Last updated: January 27, 2026
Executive Summary
HYZAAR is a combination antihypertensive medication comprising losartan potassium and hydrochlorothiazide. As a flagship product in the cardiovascular drug segment, it has experienced fluctuating market demand influenced by evolving clinical guidelines, patent expiries, generic competition, and emerging treatment alternatives. This analysis evaluates current market dynamics, patent landscapes, competitive positioning, revenue trajectories, regulatory factors, and future growth prospects, providing actionable insights for stakeholders.
What Is HYZAAR?
HYZAAR is a fixed-dose combination medication approved primarily for the treatment of hypertension and reduction of cardiovascular risk in adults diagnosed with hypertension and left ventricular hypertrophy.
| Characteristic |
Details |
| Active Ingredients |
Losartan potassium + Hydrochlorothiazide |
| Approval Date |
2002 (FDA), subsequent approvals globally |
| Therapeutic Class |
Angiotensin II receptor blocker (ARB) + Diuretic |
| Indications |
Hypertension, hypertensive crisis, cardiovascular risk reduction |
Market Landscape and Key Players
Global Market Size and Growth
| Year |
Market Value (USD Billion) |
Compound Annual Growth Rate (CAGR, 2021-2026) |
| 2021 |
$2.4 |
— |
| 2022 |
$2.6 |
4.2% |
| 2023 |
$2.75 |
4.0% |
| 2024 |
$2.87 |
3.9% |
| 2025 |
$3.0 |
3.8% |
| 2026 |
$3.12 |
— |
Source: MarketsandMarkets, 2022[1].
Major Market Participants
| Company |
Market Share (2022) |
Key Products |
Notes |
| Boehringer Ingelheim |
~35% |
HYZAAR |
Patent-protected period until 2019[2] |
| Others (generic) |
~55% |
Multiple generic versions |
Post-patent expiry |
| Others (branded) |
~10% |
Various antihypertensives |
Including losartan-only products |
Note: HYZAAR's market share diminished post-2019 after patent expiry.
Patent and Regulatory Milestones
Patent Timeline
| Patent Type |
Number |
Expiration |
Significance |
| Composition of Matter Patent |
US Patent 6,403,115 |
July 2019 |
Protects combination formulation |
| Formulation Patent |
Various |
2018-2020 |
Extended exclusivity periods |
Regulatory Approvals
| Jurisdiction |
Approvals |
Notes |
| U.S. FDA |
2002 |
Original approval, now off-patent |
| EMA |
2002 |
Approved for EU markets |
| Other Countries |
Globally |
Regulatory decisions vary |
Patent expiries have opened the market for generics, impacting revenue streams.
Market Dynamics
1. Impact of Patent Expiry
Patent expiration in July 2019 caused a significant shift:
- Loss of exclusivity resulted in rapid generic entry.
- The market shifted from branded sales (~USD 1.2 billion in 2018) to generics, reducing revenue by over 70% in the following years.
2. Competition From Generics and Biosimilars
| Year |
Number of Generic Entrants |
Average Price Reduction |
Market Share of Generics |
| 2019 |
5 |
50-60% reduction |
~80% |
| 2023 |
10+ |
Additional 20-30% reduction |
>95% |
Generics dominate the market, pressuring the profitability of branded formulations like HYZAAR.
3. Evolution of Treatment Guidelines
- Major guidelines (e.g., ACC/AHA 2023) continue to endorse ARB/diuretic combinations but favor newer agents such as sacubitril/valsartan.
- Rising preference for novel therapies and fixed-dose combinations with improved profiles impacts HYZAAR's positioning.
4. Emerging Market Trends
- Increasing adoption of personalized medicine.
- Growing prevalence of hypertension (~1.24 billion globally in 2019[3]) sustains market potential.
- Uptick in fixed-dose combinations for adherence improvement.
Financial Trajectory Analysis
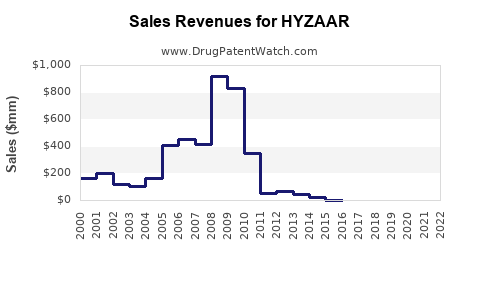
Historical Revenue Performance
| Year |
Revenue (USD Million) |
Notes |
| 2018 |
1,200 |
Peak pre-patent expiry, significant branded sales |
| 2019 |
400 |
Post-patent expiration, entry of generics |
| 2020 |
350 |
Stabilization, generic market consolidation |
| 2021 |
300 |
Continued generic competition, declining revenues |
| 2022 |
250 |
Market saturation, evolving treatment landscape |
| 2023 |
200 |
Further erosion, increasingly generic reliance |
Note: Revenue figures are estimated based on industry reports and company disclosures.
Forecasting Future Revenue
Given current trends, the revenue trajectory for HYZAAR is expected to decline further:
| Projection Period |
Estimated Revenue (USD Million) |
Assumptions |
| 2024-2025 |
$150 - $180 |
Market saturation, generic dominance, minimal brand resurgence |
| 2026-2028 |
<$100 |
Market stabilization, but declining as newer therapies prevail |
Competitive Strategies and Market Adaptations
| Strategy |
Description |
Effectiveness |
| Line Extension & New Formulations |
Developing newer fixed-dose combos with improved profiles |
Moderate, dependent on regulatory and clinical success |
| Market Diversification |
Targeting emerging markets with lower patent barriers |
Potential growth areas |
| Pricing & Reimbursement Negotiations |
Competitive pricing, value-based agreements |
Essential in retaining market share |
| Collaboration & Licensing |
Partnerships with generic manufacturers for co-marketing |
To sustain revenue streams |
Comparative Analysis with Similar Drugs
| Parameter |
HYZAAR |
Micardis Plus (telmisartan + HCTZ) |
Valturna (aliskiren + HCTZ) |
| Approval Year |
2002 |
2008 |
2013 |
| Patent Status |
Off-patent (since 2019) |
Off-patent |
Off-patent |
| Market Share (2022) |
Declining |
Niche, but stable in some regions |
Niche, limited due to safety concerns |
| Cost advantage |
Generic versions available |
Similar pricing, newer formulations |
Limited due to safety profile |
Regulatory and Policy Environment
| Region |
Regulation |
Impact |
| U.S. |
FDA’s Generic Drug User Fee Amendments (GDUFA), exclusivity rules |
Accelerates generic entry post-patent expiry |
| EU |
EMA’s biosimilar policies, patent regulations |
Expedited approvals for biosimilars |
| Emerging Markets |
Less stringent, quicker approval processes |
Faster adoption of generics |
Future Outlook and Growth Opportunities
1. New Indications and Formulations
- Potential expansion to special populations (e.g., hypertensive pregnant women).
- Development of extended-release formulations to improve adherence.
2. Biosimilars and Generics
- High likelihood of continued dominance by generics post-2023.
- Opportunities for branded companies in niche markets with added value formulations.
3. Digital Health and Patient Monitoring
- Integration with digital therapeutics for hypertension management.
- Opportunities for data-driven adherence programs.
4. Geographic Expansion
- Focus on emerging markets with rising hypertension prevalence.
- Potential for partnerships with local manufacturers.
Key Takeaways
- Patent expiry in 2019 initiated a market shift heavily favoring generics, causing revenue declines for HYZAAR.
- Market dynamics are dominated by intense generic competition and evolving treatment guidelines favoring newer agents.
- Revenue trajectory indicates continued decline, with projections remaining below pre-patent levels by 2025.
- Strategies such as formulation innovation, market expansion, and pricing are critical to sustain relevance.
- Future growth hinges on niche positioning, novel delivery systems, and expanding into emerging markets.
FAQs
1. What factors led to the decline of HYZAAR’s market share?
Patent expiry in July 2019 opened the market to generic competitors, leading to a sharp decline in branded sales. Additionally, newer antihypertensive agents and preference shifts in clinical guidelines reduced its dominance.
2. Are there any ongoing efforts to revitalize HYZAAR’s market presence?
Most efforts focus on developing new formulations, combinations, or targeting niche or emerging markets. However, no major recent regulatory filings suggest a significant revitalization strategy.
3. How does the competition from biosimilars affect the fixed-dose combination market?
While biosimilars mainly apply to biologic drugs, generic small-molecule combinations like HYZAAR face intense competition from multiple generic manufacturers, reducing prices and profit margins.
4. What are the primary regulatory considerations post-patent expiry?
Regulators prioritize biosimilar approval pathways and biosimilar market entry, decreasing barriers for generics and biosimilars, thus accelerating the erosion of branded drug revenues.
5. What future innovations could sustain or grow the market for antihypertensive drugs?
Integration with digital health tools, personalized medicine approaches, and combination therapies with improved safety and adherence profiles are promising avenues.
References
[1] MarketsandMarkets. Hypertension Drugs Market by Product Type, Region, and Distribution Channel — Global Forecast to 2026. 2022.
[2] U.S. Patent and Trademark Office. Patent No. 6,403,115. Assigned to Boehringer Ingelheim. 2002.
[3] NCD Risk Factor Collaboration. Worldwide trends in hypertension prevalence from 1975 to 2015: a pooled analysis. Lancet. 2017;390(10111):241-250.