Mark Cuban’s Cost-Plus Pricing Is Breaking Pharma’s Patent Playbook
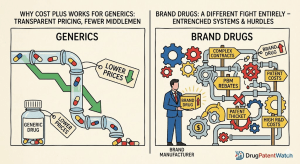
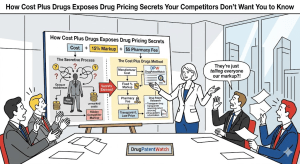
The pharmaceutical industry’s patent defense system is a machine built on a single assumption: that the price of a drug […]
Mark Cuban’s Cost-Plus Pricing Is Breaking Pharma’s Patent Playbook Read Post »