HELSINN HLTHCARE Company Profile
✉ Email this page to a colleague
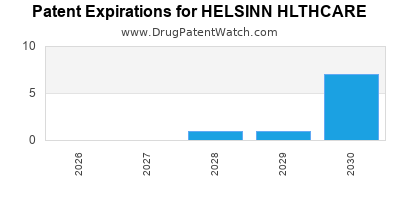
What is the competitive landscape for HELSINN HLTHCARE, and when can generic versions of HELSINN HLTHCARE drugs launch?
HELSINN HLTHCARE has five approved drugs.
There are thirty-seven US patents protecting HELSINN HLTHCARE drugs.
There are three hundred and thirty-seven patent family members on HELSINN HLTHCARE drugs in fifty-eight countries and twenty-three supplementary protection certificates in fourteen countries.
Summary for HELSINN HLTHCARE
| International Patents: | 337 |
| US Patents: | 37 |
| Tradenames: | 3 |
| Ingredients: | 4 |
| NDAs: | 5 |
Drugs and US Patents for HELSINN HLTHCARE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Helsinn Hlthcare | AKYNZEO | fosnetupitant chloride hydrochloride; palonosetron hydrochloride | SOLUTION;INTRAVENOUS | 210493-002 | May 27, 2020 | RX | Yes | Yes | 11,312,698 | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Helsinn Hlthcare | ALOXI | palonosetron hydrochloride | CAPSULE;ORAL | 022233-001 | Aug 22, 2008 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | |||||
| Helsinn Hlthcare | AKYNZEO | netupitant; palonosetron hydrochloride | CAPSULE;ORAL | 205718-001 | Oct 10, 2014 | RX | Yes | Yes | 10,676,440 | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Helsinn Hlthcare | AKYNZEO | netupitant; palonosetron hydrochloride | CAPSULE;ORAL | 205718-001 | Oct 10, 2014 | RX | Yes | Yes | 10,828,297 | ⤷ Sign Up | ⤷ Sign Up | ||||
| Helsinn Hlthcare | TRUSELTIQ | infigratinib phosphate | CAPSULE;ORAL | 214622-001 | May 28, 2021 | DISCN | Yes | No | 8,552,002 | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Helsinn Hlthcare | TRUSELTIQ | infigratinib phosphate | CAPSULE;ORAL | 214622-001 | May 28, 2021 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for HELSINN HLTHCARE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Helsinn Hlthcare | ALOXI | palonosetron hydrochloride | CAPSULE;ORAL | 022233-001 | Aug 22, 2008 | 5,202,333 | ⤷ Sign Up |
| Helsinn Hlthcare | AKYNZEO | netupitant; palonosetron hydrochloride | CAPSULE;ORAL | 205718-001 | Oct 10, 2014 | 5,202,333 | ⤷ Sign Up |
| Helsinn Hlthcare | ALOXI | palonosetron hydrochloride | INJECTABLE;INTRAVENOUS | 021372-002 | Feb 29, 2008 | 5,202,333*PED | ⤷ Sign Up |
| Helsinn Hlthcare | AKYNZEO | netupitant; palonosetron hydrochloride | CAPSULE;ORAL | 205718-001 | Oct 10, 2014 | 6,297,375 | ⤷ Sign Up |
| Helsinn Hlthcare | ALOXI | palonosetron hydrochloride | INJECTABLE;INTRAVENOUS | 021372-001 | Jul 25, 2003 | 5,202,333*PED | ⤷ Sign Up |
| Helsinn Hlthcare | ALOXI | palonosetron hydrochloride | INJECTABLE;INTRAVENOUS | 021372-002 | Feb 29, 2008 | 9,125,905 | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for HELSINN HLTHCARE Drugs
Supplementary Protection Certificates for HELSINN HLTHCARE Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2785706 | LUC00158 | Luxembourg | ⤷ Sign Up | PRODUCT NAME: AKYNZEO - FOSNETUPITANT/PALONOSETRON; AUTHORISATION NUMBER AND DATE: EU/1/15/1001 20200318 |
| 2785706 | 301047 | Netherlands | ⤷ Sign Up | PRODUCT NAME: FOSNETUPITANT, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER FOSNETUPITANTCHLORIDEHYDROCHLORIDE; REGISTRATION NO/DATE: EU/1/15/1001 20200318 |
| 1035115 | 1590040-0 | Sweden | ⤷ Sign Up | PRODUCT NAME: NETUPITANT; REG. NO/DATE: EU/1/15/1001 20150529 |
| 2785706 | 122020000046 | Germany | ⤷ Sign Up | PRODUCT NAME: FOSNETUPITANT MIT PALONOSETRON; REGISTRATION NO/DATE: C(2020)1804(FINAL) 20200316 |
| 2785706 | C20200029 00371 | Estonia | ⤷ Sign Up | PRODUCT NAME: FOSNETUPITANT;REG NO/DATE: EU/1/15/1001; 18.03.2020 |
| 2785706 | 132020000000094 | Italy | ⤷ Sign Up | PRODUCT NAME: FOSNETUPITANT O UN SUO SALE FARMACEUTICAMENTE ACCETTABILE, IN PARTICOLARE IL SUO SALE CLORURO CLORIDRATO(AKYNZEO); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/15/1001/001-002, 20200316 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.