Glenmark Pharms, Hikma, MSN, Mylan, Novugen, Qilu, Rising, Teva Pharms Usa, Wockhardt Bio Ag, Sun Pharm, and Janssen Biotech, and is included in eighteen NDAs. There are ten patents protecting this compound and three Paragraph IV challenges. Additional information is available in the individual branded drug profile pages.Abiraterone acetate has fifty-eight patent family members in twenty-six countries.
There are twenty-two drug master file entries for abiraterone acetate. Twenty-seven suppliers are listed for this compound.
Summary for abiraterone acetate
| International Patents: | 58 |
| US Patents: | 10 |
| Tradenames: | 4 |
| Applicants: | 15 |
| NDAs: | 18 |
| Drug Master File Entries: | 22 |
| Finished Product Suppliers / Packagers: | 27 |
| Raw Ingredient (Bulk) Api Vendors: | 86 |
| Clinical Trials: | 254 |
| Patent Applications: | 3,620 |
| Formulation / Manufacturing: | see details |
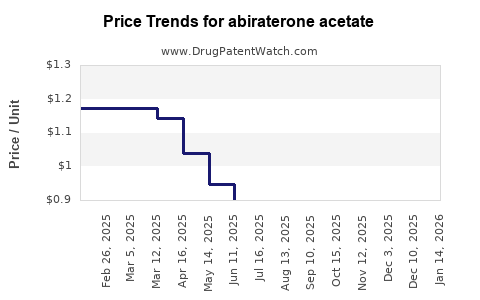
| Drug Prices: | Drug price trends for abiraterone acetate |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for abiraterone acetate |
| What excipients (inactive ingredients) are in abiraterone acetate? | abiraterone acetate excipients list |
| DailyMed Link: | abiraterone acetate at DailyMed |
Recent Clinical Trials for abiraterone acetate
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Changhai Hospital | N/A |
| Merck Sharp & Dohme LLC | Phase 3 |
| Orion Corporation, Orion Pharma | Phase 3 |
Pharmacology for abiraterone acetate
| Drug Class | Cytochrome P450 17A1 Inhibitor |
| Mechanism of Action | Cytochrome P450 17A1 Inhibitors Cytochrome P450 2C8 Inhibitors Cytochrome P450 2D6 Inhibitors |
Medical Subject Heading (MeSH) Categories for abiraterone acetate
Anatomical Therapeutic Chemical (ATC) Classes for abiraterone acetate
Paragraph IV (Patent) Challenges for ABIRATERONE ACETATE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| YONSA | Tablets | abiraterone acetate | 125 mg | 210308 | 1 | 2018-07-23 |
| ZYTIGA | Tablets | abiraterone acetate | 500 mg | 202379 | 1 | 2017-08-23 |
| ZYTIGA | Tablets | abiraterone acetate | 250 mg | 202379 | 13 | 2015-04-28 |
US Patents and Regulatory Information for abiraterone acetate
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Msn | ABIRATERONE ACETATE | abiraterone acetate | TABLET;ORAL | 210686-001 | Jul 10, 2019 | AB | RX | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Msn | ABIRATERONE ACETATE | abiraterone acetate | TABLET;ORAL | 210686-002 | Apr 24, 2024 | AB | RX | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Janssen Biotech | AKEEGA | abiraterone acetate; niraparib tosylate | TABLET;ORAL | 216793-001 | Aug 11, 2023 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Qilu | ABIRATERONE ACETATE | abiraterone acetate | TABLET;ORAL | 212462-002 | Jun 25, 2021 | AB | RX | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Sun Pharm | YONSA | abiraterone acetate | TABLET;ORAL | 210308-001 | May 22, 2018 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Sun Pharm | YONSA | abiraterone acetate | TABLET;ORAL | 210308-001 | May 22, 2018 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Janssen Biotech | AKEEGA | abiraterone acetate; niraparib tosylate | TABLET;ORAL | 216793-002 | Aug 11, 2023 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for abiraterone acetate
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Janssen Biotech | ZYTIGA | abiraterone acetate | TABLET;ORAL | 202379-001 | Apr 28, 2011 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for abiraterone acetate
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Mylan Ireland Limited | Abiraterone Mylan | abiraterone acetate | EMEA/H/C/005368 Abiraterone Mylan is indicated with prednisone or prednisolone for:the treatment of newly diagnosed high risk metastatic hormone sensitive prostate cancer (mHSPC) in adult men in combination with androgen deprivation therapy (ADT).the treatment of metastatic castration resistant prostate cancer (mCRPC) in adult men who are asymptomatic or mildly symptomatic after failure of androgen deprivation therapy in whom chemotherapy is not yet clinically indicated.the treatment of mCRPC in adult men whose disease has progressed on or after a docetaxel based chemotherapy regimen. |
Authorised | yes | no | no | 2021-08-20 | |
| Krka, d.d., Novo mesto | Abiraterone Krka | abiraterone acetate | EMEA/H/C/005649 Abiraterone Krka is indicated with prednisone or prednisolone for:the treatment of newly diagnosed high risk metastatic hormone sensitive prostate cancer (mHSPC) in adult men in combination with androgen deprivation therapy (ADT) (see section 5.1)the treatment of metastatic castration resistant prostate cancer (mCRPC) in adult men who are asymptomatic or mildly symptomatic after failure of androgen deprivation therapy in whom chemotherapy is not yet clinically indicated (see section 5.1)the treatment of mCRPC in adult men whose disease has progressed on or after a docetaxel-based chemotherapy regimen. |
Authorised | yes | no | no | 2021-06-24 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for abiraterone acetate
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Singapore | 11201701139Y | ABIRATERONE ACETATE FORMULATION AND METHODS OF USE | ⤷ Sign Up |
| Brazil | 112017003219 | formulação de acetato de abiraterona e métodos de uso | ⤷ Sign Up |
| Philippines | 12017500239 | ABIRATERONE ACETATE FORMULATION AND METHODS OF USE | ⤷ Sign Up |
| Canada | 2907415 | FORMULATION D'ACETATE D'ABIRATERONE (ABIRATERONE ACETATE FORMULATION) | ⤷ Sign Up |
| Japan | 2017528457 | 酢酸アビラテロン製剤及び使用方法 | ⤷ Sign Up |
| China | 106687112 | 醋酸阿比特龙制剂和使用方法 (Abiraterone acetate formulation and methods of use) | ⤷ Sign Up |
| South Korea | 102491439 | ⤷ Sign Up | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for abiraterone acetate
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0633893 | SPC/GB11/063 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: ABIRATERONE AND ACID ADDITION SALTS AND 3-ESTERS THEREOF ESPECIALLY ABIRATERONE ACETATE; REGISTERED: UK EU/1/11/714/001 20110905 |
| 0633893 | 2012/003 | Ireland | ⤷ Sign Up | PRODUCT NAME: ZYTIGA (ABIRATERONE) "ABIRATERONE AND ACID ADDITION SALTS AND 3-ESTERS THEREOF, ESPECIALLY ABIRATERONE ACETATE"; REGISTRATION NO/DATE: EU/1/11/714/001 20110905 |
| 0633893 | 11C0055 | France | ⤷ Sign Up | PRODUCT NAME: ABIRATERONE, SES SELS D'ADDITION D'ACIDE ET 3-ESTERS, EN PARTICULIER ACETATE D'ABIRATERONE; REGISTRATION NO/DATE: EU/1/11/714/001 20110905 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.