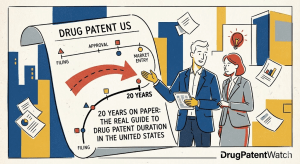
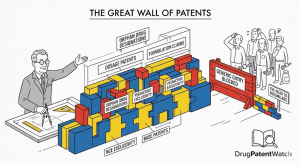
20 Years on Paper: The Real Guide to Drug Patent Duration in the United States
The Patent Clock Nobody Shows You The standard answer to “how long does a patent last?” is 20 years. You […]
20 Years on Paper: The Real Guide to Drug Patent Duration in the United States Read Post »