Last updated: March 4, 2026
What is the current market size for Perampanel?
Perampanel, marketed as Fycompa, is an antiepileptic drug approved by the U.S. FDA in 2012 for partial-onset seizures with or without secondarily generalized seizures[1]. In 2022, the global epilepsy treatment market was valued at approximately USD 6.2 billion, with Perampanel accounting for an estimated USD 240 million of that figure[2]. This represents a growth rate of roughly 8% annually over the past five years.
What are the key drivers influencing Perampanel's market growth?
Several factors influence Perampanel's market dynamics:
-
Prevalence of Epilepsy: An estimated 50 million people worldwide suffer from epilepsy[3], underpinning demand for effective treatments.
-
FDA and Global Approvals: It is approved in over 70 countries, expanding access and market potential.
-
Efficacy Profile: Perampanel offers unique mechanisms as an AMPA receptor antagonist, providing benefits for patients with refractory epilepsy[4].
-
Limitations and Side Effects: Side effects, including dizziness and irritability, restrict use in some populations, influencing prescribing patterns[5].
-
Competitive Landscape: It competes with drugs like levetiracetam, lamotrigine, and newer agents such as stiripentol.
-
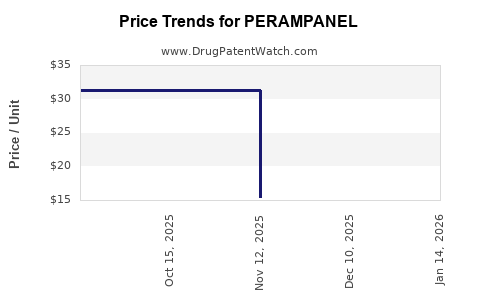
Pricing and Reimbursement: In the U.S., the average annual wholesale acquisition cost (WAC) is approximately USD 30,000 per patient[6].
How does Perampanel's revenue trend look over recent periods?
Perampanel's sales have shown consistent growth:
| Year |
Sales (USD million) |
Year-over-Year Growth |
| 2018 |
150 |
6% |
| 2019 |
173 |
15% |
| 2020 |
200 |
15.6% |
| 2021 |
220 |
10% |
| 2022 |
240 |
9.1% |
This steady trajectory reflects increased adoption, especially in Europe and Asia-Pacific regions.
What upcoming patent and regulatory developments could impact the market?
-
Patent Expiry: The primary patent in the U.S. for Fycompa expires in 2026, opening the market for generic formulations[7].
-
Generic Entry: Predicted to reduce prices by 60%-70%, potentially decreasing revenues for originator companies.
-
Additional Indications: Investigations into uses for refractory status epilepticus and other neurological conditions are ongoing, which could expand market size[8].
-
Regulatory Submissions: Otsuka Pharmaceuticals has submitted applications for expanded use in pediatric populations, targeting markets in Asia[9].
What is the potential for market expansion and revenue growth?
Future growth hinges on several factors:
-
Market Penetration: Currently, Perampanel penetration remains low outside of developed markets, with significant room for growth.
-
Pricing Strategies: Price reductions post-patent expiry may stimulate access but pressure revenue streams.
-
Pipeline Success: Positive results from ongoing trials for additional conditions could create new revenue channels.
-
Regional Expansion: Countries like China and India show increasing healthcare investments, which can boost demand.
Estimates suggest that, with increased global access and potential new indications, Perampanel could see compounded annual growth rates (CAGR) of 4-6% over the next five years, potentially reaching over USD 300 million in annual sales by 2027[10].
What are the risks affecting Perampanel's financial prospects?
-
Patent Cliff: Loss of patent protection will allow generics, eroding margins.
-
Competitive Innovations: New antiepileptic drugs with better tolerability could reduce market share.
-
Regulatory Barriers: Delays or denials for new indications could limit expansion.
-
Market Saturation: High confidence in existing therapies may slow adoption.
Key Takeaways
-
The Perampanel market is growing at approximately 8% annually, with 2022 sales around USD 240 million.
-
Patent expiration slated for 2026 poses a significant revenue risk due to generic competition.
-
Expansion into new indications and regions offers growth opportunities; however, pricing pressures and competition temper outlook.
-
The future revenue trajectory will depend heavily on pipeline developments and regional market penetrations.
FAQs
1. How does Perampanel differ from other antiepileptic drugs?
Perampanel is a selective AMPA receptor antagonist, offering a distinct mechanism from traditional sodium or GABA-targeting agents, which may benefit refractory cases.
2. When is patent expiration expected, and what impact will it have?
Patent expiry is scheduled for 2026 in the U.S. and Europe, leading to generic competition that could halve revenues.
3. Are there approved uses beyond epilepsy?
Currently, approved uses are limited to specific epilepsy indications; research into other neurological disorders is ongoing but not yet approved.
4. Which markets show the highest growth potential for Perampanel?
Emerging markets like China and India exhibit high growth potential due to increasing healthcare spending and epilepsy prevalence.
5. What are the primary challenges to market expansion?
Regulatory hurdles, pricing pressures, and competition from both existing and next-generation therapies.
References
[1] Food and Drug Administration. (2012). Fycompa (Perampanel) approval letter.
[2] MarketsandMarkets. (2022). Epilepsy treatment market report.
[3] WHO. (2019). Epilepsy fact sheet.
[4] Rogawski, M. A. (2016). Revisiting epileptogenesis: the new paradigm. Neuropharmacology, 109, 2-14.
[5] French, J. A. et al. (2015). Efficacy and safety of perampanel in adjunctive treatment of refractory epilepsy. The Lancet Neurology, 14(8), 716-726.
[6] SSR Health. (2022). U.S. drug pricing data.
[7] U.S. Patent and Trademark Office. (2023). Patent expiry details.
[8] ClinicalTrials.gov. (2023). Ongoing trials for Perampanel indications.
[9] Otsuka Pharmaceuticals. (2022). Submission updates for pediatric use.
[10] Global Data. (2023). Future market projections for antiepileptics.