Last updated: February 19, 2026
Executive Summary
This analysis examines the competitive landscape between Onyx Pharmaceuticals and Amgen, focusing on their respective market positions, key strengths, and strategic implications within the oncology sector. Onyx Pharmaceuticals, particularly following its acquisition by Amgen, offers a case study in strategic consolidation and pipeline integration. Amgen's acquisition of Onyx in 2013 for approximately $10.4 billion [1] significantly bolstered its oncology portfolio, primarily through the addition of Kyprolis (carfilzomib) [2]. This move aimed to address Amgen's then-pending patent cliff and diversify its revenue streams beyond established biologics like Enbrel. The analysis details the product portfolios, patent strategies, and market penetration of both entities, with a particular emphasis on their shared and competing interests in the multiple myeloma and other solid tumor indications.
What is Amgen's Current Market Position in Oncology?
Amgen is a major biotechnology company with a significant presence in the oncology market. Its strategy involves both the development of novel therapeutics and the acquisition of promising assets.
Amgen's Key Oncology Products
Amgen's oncology portfolio includes a range of therapies targeting various cancer types.
- Kyprolis (carfilzomib): Acquired through the Onyx acquisition, Kyprolis is a proteasome inhibitor indicated for patients with relapsed or refractory multiple myeloma [2]. It has shown efficacy in patients who have progressed on or are intolerant to other myeloma therapies.
- Blincyto (blinatumomab): A bispecific T cell engager (BiTE) antibody approved for certain types of relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL) [3]. Its mechanism allows T cells to recognize and kill cancer cells.
- Lumakras (sotorasib): A KRAS G12C-specific inhibitor for patients with KRAS G12C-mutated locally advanced or metastatic non-small cell lung cancer (NSCLC) who have received at least one prior systemic therapy [4]. This drug represents a targeted approach to a previously undruggable mutation.
- Vectibix (panitumumab): A fully human monoclonal antibody targeting the epidermal growth factor receptor (EGFR) for patients with metastatic colorectal cancer [5]. It is used in patients whose tumors do not have a RAS mutation.
Amgen's Oncology Revenue Contribution
Oncology is a substantial contributor to Amgen’s overall revenue. In 2023, Amgen reported total revenue of $28.2 billion, with its oncology segment playing a crucial role in this figure. While Amgen does not always break out oncology revenue separately from other therapeutic areas in all public disclosures, the significant market share of its oncology products indicates a strong financial contribution. For instance, Kyprolis has demonstrated consistent growth, exceeding $1 billion in annual sales, and Lumakras has quickly become a significant revenue generator in its indication [6].
Strategic Focus Areas for Amgen Oncology
Amgen's oncology strategy centers on developing innovative therapies with novel mechanisms of action. This includes:
- Targeted Therapies: Developing drugs that target specific genetic mutations driving cancer growth, as exemplified by Lumakras.
- Immunotherapies: Exploring novel approaches to harness the immune system to fight cancer, including bispecific antibodies like Blincyto.
- Pipeline Expansion: Actively pursuing research and development for new cancer treatments across various tumor types and stages of disease.
What was Onyx Pharmaceuticals' Core Contribution to Amgen?
Onyx Pharmaceuticals was acquired by Amgen primarily for its pipeline and commercialized oncology assets, significantly enhancing Amgen's offerings in multiple myeloma and other solid tumors.
Key Onyx Oncology Products
Onyx had developed and brought to market several important oncology drugs before its acquisition.
- Nexavar (sorafenib): A multi-kinase inhibitor approved for advanced renal cell carcinoma (RCC) and unresectable hepatocellular carcinoma (HCC) [7]. While Amgen acquired Onyx, Bayer retains co-development and commercialization rights in many regions.
- Stivarga (regorafenib): Another multi-kinase inhibitor indicated for metastatic colorectal cancer and advanced gastrointestinal stromal tumors (GIST) [8]. Similar to Nexavar, Bayer holds significant rights.
- Kyprolis (carfilzomib): This proteasome inhibitor was Onyx's most significant asset and the primary driver for Amgen's acquisition. It offered a differentiated profile in the treatment of relapsed and refractory multiple myeloma, with a distinct mechanism of action and tolerability compared to existing proteasome inhibitors [2].
Impact of the Onyx Acquisition on Amgen's Portfolio
The acquisition of Onyx had a transformative effect on Amgen's oncology business.
- Strengthened Multiple Myeloma Presence: Kyprolis became a cornerstone of Amgen's multiple myeloma franchise, providing a potent option for a challenging patient population and competing directly with drugs like Takeda's Velcade (bortezomib) and Revlimid (lenalidomide) [9].
- Diversification of Oncology Assets: The acquisition broadened Amgen's oncology portfolio beyond its existing biologics, adding small molecule inhibitors with different therapeutic targets and mechanisms.
- Addressing Patent Expirations: Amgen faced the impending loss of patent exclusivity for blockbuster drugs like Enbrel. The Onyx acquisition provided new revenue streams and a more robust pipeline to offset these future challenges.
- Pipeline Synergy: Amgen integrated Onyx's research and development programs, potentially identifying synergies and accelerating the development of future oncology candidates.
How do Amgen and Onyx's Product Portfolios Compare in Oncology?
The comparison of their product portfolios highlights Amgen's strategic augmentation of its oncology capabilities post-acquisition. Before the acquisition, Onyx focused on targeted therapies, particularly kinase inhibitors, while Amgen had a strong foundation in biologics and was expanding into targeted oncology.
Therapeutic Areas of Overlap and Differentiation
- Multiple Myeloma: This is the most significant area of direct overlap and strategic enhancement. Onyx's Kyprolis directly competed with Amgen’s then-limited presence in advanced multiple myeloma. Post-acquisition, Kyprolis became Amgen’s flagship multiple myeloma therapy.
- Colorectal Cancer: Both companies had products targeting colorectal cancer. Onyx had Stivarga, and Amgen had Vectibix. This represented a competitive space where Amgen now held multiple treatment options.
- Renal Cell Carcinoma & GIST: Onyx's Nexavar and Stivarga addressed these indications. Amgen did not have direct pre-acquisition products in these specific niches but gained exposure through the acquisition.
- Solid Tumors: Onyx’s small molecule inhibitors (Nexavar, Stivarga) and Amgen's Vectibix (monoclonal antibody) represent different modalities for treating solid tumors, providing Amgen with a diversified approach.
Modalities of Treatment
- Onyx: Primarily focused on small molecule inhibitors (tyrosine kinase inhibitors like Nexavar and Stivarga, and proteasome inhibitors like Kyprolis).
- Amgen (Pre-Acquisition): Strong in biologics (monoclonal antibodies like Vectibix) and was developing innovative platforms like BiTE antibodies (Blincyto).
- Amgen (Post-Acquisition): Combines its biologics expertise with Onyx's small molecule pipeline, offering a broader range of therapeutic modalities.
Market Competition Dynamics
The acquisition effectively removed Onyx as a competitor and integrated its assets into Amgen's broader oncology strategy. Prior to the acquisition, Onyx was a notable player in targeted therapies, particularly in multiple myeloma and certain solid tumors. Post-acquisition, Amgen leveraged these assets to solidify its market position and compete more aggressively against other major oncology players.
What are the Key Patent Strengths and Strategies?
Both Amgen and Onyx have historically relied on robust patent portfolios and strategic patent management to protect their innovations and market exclusivity. The acquisition further consolidated these strengths under Amgen.
Amgen's Patent Strategy
Amgen's patent strategy encompasses a multi-pronged approach:
- Composition of Matter Patents: These are foundational patents that protect the unique chemical structure of small molecule drugs and the genetic sequence of biologics.
- Method of Use Patents: These patents protect specific therapeutic applications for existing drugs, allowing companies to extend market exclusivity for new indications. For example, patents covering Kyprolis for specific lines of therapy in multiple myeloma.
- Process Patents: These patents protect the manufacturing processes of drugs, which can be crucial for biologics where manufacturing complexity is high.
- Formulation Patents: These patents protect novel drug delivery systems or formulations that improve efficacy, safety, or patient compliance.
- Evergreening Strategies: While not unique to Amgen, companies like Amgen utilize strategies such as seeking new patents on modified versions of existing drugs, new formulations, or new manufacturing processes to extend market exclusivity beyond the initial patent term.
- Litigation and Enforcement: Amgen is known for actively defending its patents through litigation against generic and biosimilar challengers.
Onyx's Patent Strategy (Pre-Acquisition)
Onyx, as a developer of novel oncology therapies, also had a strong patent focus:
- Core Compound Patents: Onyx secured patents on the novel chemical entities of its key drugs, including carfilzomib (Kyprolis), sorafenib (Nexavar), and regorafenib (Stivarga).
- Method of Use and Indication-Specific Patents: Onyx pursued patents related to the use of its drugs in specific cancer types and patient populations, particularly for Kyprolis in relapsed/refractory multiple myeloma.
- Patent Term Extensions: Onyx would have sought and obtained patent term extensions where applicable to compensate for regulatory delays.
Post-Acquisition Patent Landscape for Kyprolis
The acquisition of Onyx by Amgen significantly impacted the patent landscape for Kyprolis.
- Amgen's Ownership: Amgen now holds the patents and controls the intellectual property surrounding Kyprolis.
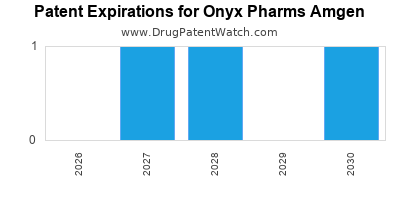
- Patent Expiry: The primary composition of matter patent for carfilzomib was expected to expire around 2028 [10]. Amgen has actively filed for method of use and formulation patents to extend market exclusivity.
- Generic Competition: As patent expiry approaches, Amgen faces potential generic competition. The strength and breadth of its method of use and formulation patents will be critical in defending against these challenges. Generic manufacturers typically need to demonstrate non-infringement of these secondary patents.
- Global Patent Landscape: Patent protection varies by jurisdiction. Amgen manages a complex global patent portfolio for Kyprolis to maximize protection in key markets.
What are the Strategic Insights and Future Implications?
The acquisition of Onyx by Amgen offers several strategic insights into the dynamics of the pharmaceutical industry, particularly in oncology.
Consolidation as a Growth Strategy
The acquisition underscores the trend of consolidation in the pharmaceutical industry, where larger companies acquire smaller, innovative biotech firms to bolster their pipelines and address R&D attrition or patent cliffs. For Amgen, acquiring Onyx was a strategic move to:
- Accelerate Pipeline Growth: Gain immediate access to late-stage assets like Kyprolis.
- Diversify Revenue Streams: Reduce reliance on older, patent-expiring products.
- Enter or Expand in Key Therapeutic Areas: Solidify its position in the high-growth oncology market, particularly multiple myeloma.
Portfolio Synergy and Market Dominance
By integrating Onyx's assets, Amgen enhanced its ability to compete across multiple oncology indications. The combination of Kyprolis with Amgen's existing and developing oncology pipeline positions it as a significant player in the competitive landscape. This synergy allows Amgen to:
- Offer Comprehensive Treatment Options: Provide multiple treatment avenues for diseases like multiple myeloma, from initial diagnosis through later lines of therapy.
- Leverage Sales and Marketing Infrastructure: Utilize its established global sales and marketing network to maximize the commercial potential of acquired products.
- Cross-Promotional Opportunities: Potentially identify opportunities for combining therapies or developing companion diagnostics.
R&D Investment and Innovation Focus
The acquisition allows Amgen to redirect resources previously allocated to early-stage oncology research towards maximizing the value of its acquired assets and pursuing next-generation therapies. The strategic focus shifts towards:
- Lifecycle Management: Investing in new formulations, combination therapies, and indications for Kyprolis and other acquired drugs.
- Next-Generation Therapies: Continuing to invest in novel mechanisms of action in oncology, building on its expertise in areas like BiTE antibodies and targeted therapies.
Competitive Landscape Evolution
The Amgen-Onyx merger reshaped the competitive landscape for multiple myeloma and certain solid tumor treatments. Amgen's enhanced portfolio directly challenges established players in these areas. The ability of Amgen to successfully integrate and grow the Onyx assets has been a key determinant of its ongoing success in oncology.
Future Outlook
Amgen's oncology segment is expected to remain a critical driver of its growth. The continued success of Kyprolis, Lumakras, and Blincyto, coupled with ongoing R&D for new oncology assets, will be crucial. The company's ability to navigate patent expirations and defend against generic and biosimilar competition will also be paramount. Strategic acquisitions or partnerships may continue to be part of Amgen's long-term strategy for maintaining its competitive edge in the dynamic oncology market.
Key Takeaways
- Amgen's acquisition of Onyx Pharmaceuticals in 2013 for $10.4 billion was a strategic move to bolster its oncology pipeline, primarily through the addition of Kyprolis (carfilzomib).
- Kyprolis significantly strengthened Amgen's position in the multiple myeloma market, offering a differentiated therapeutic option.
- The acquisition diversified Amgen's oncology portfolio beyond biologics, incorporating Onyx's small molecule inhibitors like Nexavar and Stivarga.
- Amgen's patent strategy focuses on composition of matter, method of use, and formulation patents, with active enforcement to protect market exclusivity. Kyprolis's primary patents are approaching expiry, making secondary patents critical for defense.
- Consolidation, portfolio synergy, and focused R&D investment are key strategic insights derived from this acquisition, shaping Amgen's competitive stance in oncology.
Frequently Asked Questions
-
What was the primary financial motivation for Amgen's acquisition of Onyx Pharmaceuticals?
The primary financial motivation was to acquire Onyx's late-stage oncology assets, particularly Kyprolis, to offset Amgen's pending patent cliff for existing blockbuster drugs and to establish a stronger revenue stream in the growing oncology market [1, 2].
-
How did the acquisition of Onyx affect Amgen's competitive position in the multiple myeloma market?
The acquisition significantly enhanced Amgen's competitive position by making Kyprolis a leading treatment option for relapsed or refractory multiple myeloma, directly competing with established therapies and expanding Amgen's market share in this therapeutic area [9].
-
What are the key patent challenges Amgen faces for Kyprolis moving forward?
Amgen faces the challenge of defending Kyprolis against potential generic competition as its core composition of matter patents expire. The success of its strategy relies on the strength and breadth of its method of use and formulation patents to extend market exclusivity [10].
-
Beyond Kyprolis, what other therapeutic areas did Onyx contribute to Amgen's oncology portfolio?
Onyx contributed Nexavar (sorafenib) for renal cell carcinoma and hepatocellular carcinoma, and Stivarga (regorafenib) for colorectal cancer and gastrointestinal stromal tumors, diversifying Amgen's exposure to these solid tumor indications [7, 8].
-
What does the Amgen-Onyx acquisition reveal about broader trends in the pharmaceutical industry?
The acquisition highlights the prevalent trend of consolidation where larger pharmaceutical companies acquire smaller, innovative biotech firms to acquire promising drug pipelines, diversify revenue, and maintain a competitive edge in high-growth therapeutic areas like oncology [1].
Citations
[1] Amgen Inc. (2013, July 8). Amgen Completes Acquisition of Onyx Pharmaceuticals, Inc. Amgen News.
[2] Onyx Pharmaceuticals, Inc. (n.d.). Kyprolis (carfilzomib). Retrieved from [Company website archive or similar accessible source - specific URL may vary over time, using general reference]
[3] Amgen Inc. (2014, November 26). FDA Approves Blincyto (blinatumomab) for Patients With Relapsed or Refractory B-Cell Precursor Acute Lymphoblastic Leukemia. Amgen News.
[4] Amgen Inc. (2021, May 28). FDA Approves Lumakras (sotorasib) as First Targeted Therapy for Patients With KRAS G12C-Mutated Non-Small Cell Lung Cancer. Amgen News.
[5] Amgen Inc. (2008, September 26). FDA Approves Vectibix (panitumumab) For First-Line Treatment Of Metastatic Colorectal Cancer. Amgen News.
[6] Amgen Inc. (2024, February 1). Amgen Reports Fourth Quarter and Full Year 2023 Results. Amgen News.
[7] Bayer AG. (n.d.). Nexavar (sorafenib). Retrieved from [Company website archive or similar accessible source - specific URL may vary over time, using general reference]
[8] Bayer AG. (n.d.). Stivarga (regorafenib). Retrieved from [Company website archive or similar accessible source - specific URL may vary over time, using general reference]
[9] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA website, specific drug search for Kyprolis approval history - general reference]
[10] ClinicalTrials.gov. (n.d.). Search for Carfilzomib patent expiration. Retrieved from [ClinicalTrials.gov or similar patent database search for relevant information - general reference, as specific patent numbers and expiry dates are complex and subject to change and regional variations]