UPJOHN Company Profile
✉ Email this page to a colleague
What is the competitive landscape for UPJOHN, and when can generic versions of UPJOHN drugs launch?
UPJOHN has thirteen approved drugs.
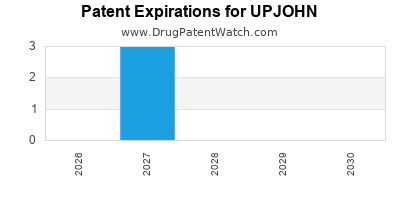
There are three US patents protecting UPJOHN drugs.
There are thirty-three patent family members on UPJOHN drugs in thirty-three countries and thirty-nine supplementary protection certificates in thirteen countries.
Drugs and US Patents for UPJOHN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Upjohn | EFFEXOR XR | venlafaxine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 020699-001 | Oct 20, 1997 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Upjohn | EFFEXOR XR | venlafaxine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 020699-002 | Oct 20, 1997 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Upjohn | LYRICA | pregabalin | CAPSULE;ORAL | 021446-008 | Dec 30, 2004 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Upjohn | EFFEXOR XR | venlafaxine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 020699-003 | Oct 20, 1997 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Upjohn | XALATAN | latanoprost | SOLUTION/DROPS;OPHTHALMIC | 020597-001 | Jun 5, 1996 | AT | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Upjohn | INSPRA | eplerenone | TABLET;ORAL | 021437-003 | Sep 27, 2002 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for UPJOHN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Upjohn | LIPITOR | atorvastatin calcium | TABLET;ORAL | 020702-002 | Dec 17, 1996 | 4,681,893*PED | ⤷ Try a Trial |
| Upjohn | EFFEXOR XR | venlafaxine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 020699-003 | Oct 20, 1997 | 4,535,186*PED | ⤷ Try a Trial |
| Upjohn | LYRICA | pregabalin | CAPSULE;ORAL | 021446-003 | Dec 30, 2004 | 5,563,175 | ⤷ Try a Trial |
| Upjohn | INSPRA | eplerenone | TABLET;ORAL | 021437-002 | Sep 27, 2002 | 6,747,020*PED | ⤷ Try a Trial |
| Upjohn | INSPRA | eplerenone | TABLET;ORAL | 021437-002 | Sep 27, 2002 | 6,863,902*PED | ⤷ Try a Trial |
| Upjohn | INSPRA | eplerenone | TABLET;ORAL | 021437-001 | Sep 27, 2002 | 7,157,101*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for UPJOHN drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 20 mg and 40 mg | ➤ Subscribe | 2010-03-29 |
| ➤ Subscribe | Extended-release Tablets | 37.5 mg, 75 mg and 150 mg | ➤ Subscribe | 2007-05-03 |
| ➤ Subscribe | Capsules | 25 mg, 50 mg, 75 mg, 100 mg, 150 mg, 200 mg, 225 mg and 300 mg | ➤ Subscribe | 2008-12-30 |
| ➤ Subscribe | Extended-release Tablets | 330 mg | ➤ Subscribe | 2018-01-29 |
| ➤ Subscribe | Extended-release Tablets | 330 mg | ➤ Subscribe | 2018-01-29 |
| ➤ Subscribe | Tablets | 25 mg and 50 mg | ➤ Subscribe | 2006-09-27 |
| ➤ Subscribe | Extended-release Capsules | 2 mg and 4 mg | ➤ Subscribe | 2007-07-30 |
| ➤ Subscribe | Capsules | 50 mg | ➤ Subscribe | 2008-03-21 |
| ➤ Subscribe | Oral Solution | 20 mg/mL | ➤ Subscribe | 2010-05-19 |
| ➤ Subscribe | Extended-release Tablets | 82.5 mg and 165 mg | ➤ Subscribe | 2018-02-02 |
| ➤ Subscribe | Extended-release Tablets | 82.5 mg and 165 mg | ➤ Subscribe | 2018-02-02 |
International Patents for UPJOHN Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Brazil | PI0618211 | ⤷ Try a Trial |
| European Patent Office | 1945186 | ⤷ Try a Trial |
| Slovenia | 1945186 | ⤷ Try a Trial |
| Netherlands | 2000281 | ⤷ Try a Trial |
| Argentina | 058175 | ⤷ Try a Trial |
| Cyprus | 1115009 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for UPJOHN Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0641330 | 2004C/022 | Belgium | ⤷ Try a Trial | PRODUCT NAME: PREGABALIN; REGISTRATION NO/DATE: EU/1/04/279/001 20040708 |
| 0641330 | CR 2004 00036 | Denmark | ⤷ Try a Trial | PRODUCT NAME: PREGABALIN ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/04/279/001-025 20040706 |
| 1003503 | 05C0048 | France | ⤷ Try a Trial | PRODUCT NAME: AMLODIPINE OU UN DE SES SELS D?ADDITION D?ACIDES PHARMACAUTIQUEMENT ACCEPTABLES/ ATORVASTATINE OU UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES; REGISTRATION NO/DATE IN FRANCE: NL 29929 DU 20050707; REGISTRATION NO/DATE AT EEC: NL 29929 DU 20050707 |
| 0641330 | 27/2004 | Austria | ⤷ Try a Trial | PRODUCT NAME: PREGABALIN, GEGEBENENFALLS IN FORM EINES PHARMAZEUTISCH ANNEHMBAREN SALZES; REGISTRATION NO/DATE: EU/1/04/279/001 - EU/1/04/279/025 20040706 |
| 1003503 | C01003503/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: AMLODIPIN + ATORVASTATIN; REGISTRATION NUMBER/DATE: SWISSMEDIC 57633 05.07.2006 |
| 3461484 | 21C1024 | France | ⤷ Try a Trial | PRODUCT NAME: ASSOCIATION DE NETARSUDIL OU L'UN DE SES SELS ET DE LATANOPROST; REGISTRATION NO/DATE: EU/1/20/1502 20210108 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.