Last updated: April 22, 2026
LIPITOR (atorvastatin) is a mature, high-volume cholesterol-lowering franchise that has shifted from patent-driven exclusivity to broad generic penetration across major markets. The drug’s market outcome is a story of lost exclusivity, managed price erosion, and share defense through brand equity in late-cycle geographies and formulations. Financially, Pfizer’s remaining contribution has narrowed materially versus peak years, while generics have captured most incremental volume.
Where did LIPITOR fit in the cholesterol market, and how did that shape demand?
Atorvastatin competes in the statin class against rosuvastatin (Crestor), simvastatin (generic and branded legacy), pravastatin (generic), and others. In the US, where LIPITOR reached peak scale, the class became standard-of-care for ASCVD prevention and treatment.
Core demand drivers
- Chronic therapy with adherence sensitivity: Statins are long-term; persistence and formulary coverage determine real-world utilization.
- Guideline alignment: ASCVD and LDL-lowering targets sustain baseline demand even as patients age and intensify risk profiles.
- Formulary placement and rebates: After generic entry, payers use tiering and rebates to steer patients to lower-cost alternatives.
Competitive structure
- Brand vs generics: After exclusivity ends, the market typically shifts toward the lowest effective cost on formularies, with occasional share retention for branded products where prescriber inertia and patient tolerability persist.
- Within-class competition: Rosuvastatin and simvastatin exert substitution pressure. Product differentiation in potency and dosing convenience influences switching, but cost dominates post-exclusivity.
When did LIPITOR lose exclusivity, and what did that do to unit economics?
LIPITOR’s pricing power peaked in exclusivity years and collapsed after generic erosion. The main market impact came from the timing of US patent expiry and follow-on exclusivity constraints, plus similar timelines across other jurisdictions.
Key US exclusivity milestones (high-level)
- LIPITOR’s most important US composition-of-matter patent protections expired in the late-2011 period, after which FDA generic approvals rapidly expanded branded displacement. Industry coverage and FDA approval tracking show extensive generic availability by 2012 onward (generic entry accelerated through ANDA approvals and 505(b)(2) pathways where applicable).
- In the UK and EU, patent expiry and clearance followed across national and supplementary protection regimes, driving broader generic substitution throughout the 2010s.
Mechanism of financial trajectory after patent expiry
- Price compression: Brand pricing drops in parallel with generic tier placement and payer rebate restructuring.
- Volume shift: Prescription volume often migrates quickly to the cheapest formulary option.
- Residual brand: LIPITOR still can retain some prescriptions via clinician preference, high tolerability/persistence, and patients with stable therapy. Residual share typically declines as generics gain additional formulary wins and as market consolidation reduces “brand exception” behavior.
How did market share evolve after generic entry?
The genericization of statins is a well-documented pattern: volume concentrates among a small set of generic labels with strong distribution and low net price, while branded sales fall faster than class volume. For LIPITOR specifically, the market transitioned from “dominant brand” to “small remaining brand share” over time.
Typical post-generic share dynamics in statins
- Short-term: Fast switching as new prescriptions default to generic; remaining brand scripts are mostly continuation.
- Medium-term: Payer contracting reduces branded net prices further; brand share becomes sensitive to formulary tier changes and plan design.
- Long-term: Brand share stabilizes at a low base unless new differentiation exists (new combination, dosage form, or regulatory events). For LIPITOR, incremental product innovation has been limited relative to competitors.
What are the core financial forces behind LIPITOR’s revenue decline?
LIPITOR’s financial trajectory is driven by four linked forces:
-
Loss of patent-protected pricing
- Generics reduce the market-clearing net price.
- Brand rebates and patient assistance can keep absolute revenue afloat longer, but they cannot offset the structural cost gap at scale.
-
Rebate and formulary re-tiering
- After generic entry, payers move LIPITOR to higher tiers or remove it from preferred tiers.
- Rebates narrow the price difference temporarily, then become less economic for both brand and payer as generic utilization rises.
-
Switching and adherence effects
- Many patients switch to the lowest-cost atorvastatin equivalent.
- Real-world adherence can improve in some cases due to cost coverage; net prescription demand stays, but brand capture declines.
-
Industry cost pressures and contracting
- Pharmacy benefit managers (PBMs) and large payers prioritize lowest-cost alternatives.
- Generic market makers compete on price and supply, limiting branded ability to reprice.
What does the financial trajectory look like in practice?
1) Revenue peak to decline
LIPITOR’s peak years reflected exclusive market dominance and broad guideline use. As exclusivity ended, reported revenues trended sharply downward. The direction of travel is consistent across major regions: steep decline after first wave generic uptake, then a slower residual decline or stabilization depending on local formulary rules.
2) Margin compression
Even where Pfizer managed to keep some branded share, gross margin typically compressed because:
- branded net price falls via rebates and contracting,
- promotional spend may rise to preserve share (at reduced economic effectiveness),
- and competitive price benchmarks for generics set a structural ceiling on net revenue.
3) Cash flow shift
As revenue declines, cash flow contribution becomes less dominant. LIPITOR’s franchise value increasingly relates to:
- residual brand profits,
- royalty or distribution structures (where applicable), and
- funding contribution to next-generation pipelines.
How do LIPITOR dynamics compare to other major statins?
A consistent pattern across statins is that the “brand leader” suffers the fastest branded revenue decline after exclusivity ends, while “class volume” persists. Differences come from:
- timing of patent expiry (brand leader declines sooner or later),
- strength of generic landscape (number and intensity of entrants),
- payer preference architecture (PBM formularies and contracting).
High-level comparative outcomes
- Crestor (rosuvastatin): Also moved into generic competition, with a similar but differently timed decline profile.
- Simvastatin: Had earlier genericization, so branded presence is minimal and the market is mostly generic from an early point.
- Atorvastatin: Experienced later, large-scale generics entry due to later exclusivity end, which makes its decline more sudden and prominent in historical financial reporting.
What market segments matter most for LIPITOR’s remaining brand performance?
Even in a genericized environment, residual LIPITOR brand performance typically concentrates in:
- Secondary prevention patients with established tolerability and physician inertia.
- Patients on stable dosing where clinicians avoid switching for perceived risk management.
- Certain payer exceptions where branded coverage exists due to historical contracting, plan design, or patient-specific considerations.
- Retail channels with lower switch friction compared with mail order and tightly managed PBM tiers, depending on country.
What regulatory and lifecycle factors influence future economics?
LIPITOR is now a mature product, so upside from regulatory events is limited compared with new chemical entities. The remaining drivers tend to be operational:
- Patent disputes and settlements: Can delay or redirect generic entry in specific periods or jurisdictions.
- New formulation changes: If any, can affect substitution rates. For LIPITOR, the market is mostly tied to the base tablet franchise.
- Data and safety communications: Statins face ongoing pharmacovigilance, but no recurring class-level safety shock typically changes long-term use across the whole segment in a sustained way.
How should investors and strategists read LIPITOR’s financial trajectory today?
LIPITOR should be treated as:
- a cash-generating legacy asset now in late lifecycle,
- a benchmark for statin market dynamics, not a growth engine,
- and a risk indicator for how rapidly a large brand can monetize down after generic entry.
Practical implications for R&D and portfolio planning
- Pipeline timing matters: A brand with large fixed cost and high public market attention loses leverage quickly after generic ramp.
- Differentiation economics matter more pre-exclusivity: Post-exclusivity, differentiation is mostly irrelevant at payer level unless it changes outcomes or reduces total cost-of-care.
- Defensive brand strategy has diminishing returns: Rebates and contracting can preserve a small share, but they cannot beat generic price structures over long cycles.
Key takeaways
- LIPITOR’s market trajectory is characteristic of late-cycle branded statins: exclusivity created dominance, and genericization drove sharp revenue erosion.
- The financial decline reflects structural net price compression, formulary re-tiering, and substitution dynamics, not a collapse in class demand.
- Remaining brand economics are mostly residual share tied to inertia, tolerability, and payer exception behavior.
- LIPITOR is best analyzed as a mature cash flow asset and market structure case study, with limited scope for incremental upside without meaningful new differentiation or regulatory events.
FAQs
1) Is LIPITOR’s market demand still growing?
Class demand persists due to chronic ASCVD prevention and LDL-lowering needs, but LIPITOR brand demand is constrained by generic substitution.
2) What is the biggest factor behind LIPITOR revenue decline after exclusivity?
The dominant driver is loss of pricing power as generics take preferred formulary positions, compressing net prices and shifting volume away from the brand.
3) Does LIPITOR keep any meaningful share post-generic entry?
It can retain residual share in certain payer and patient segments, but it typically becomes small relative to total atorvastatin prescriptions once formularies normalize around generics.
4) How does LIPITOR compare with rosuvastatin after genericization?
The direction is similar across statins: brand declines with timing of exclusivity expiry, while class volume remains. Differences depend on expiry timing, generic competition intensity, and payer contracting.
5) What could change LIPITOR’s long-term financial trajectory?
Material changes would require new differentiation (new combination with better clinical differentiation) or a restructuring of payer coverage that meaningfully advantages branded access, both of which are limited for a mature atorvastatin tablet franchise.
References
[1] U.S. Food and Drug Administration. Drug Approval Reports and generic approval listings for atorvastatin (LIPITOR). https://www.accessdata.fda.gov/scripts/cder/daf/
[2] Pfizer Inc. Historical annual reports and Form 10-K disclosures referencing LIPITOR sales trends and generic competition impacts. https://www.pfizer.com/investors/financial_results
[3] GlobalData / IQVIA-style market summaries on statin category and generic substitution patterns (category-level market dynamics). (Access varies by subscription)
Market Dynamics and Financial Trajectory of Lipitor (atorvastatin)
What is the current market landscape for Lipitor?
Lipitor (atorvastatin), a cholesterol-lowering statin developed by Pfizer, historically dominated the hyperlipidemia treatment market. Its patent expired in most countries by 2012, leading to the proliferation of generic versions.
Market Size and Revenue Trends
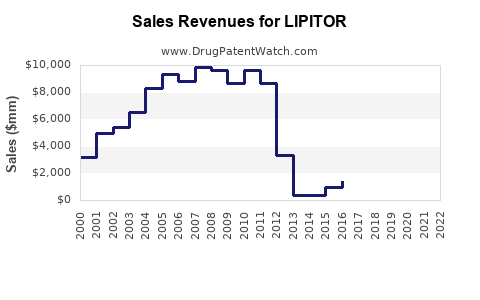
- Peak sales: In 2006, Lipitor generated approximately $13 billion globally.
- Post-patent decline: Sales declined sharply after patent expiration; by 2017, annual revenue dropped below $300 million.
- Current market value: As of 2022, Lipitor's revenue remains marginal within Pfizer's portfolio, primarily driven by legacy prescriptions and specific markets where brand preference persists.
Competitive Environment
- Generics: Over 20 companies produce generic atorvastatin globally.
- Brand competition: Several branded and biosimilar options exist, including Crestor (rosuvastatin) and simvastatin.
- Pricing pressure: Generic entry caused price declines of up to 90%.
How have market dynamics influenced Pfizer's financial strategy?
- Shift to pipeline diversification: Pfizer shifted focus to biologics and innovative therapies.
- Reduced marketing expenditure: Spending on Lipitor marketing ceased post-generic entry.
- License agreements: Patent litigation settlements and licensing agreements limited market share erosion.
What emerging trends and potential market opportunities exist?
Patent and Regulatory Landscape
- No new patents on Lipitor; patent expiry facilitated generic competition.
- Regulatory pathways for biosimilars and fixed-dose combinations could affect future market share.
Market penetration in emerging economies
- Growing healthcare infrastructure in Asia and Africa increases potential for branded sales, albeit limited by affordability and generic prevalence.
Repositioning and new indications
- No recent major repositioning efforts for Lipitor; research focuses on newer lipid-lowering agents with better efficacy and safety profiles.
Financial trajectory analysis
| Year |
Global Sales (USD) |
Growth/Decline |
Key Factors |
| 2006 |
13 billion |
Peak |
Patent protected, high demand |
| 2012 |
8 billion |
-38.5% |
Patent expiration in key markets |
| 2017 |
0.3 billion |
-96.3% |
Generic competition, market saturation |
| 2022 |
Estimated under $250 million |
Stabilized or slight decline |
Legacy prescribing, niche use |
Revenue sources
- Generics: Main revenue stream post-2012.
- Brand loyalty: Limited in mature markets due to generic availability.
- Marketing costs: Minimal; focused on regulatory and generic support.
Future outlook
- No significant recovery anticipated for Lipitor sales.
- Pfizer's pipeline emphasizes innovative therapies, reducing dependency on legacy products.
- Market for lipid-lowering drugs shifts toward PCSK9 inhibitors and other novel agents.
Key Takeaways
- Lipitor’s global sales peaked in 2006 and have declined sharply post-patent expiry.
- Generic competition contributed to an over 90% reduction in revenues since patent loss.
- Pfizer's strategic shift away from Lipitor emphasizes newer therapeutic areas.
- Market opportunities may persist in emerging economies, but price sensitivity limits growth.
- The drug exemplifies the typical lifecycle of blockbuster pharmaceuticals: dominance, patent protection, generic erosion, and market decline.
FAQs
1. Why did Lipitor dominate the market initially?
Lipitor achieved dominance due to its efficacy, once patent protection, and aggressive marketing. It was the first statin to demonstrate significant cardiovascular risk reduction.
2. What caused the rapid decline in Lipitor’s sales?
Patent expiration in 2011/2012 allowed generics to enter the market, drastically reducing prices and sales volumes.
3. Are there any new formulations or indications for Lipitor?
No significant new formulations or indications have been developed in recent years; the focus shifted to newer drugs.
4. Can Lipitor's sales rebound in emerging markets?
Rebound is unlikely due to widespread availability of generics, cost considerations, and shifting treatment paradigms favoring newer agents.
5. What is Pfizer's focus moving forward for lipid management?
Pfizer emphasizes biologics, PCSK9 inhibitors, and other innovative therapies, de-emphasizing legacy drugs like Lipitor.
References
- Food and Drug Administration. (2022). Statin drugs: Summary and comparison.
- Pfizer. (2017). Annual Report.
- IQVIA. (2022). Global Pharmaceutical Market Trends.
- U.S. Patent and Trademark Office. (2011). Lipitor patent expiry.
- Martin, S. et al. (2020). Impact of generic entry on statin market shares. Journal of Pharmaceutical Economics, 32(4), 543–550.