Novadaq Tech Company Profile
✉ Email this page to a colleague
What is the competitive landscape for NOVADAQ TECH
NOVADAQ TECH has one approved drug.
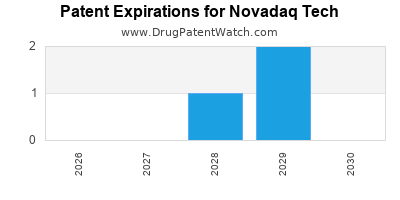
There are five US patents protecting NOVADAQ TECH drugs.
There are fifty-six patent family members on NOVADAQ TECH drugs in eleven countries.
Drugs and US Patents for Novadaq Tech
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novadaq Tech | SPY AGENT GREEN KIT | indocyanine green | POWDER;INTRAVENOUS, INTERSTITIAL | 211580-001 | Nov 21, 2018 | RX | Yes | Yes | 10,631,746 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Novadaq Tech | SPY AGENT GREEN KIT | indocyanine green | POWDER;INTRAVENOUS, INTERSTITIAL | 211580-001 | Nov 21, 2018 | RX | Yes | Yes | 8,647,605 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Novadaq Tech | SPY AGENT GREEN KIT | indocyanine green | POWDER;INTRAVENOUS, INTERSTITIAL | 211580-001 | Nov 21, 2018 | RX | Yes | Yes | 8,185,176 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Novadaq Tech | SPY AGENT GREEN KIT | indocyanine green | POWDER;INTRAVENOUS, INTERSTITIAL | 211580-001 | Nov 21, 2018 | RX | Yes | Yes | 11,712,320 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Novadaq Tech | SPY AGENT GREEN KIT | indocyanine green | POWDER;INTRAVENOUS, INTERSTITIAL | 211580-001 | Nov 21, 2018 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | |||||
| Novadaq Tech | SPY AGENT GREEN KIT | indocyanine green | POWDER;INTRAVENOUS, INTERSTITIAL | 211580-001 | Nov 21, 2018 | RX | Yes | Yes | 8,406,860 | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Novadaq Tech
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Novadaq Tech | SPY AGENT GREEN KIT | indocyanine green | POWDER;INTRAVENOUS, INTERSTITIAL | 211580-001 | Nov 21, 2018 | 8,892,190 | ⤷ Start Trial |
| Novadaq Tech | SPY AGENT GREEN KIT | indocyanine green | POWDER;INTRAVENOUS, INTERSTITIAL | 211580-001 | Nov 21, 2018 | 6,915,154 | ⤷ Start Trial |
| Novadaq Tech | SPY AGENT GREEN KIT | indocyanine green | POWDER;INTRAVENOUS, INTERSTITIAL | 211580-001 | Nov 21, 2018 | 9,421,280 | ⤷ Start Trial |
| Novadaq Tech | SPY AGENT GREEN KIT | indocyanine green | POWDER;INTRAVENOUS, INTERSTITIAL | 211580-001 | Nov 21, 2018 | 7,881,777 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Novadaq Tech Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 6577877 | ⤷ Start Trial |
| Japan | 6487544 | ⤷ Start Trial |
| European Patent Office | 4721775 | ⤷ Start Trial |
| South Korea | 101621107 | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2012038824 | ⤷ Start Trial |
| Japan | 5476317 | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2018175583 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Similar Applicant Names
Here is a list of applicants with similar names.