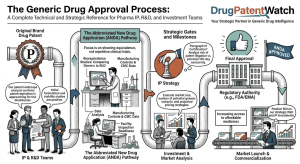
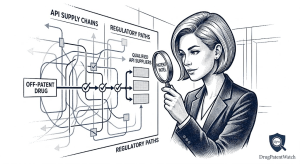
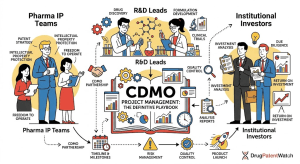
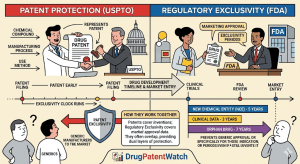
The Generic Drug Approval Process: A Complete Technical and Strategic Reference for Pharma IP, R&D, and Investment Teams
A deep dive covering the ANDA pathway, EU MAA procedures, bioequivalence science, Paragraph IV litigation strategy, patent cliff analysis, evergreening […]