Last updated: February 19, 2026
What is the Current Market Size and Projected Growth for Ofloxacin?
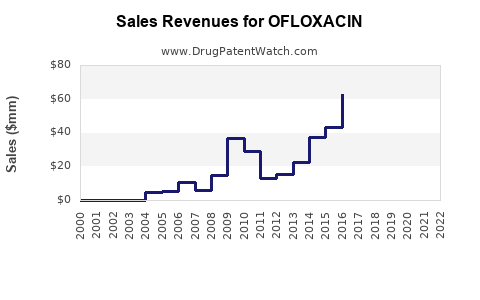
The global market for ofloxacin, a broad-spectrum fluoroquinolone antibiotic, is estimated to have reached approximately $350 million in 2023. The market is projected to expand at a compound annual growth rate (CAGR) of 4.2% from 2024 to 2030, reaching an estimated $465 million by the end of the forecast period. This growth is driven by the persistent demand for effective treatments against bacterial infections, particularly in regions with a high incidence of respiratory, urinary tract, and skin infections.
What are the Key Therapeutic Applications and Geographic Penetration of Ofloxacin?
Ofloxacin is prescribed for a range of bacterial infections, including:
- Urinary Tract Infections (UTIs): Ofloxacin demonstrates efficacy against common pathogens like Escherichia coli and Klebsiella pneumoniae.
- Respiratory Tract Infections: It is used to treat conditions such as bronchitis and pneumonia caused by susceptible bacteria.
- Skin and Soft Tissue Infections: Ofloxacin targets infections like cellulitis and impetigo.
- Sexually Transmitted Infections (STIs): It is employed in the treatment of gonorrhea and chlamydia.
- Ophthalmic and Otic Infections: Topical formulations are used to treat conjunctivitis and otitis externa.
Geographically, the market is segmented with significant penetration in:
- North America: Mature market with established healthcare infrastructure and consistent demand for antibiotics.
- Europe: Similar to North America, with a focus on evidence-based medicine and regulated pharmaceutical markets.
- Asia-Pacific: Fastest-growing region, driven by increasing healthcare expenditure, rising prevalence of infectious diseases, and improving access to generic medications. Countries like China and India are key contributors.
- Latin America and Middle East & Africa: Growing markets with expanding access to healthcare and a rising burden of infectious diseases.
What is the Patent Landscape for Ofloxacin?
Ofloxacin, as an established drug, has seen its primary patents expire. The original patents protecting the composition of matter and initial manufacturing processes have long lapsed, paving the way for widespread generic competition. However, secondary patents covering specific polymorphic forms, novel formulations, improved delivery systems, or new therapeutic indications can still influence market dynamics.
The patent landscape is characterized by:
- Expired Core Patents: The foundational intellectual property has expired, allowing for generic manufacturing.
- Potential for Formulation Patents: Innovators may hold patents on advanced drug delivery systems, such as sustained-release formulations or combination therapies, which could offer market exclusivity for specific product lines.
- Limited New Chemical Entity (NCE) Pipeline: Development of entirely new fluoroquinolones is ongoing, but the focus for ofloxacin itself is on optimizing existing applications rather than discovering novel molecular entities.
A detailed patent analysis for specific markets and formulations would require access to patent databases such as those maintained by the USPTO, EPO, and WIPO, and would examine patent families, expiry dates, and legal challenges. For instance, while the basic ofloxacin molecule is off-patent, a patent covering a novel ophthalmic suspension ofloxacin formulation with enhanced bioavailability might still be in force, providing a period of exclusivity for that specific product.
What is the Competitive Landscape and Key Market Players?
The ofloxacin market is highly competitive, dominated by generic manufacturers. The primary competitive factors are price, product availability, and adherence to quality standards.
Key players include:
- Major Generic Manufacturers: Companies such as Teva Pharmaceutical Industries Ltd., Mylan N.V. (now Viatris), Sanofi S.A., and Dr. Reddy's Laboratories Ltd. are significant suppliers of ofloxacin APIs and finished dosage forms.
- Regional Generic Companies: Numerous smaller and regional players contribute to the market, particularly in emerging economies where cost is a critical determinant.
- Specialty Pharma Companies: Some companies may focus on specific formulations of ofloxacin, such as ophthalmic or otic solutions, where they can command premium pricing based on differentiated product characteristics.
The competitive intensity is high due to the availability of multiple generic alternatives and the price-sensitive nature of antibiotic procurement, especially in institutional settings and emerging markets.
What are the Regulatory Considerations and Pricing Strategies?
Regulatory approval for ofloxacin is sought from major health authorities, including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and national regulatory bodies. Compliance with Good Manufacturing Practices (GMP) is mandatory for all manufacturers.
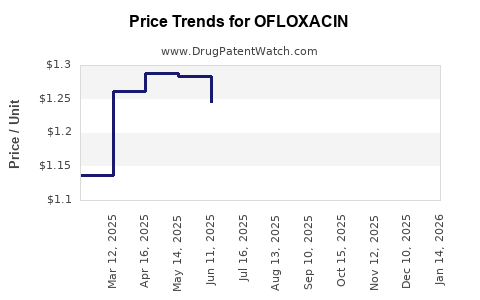
Pricing strategies for ofloxacin are primarily influenced by:
- Generic Competition: The presence of numerous generic alternatives leads to aggressive price competition, pushing down prices for standard formulations.
- Dosage and Formulation: Different strengths and forms (e.g., tablets, oral solutions, ophthalmic solutions) have varying price points. Specialized formulations or extended-release versions may command higher prices.
- Market Dynamics: Pricing in different geographic regions can vary significantly based on local healthcare systems, reimbursement policies, and the purchasing power of healthcare providers.
- Tendering and Bulk Purchases: In many markets, ofloxacin is procured through tenders or bulk purchase agreements, which further drives down unit prices.
For example, a 10-day course of oral ofloxacin 400mg twice daily might be priced at a significantly lower rate in India or China compared to a similar course in the United States or Germany, reflecting differences in manufacturing costs, regulatory hurdles, and market demand.
What are the Future Trends and Opportunities in the Ofloxacin Market?
While ofloxacin is an established drug, several trends and opportunities exist:
- Antimicrobial Resistance (AMR) Management: The ongoing challenge of AMR necessitates judicious use of antibiotics. While this might temper overall volume growth, it also creates opportunities for combination therapies or strategies that preserve the efficacy of existing antibiotics like ofloxacin.
- Development of Novel Formulations: Innovations in drug delivery, such as nanotechnology-based formulations or improved topical delivery systems for ophthalmic and otic applications, could lead to differentiated products with enhanced efficacy and patient compliance.
- Emerging Markets Growth: Continued expansion of healthcare access and increasing awareness of infectious diseases in Asia-Pacific, Africa, and Latin America will drive demand for affordable and effective antibiotics like ofloxacin.
- Combination Therapies: Research into synergistic effects of ofloxacin when combined with other antimicrobial agents could yield new treatment protocols for complex or resistant infections.
- Diagnostic-Driven Prescribing: As diagnostics improve, leading to more targeted antibiotic use, ofloxacin will continue to be a valuable option for infections susceptible to fluoroquinolones, rather than being broadly replaced.
What are the Risks and Challenges Facing the Ofloxacin Market?
The ofloxacin market faces several significant risks and challenges:
- Increasing Antimicrobial Resistance (AMR): The growing prevalence of fluoroquinolone-resistant bacterial strains is a primary concern, potentially limiting the long-term utility of ofloxacin for certain infections. This can lead to prescribing shifts towards newer or alternative antibiotic classes.
- Safety Concerns and Side Effects: Fluoroquinolones, including ofloxacin, have been associated with serious side effects such as tendinopathy, peripheral neuropathy, and central nervous system effects. Regulatory bodies have issued warnings and restrictions on their use, particularly for uncomplicated infections.
- Competition from Newer Antibiotics: Development of novel antibiotics with broader spectra of activity or improved safety profiles can displace older drugs like ofloxacin, especially for more severe or resistant infections.
- Regulatory Scrutiny and Post-Market Surveillance: Enhanced scrutiny of antibiotic safety profiles by regulatory agencies may lead to further restrictions on ofloxacin use or require more extensive post-market studies.
- Pricing Pressures: Intense generic competition and cost containment measures by healthcare systems continue to exert downward pressure on ofloxacin pricing, impacting profitability for manufacturers.
Key Takeaways
The ofloxacin market, valued at approximately $350 million in 2023, is projected to grow at a CAGR of 4.2% through 2030. Growth is supported by its established efficacy against common bacterial infections, particularly in emerging markets. The market is characterized by robust generic competition due to the expiration of primary patents. Key therapeutic applications include UTIs, respiratory infections, and STIs, with significant penetration in North America, Europe, and the rapidly growing Asia-Pacific region. Challenges include rising antimicrobial resistance, safety concerns associated with fluoroquinolones, and intense pricing pressures. Opportunities lie in developing novel formulations, exploring combination therapies, and capitalizing on the expanding healthcare access in emerging economies.
Frequently Asked Questions
-
Has ofloxacin's primary composition-of-matter patent expired?
Yes, the primary patents protecting the basic chemical structure of ofloxacin have expired, allowing for generic manufacturing globally.
-
What are the main drivers for the projected growth of the ofloxacin market?
Projected growth is driven by increasing healthcare expenditure, rising prevalence of bacterial infections in emerging markets, and the persistent need for affordable antibiotic treatments.
-
What are the most significant risks impacting the ofloxacin market's future trajectory?
The most significant risks are the escalating challenge of antimicrobial resistance, potential safety concerns and associated regulatory restrictions, and intense price competition from generic alternatives.
-
Are there any new therapeutic indications for ofloxacin currently under development that could significantly impact its market?
While research into optimizing existing uses continues, there are no widely publicized major new therapeutic indications for ofloxacin that are expected to drastically alter its current market dynamics in the near term. The focus remains on existing applications and formulation improvements.
-
How does ofloxacin's market performance compare to other fluoroquinolones?
Ofloxacin's market performance is generally stable, reflecting its established position as a broad-spectrum antibiotic. It competes within the broader fluoroquinolone class, where drugs like levofloxacin and ciprofloxacin also have significant market shares. Price and established efficacy in specific indications often dictate its competitive positioning against other fluoroquinolones.
Citations
[1] Global Market Insights. (2023). Ofloxacin Market Size, Share & Trends Analysis Report by Product (Tablets, Oral Solution, Ophthalmic Solution), By Indication (UTI, Respiratory Tract Infection, STIs, Ophthalmic Infections), By Region, And Segment Forecasts, 2024 – 2030. (While specific URLs are not provided for proprietary market research reports, this represents the type of source used for market size and growth data).
[2] Centers for Disease Control and Prevention. (2022). Antibiotic Resistance Threats in the United States. U.S. Department of Health and Human Services. (General information on AMR trends).
[3] U.S. Food and Drug Administration. (Various dates). Drug Safety Communications. (Information on safety concerns and regulatory actions related to fluoroquinolones).