ORGANON Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ORGANON, and when can generic versions of ORGANON drugs launch?
ORGANON has fifty-nine approved drugs.
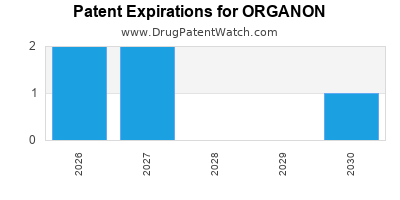
There are six US patents protecting ORGANON drugs. There is one tentative approval on ORGANON drugs.
There are three hundred and thirteen patent family members on ORGANON drugs in forty-four countries and one hundred and thirty-six supplementary protection certificates in fifteen countries.
Drugs and US Patents for ORGANON
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Organon Llc | XACIATO | clindamycin phosphate | GEL;VAGINAL | 215650-001 | Dec 7, 2021 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Organon | VYTORIN | ezetimibe; simvastatin | TABLET;ORAL | 021687-002 | Jul 23, 2004 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Organon Llc | SINEMET CR | carbidopa; levodopa | TABLET, EXTENDED RELEASE;ORAL | 019856-002 | Dec 24, 1992 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Organon | FOSAMAX | alendronate sodium | TABLET;ORAL | 020560-001 | Sep 29, 1995 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Organon Usa Inc | LIQUAEMIN SODIUM PRESERVATIVE FREE | heparin sodium | INJECTABLE;INJECTION | 000552-012 | Apr 11, 1986 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ORGANON
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Organon Llc | CLARINEX-D 12 HOUR | desloratadine; pseudoephedrine sulfate | TABLET, EXTENDED RELEASE;ORAL | 021313-001 | Feb 1, 2006 | 7,214,684*PED | ⤷ Try a Trial |
| Organon | FOSAMAX | alendronate sodium | TABLET;ORAL | 020560-004 | Oct 20, 2000 | 5,849,726*PED | ⤷ Try a Trial |
| Organon | HYZAAR | hydrochlorothiazide; losartan potassium | TABLET;ORAL | 020387-001 | Apr 28, 1995 | 5,608,075 | ⤷ Try a Trial |
| Organon Llc | MAXALT | rizatriptan benzoate | TABLET;ORAL | 020864-001 | Jun 29, 1998 | 5,602,162*PED | ⤷ Try a Trial |
| Organon | COZAAR | losartan potassium | TABLET;ORAL | 020386-001 | Apr 14, 1995 | 5,210,079*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for ORGANON drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Orally Disintegrating Tablets | 2.5 mg and 5 mg | ➤ Subscribe | 2006-06-21 |
| ➤ Subscribe | Oral Solution | 70 mg/75 mL | ➤ Subscribe | 2007-09-07 |
| ➤ Subscribe | Tablets | 100 mg/12.5 mL | ➤ Subscribe | 2006-04-04 |
| ➤ Subscribe | Tablets | 10 mg | ➤ Subscribe | 2006-10-25 |
| ➤ Subscribe | Tablets | 70 mg/2800 IU and 70 mg/5600 IU | ➤ Subscribe | 2007-11-20 |
| ➤ Subscribe | Injection | 250 mcg/0.5 mL, 1 mL PFS | ➤ Subscribe | 2012-03-30 |
| ➤ Subscribe | Tablets | 10 mg | ➤ Subscribe | 2007-02-20 |
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2006-06-21 |
| ➤ Subscribe | Orally Disintegrating Tablets | 5 mg and 10 mg | ➤ Subscribe | 2006-02-17 |
| ➤ Subscribe | Tablets | 50 mg/12.5 mg and 100 mg/25 mg | ➤ Subscribe | 2004-05-24 |
| ➤ Subscribe | Tablets | 10 mg/10 mg, 10 mg/20 mg, 10 mg/40 mg and 10 mg/80 mg | ➤ Subscribe | 2009-07-27 |
| ➤ Subscribe | Nasal Spray | 50 mcg/ Spray | ➤ Subscribe | 2009-08-07 |
| ➤ Subscribe | Tablets | 5 mg and 10 mg | ➤ Subscribe | 2004-09-02 |
| ➤ Subscribe | Vaginal Ring | 0.015 mg/24 hour and 0.12 mg/24 hour | ➤ Subscribe | 2013-06-17 |
| ➤ Subscribe | Topical Solution (Lotion) | 0.10% | ➤ Subscribe | 2004-06-10 |
| ➤ Subscribe | Chewable Tablets | 4 mg and 5 mg | ➤ Subscribe | 2006-12-26 |
International Patents for ORGANON Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Hong Kong | 1115073 | ⤷ Try a Trial |
| Taiwan | 200840563 | ⤷ Try a Trial |
| Brazil | 0212907 | ⤷ Try a Trial |
| European Patent Office | 1841492 | ⤷ Try a Trial |
| Mexico | PA03006724 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for ORGANON Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0480717 | 98C0022 | France | ⤷ Try a Trial | PRODUCT NAME: MONTELUKAST SODIUM; REGISTRATION NO/DATE IN FRANCE: NL 23 133 DU 19980320; REGISTRATION NO/DATE AT EEC: 13 651 DU 19970825 |
| 1304992 | C300617 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE VAN CLINDAMYCINE, DESGEWENST IN DE VORM VAN CLINDAMYCINEFOSFAAT EN TRETINOINE; NAT. REGISTRATION NO/DATE: RVG 109745 20130626; FIRST REGISTRATION: PA1332/043/001 20130322 |
| 0720599 | 03C0028 | France | ⤷ Try a Trial | PRODUCT NAME: EZETIMIBE; NAT. REGISTRATION NO/DATE: NL28237 20030611; FIRST REGISTRATION: DE - 54486.00.00 A 54489.00.00 20021017 |
| 1110543 | SPC/GB08/005 | United Kingdom | ⤷ Try a Trial | SUPPLEMENTARY PROTECTION CERTIFICATE NO SPC/GB08/005 GRANTED TO MERCK SHARP + DOHME CORP. IN RESPECT OF THE PRODUCT DESLORATADINE OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, IN COMBINATION WITH PSEUDOEPHEDRINE OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT OR ESTER, THE GRANT OF WHICH WAS ADVERTISED IN JOURNAL NO 6322 DATED 21 JULY 2010 HAS HAD ITS MAXIMUM PERIOD OF DURATION CORRECTED, SUBJECT TO THE PAYMENT OF THE PRESCRIBED FEES IT WILL EXPIRE ON 31 JULY 2022. |
| 2435025 | PA2019014 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: GLIKOPIROLATO (ISKAITANT BET KOKIAS JO FARMACINIU POZIURIU PRIIMTINAS DRUSKAS, ESTERIUS, ENANTIOMERUS AR KITUS DARINIUS) IR FORMOTEROLIO (ISKAITANT BET KOKIAS JO FARMACINIU POZIURIU PRIIMTINAS DRUSKAS, ESTERIUS, ENANTIOMERUS AR KITUS DARINIUS) DERINYS; REGISTRATION NO/DATE: EU/1/18/1339 20181218 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.