Sumitomo Pharma Am Company Profile
✉ Email this page to a colleague
What is the competitive landscape for SUMITOMO PHARMA AM, and when can generic versions of SUMITOMO PHARMA AM drugs launch?
SUMITOMO PHARMA AM has three approved drugs.
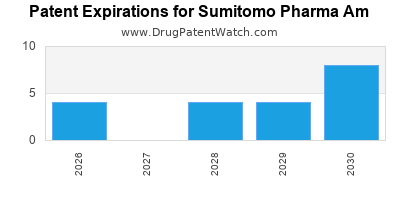
There are thirty-six US patents protecting SUMITOMO PHARMA AM drugs.
There are four hundred and two patent family members on SUMITOMO PHARMA AM drugs in thirty-three countries and twelve supplementary protection certificates in seven countries.
Summary for Sumitomo Pharma Am
| International Patents: | 402 |
| US Patents: | 36 |
| Tradenames: | 3 |
| Ingredients: | 3 |
| NDAs: | 3 |
Drugs and US Patents for Sumitomo Pharma Am
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-005 | May 21, 2020 | DISCN | Yes | No | 9,669,019 | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Sumitomo Pharma Am | APTIOM | eslicarbazepine acetate | TABLET;ORAL | 022416-002 | Nov 8, 2013 | AB | RX | Yes | No | 10,702,536 | ⤷ Sign Up | ⤷ Sign Up | |||
| Sumitomo Pharma Am | APTIOM | eslicarbazepine acetate | TABLET;ORAL | 022416-002 | Nov 8, 2013 | AB | RX | Yes | No | 9,643,929 | ⤷ Sign Up | Y | ⤷ Sign Up | ||
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-001 | May 21, 2020 | DISCN | Yes | No | 10,821,074 | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-001 | May 21, 2020 | DISCN | Yes | No | 10,449,146 | ⤷ Sign Up | ⤷ Sign Up | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Sumitomo Pharma Am
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Sumitomo Pharma Am | LONHALA MAGNAIR KIT | glycopyrrolate | SOLUTION;INHALATION | 208437-001 | Dec 5, 2017 | 7,316,067 | ⤷ Sign Up |
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-002 | May 21, 2020 | 9,931,305 | ⤷ Sign Up |
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-001 | May 21, 2020 | 9,855,221 | ⤷ Sign Up |
| Sumitomo Pharma Am | KYNMOBI | apomorphine hydrochloride | FILM;SUBLINGUAL | 210875-001 | May 21, 2020 | 11,077,068 | ⤷ Sign Up |
| Sumitomo Pharma Am | APTIOM | eslicarbazepine acetate | TABLET;ORAL | 022416-003 | Nov 8, 2013 | 5,753,646 | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Sumitomo Pharma Am Drugs
Supplementary Protection Certificates for Sumitomo Pharma Am Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2435024 | SPC/GB21/029 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: A COMBINATION OF FORMOTEROL, INCLUDING PHARMACEUTICALLY ACCEPTABLE SALTS, ESTERS AND SOLVATES THEREOF, GLYCOPYRROLATE, INCLUDING PHARMACEUTICALLY ACCEPTABLE SALTS, ESTERS AND SOLVATES THEREOF, AND BUDESONIDE INCLUDING PHARMACEUTICALLY ACCEPTABLE SALTS, ES; REGISTERED: UK EU/1/20/1498 (NI) 20201210; UK PLGB 17901/0352-001 20201210 |
| 2214636 | C02214636/01 | Switzerland | ⤷ Sign Up | PRODUCT NAME: ESLICARBAZEPINACETAT; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 67375 02.04.2020 |
| 2435025 | LUC00124 | Luxembourg | ⤷ Sign Up | PRODUCT NAME: UNE COMBINAISON DE GLYCOPYRROLATE (Y COMPRIS SES SELS, ESTERS, ENANTIOMERES OU AUTRES DERIVES PHARMACEUTIQUEMENT ACCEPTABLES) ET DE FORMOTEROL (Y COMPRIS SES SELS, ESTERS, ENANTIOMERES OU AUTRES DERIVES PHARMACEUTIQUEMENT ACCEPTABLES); AUTHORISATION NUMBER AND DATE: EU/1/18/1339 20181220 |
| 2435024 | 21C1020 | France | ⤷ Sign Up | PRODUCT NAME: COMBINAISON DE FORMOTEROL (Y COMPRIS LES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES DE CELUI-CI), GLYCOPYRROLATE (Y COMPRIS LES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES DE CELUI-CI) ET BUDESONIDE (Y COMPRIS LES SELS, ESTERS, SOLVATES OU ENANTIOMERES PHARMACEUTIQUEMENT ACCEPTABLES DE CELUI-CI); REGISTRATION NO/DATE: EU/1/20/1498 20201210 |
| 1915346 | C01915346/01 | Switzerland | ⤷ Sign Up | PRODUCT NAME: ESLICARBAZEPINACETAT; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 67375 02.04.2020 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.