kalydeco Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Kalydeco, and what generic alternatives are available?
Kalydeco is a drug marketed by Vertex Pharms Inc and Vertex Pharms and is included in two NDAs. There are fourteen patents protecting this drug and two Paragraph IV challenges.
This drug has two hundred and fifty-seven patent family members in thirty-six countries.
The generic ingredient in KALYDECO is ivacaftor. There are three drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the ivacaftor profile page.
DrugPatentWatch® Generic Entry Outlook for Kalydeco
Kalydeco was eligible for patent challenges on January 31, 2016.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be February 13, 2030. This may change due to patent challenges or generic licensing.
There have been five patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There are two tentative approvals for the generic drug (ivacaftor), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for kalydeco?
- What are the global sales for kalydeco?
- What is Average Wholesale Price for kalydeco?
Summary for kalydeco
| International Patents: | 257 |
| US Patents: | 14 |
| Applicants: | 2 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 94 |
| Clinical Trials: | 26 |
| Patent Applications: | 1,513 |
| Drug Prices: | Drug price information for kalydeco |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for kalydeco |
| What excipients (inactive ingredients) are in kalydeco? | kalydeco excipients list |
| DailyMed Link: | kalydeco at DailyMed |
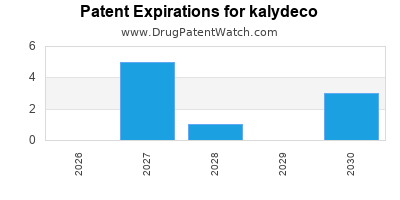

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for kalydeco
Generic Entry Dates for kalydeco*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
GRANULE;ORAL |
Generic Entry Dates for kalydeco*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for kalydeco
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) | Phase 2 |
| University of Miami | Early Phase 1 |
| University of Kansas Medical Center | Early Phase 1 |
Pharmacology for kalydeco
US Patents and Regulatory Information for kalydeco
kalydeco is protected by fourteen US patents and ten FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of kalydeco is ⤷ Start Trial.
This potential generic entry date is based on patent 10,646,481.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vertex Pharms Inc | KALYDECO | ivacaftor | GRANULE;ORAL | 207925-001 | Mar 17, 2015 | RX | Yes | No | 12,214,083*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Vertex Pharms | KALYDECO | ivacaftor | TABLET;ORAL | 203188-001 | Jan 31, 2012 | RX | Yes | Yes | 7,495,103*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Vertex Pharms Inc | KALYDECO | ivacaftor | GRANULE;ORAL | 207925-003 | Apr 29, 2019 | RX | Yes | No | 7,495,103*PED | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for kalydeco
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Vertex Pharms Inc | KALYDECO | ivacaftor | GRANULE;ORAL | 207925-004 | May 3, 2023 | 8,629,162 | ⤷ Start Trial |
| Vertex Pharms Inc | KALYDECO | ivacaftor | GRANULE;ORAL | 207925-003 | Apr 29, 2019 | 8,629,162 | ⤷ Start Trial |
| Vertex Pharms Inc | KALYDECO | ivacaftor | GRANULE;ORAL | 207925-005 | May 3, 2023 | 8,629,162 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for kalydeco
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Vertex Pharmaceuticals (Ireland) Limited | Kalydeco | ivacaftor | EMEA/H/C/002494Kalydeco tablets are indicated:As monotherapy for the treatment of adults, adolescents, and children aged 6 years and older and weighing 25 kg or more with cystic fibrosis (CF) who have an R117H CFTR mutation or one of the following gating (class III) mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene: G551D, G1244E, G1349D, G178R, G551S, S1251N, S1255P, S549N or S549R (see sections 4.4 and 5.1).In a combination regimen with tezacaftor/ivacaftor tablets for the treatment of adults, adolescents, and children aged 6 years and older with cystic fibrosis (CF) who are homozygous for the F508del mutation or who are heterozygous for the F508del mutation and have one of the following mutations in the CFTR gene: P67L, R117C, L206W, R352Q, A455E, D579G, 711+3A→G, S945L, S977F, R1070W, D1152H, 2789+5G→A, 3272 26A→G, and 3849+10kbC→T.In a combination regimen with ivacaftor/tezacaftor/elexacaftor tablets for the treatment of adults, adolescents, and children aged 6 years and older with cystic fibrosis (CF) who have at least one F508del mutation in the CFTR gene (see section 5.1).Kalydeco granules are indicated for the treatment of infants aged at least 4 months, toddlers and children weighing 5 kg to less than 25 kg with cystic fibrosis (CF) who have an R117H CFTR mutation or one of the following gating (class III) mutations in the CFTR gene: G551D, G1244E, G1349D, G178R, G551S, S1251N, S1255P, S549N or S549R (see sections 4.4 and 5.1). | Authorised | no | no | no | 2012-07-23 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for kalydeco
When does loss-of-exclusivity occur for kalydeco?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 09282419
Estimated Expiration: ⤷ Start Trial
Patent: 10282986
Estimated Expiration: ⤷ Start Trial
Patent: 13226076
Estimated Expiration: ⤷ Start Trial
Patent: 16216569
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0916877
Estimated Expiration: ⤷ Start Trial
Patent: 2012008082
Estimated Expiration: ⤷ Start Trial
Patent: 2014021090
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 33908
Estimated Expiration: ⤷ Start Trial
Patent: 69695
Estimated Expiration: ⤷ Start Trial
Patent: 65519
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 12000348
Estimated Expiration: ⤷ Start Trial
China
Patent: 2231990
Estimated Expiration: ⤷ Start Trial
Patent: 2497859
Estimated Expiration: ⤷ Start Trial
Patent: 4470518
Patent: Pharmaceutical composition and administration thereof
Estimated Expiration: ⤷ Start Trial
Patent: 9966264
Patent: 药物组合物及其施用 (Pharmaceutical composition and administration thereof)
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0180328
Estimated Expiration: ⤷ Start Trial
Patent: 0190660
Estimated Expiration: ⤷ Start Trial
Patent: 0210208
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 19945
Estimated Expiration: ⤷ Start Trial
Patent: 21572
Estimated Expiration: ⤷ Start Trial
Patent: 23901
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 28618
Estimated Expiration: ⤷ Start Trial
Patent: 64337
Estimated Expiration: ⤷ Start Trial
Patent: 45625
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 1706
Estimated Expiration: ⤷ Start Trial
Patent: 1170330
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 28618
Estimated Expiration: ⤷ Start Trial
Patent: 64337
Estimated Expiration: ⤷ Start Trial
Patent: 19670
Patent: COMPOSITION PHARMACEUTIQUE ET SON ADMINISTRATION (PHARMACEUTICAL COMPOSITION AND ADMINISTRATION THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 45625
Estimated Expiration: ⤷ Start Trial
Patent: 42037
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 64337
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 61140
Estimated Expiration: ⤷ Start Trial
Patent: 03840
Patent: 藥物組合物及其施用 (PHARMACEUTICAL COMPOSITION AND ADMINISTRATION THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 05690
Patent: 藥物组合物及其施用 (PHARMACEUTICAL COMPOSITION AND ADMINISTRATION THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 56805
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 35931
Estimated Expiration: ⤷ Start Trial
Patent: 42437
Estimated Expiration: ⤷ Start Trial
Patent: 53357
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 1203
Estimated Expiration: ⤷ Start Trial
Patent: 4307
Patent: תכשירי רוקחות המכילים פיזור מוצק של n-[2,4-ביס(1,1-דימטילאטיל)-5-הידרוקסיפניל]-1,4-דיהידרו-4-אוקסוקוינולין-3-קארבוקסאמיד ושימושים בהם (Pharmaceutical compositions containing a solid dispersion of n-[2,4-bis(1,1-dimethylethyl)-5-hydroxyphenyl]-1,4-dihydro-4-oxoquinoline-3-carboxamode and uses thereof)
Estimated Expiration: ⤷ Start Trial
Patent: 2421
Estimated Expiration: ⤷ Start Trial
Patent: 5430
Patent: תכשירי רוקחות המכילים פיזור מוצק של n-[2,4-ביס(1,1-דימטילאטיל)-5-הידרוקסיפניל]-1,4-דיהידרו-4-אוקסוקוינולין-3-קארבוקסאמיד ושימושים בהם (Pharmaceutical compositions containing a solid dispersion of n-[2,4-bis(1,1-dimethylethyl)-5-hydroxyphenyl]-1,4-dihydro-4-oxoquinoline-3-carboxamode and uses thereof)
Estimated Expiration: ⤷ Start Trial
Patent: 5854
Estimated Expiration: ⤷ Start Trial
Patent: 1180
Patent: תכשירי רוקחות המכילים פיזור מוצק של n-[2,4-ביס(1,1-דימטילאטיל)-5-הידרוקסיפניל]-1,4-דיהידרו-4-אוקסוקוינולין-3-קארבוקסאמיד ושימושים בהם (Pharmaceutical compositions containing a solid dispersion of n-[2,4-bis(1,1-dimethylethyl)-5-hydroxyphenyl]-1,4-dihydro-4-oxoquinoline-3-carboxamode and uses thereof)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 75768
Estimated Expiration: ⤷ Start Trial
Patent: 34041
Estimated Expiration: ⤷ Start Trial
Patent: 11530598
Estimated Expiration: ⤷ Start Trial
Patent: 13501787
Estimated Expiration: ⤷ Start Trial
Patent: 14111656
Estimated Expiration: ⤷ Start Trial
Patent: 15511583
Patent: 薬学的組成物およびその投与
Estimated Expiration: ⤷ Start Trial
Patent: 17190356
Patent: 薬学的組成物およびその投与 (PHARMACEUTICAL COMPOSITION AND ADMINISTRATION THEREOF)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 28618
Estimated Expiration: ⤷ Start Trial
Patent: 64337
Estimated Expiration: ⤷ Start Trial
Patent: 45625
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 6161
Estimated Expiration: ⤷ Start Trial
Patent: 3230
Estimated Expiration: ⤷ Start Trial
Patent: 9751
Patent: COMPOSICIÓN FARMACÉUTICA Y ADMINISTRACIONES DE LA MISMA. (PHARMACEUTICAL COMPOSITION AND ADMINISTRATION THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 11001782
Estimated Expiration: ⤷ Start Trial
Patent: 12001939
Estimated Expiration: ⤷ Start Trial
Patent: 14010253
Patent: COMPOSICION FARMACEUTICA Y ADMINISTRACIONES DE LA MISMA. (PHARMACEUTICAL COMPOSITION AND ADMINISTRATION THEREOF.)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 019
Estimated Expiration: ⤷ Start Trial
Patent: 356
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 1535
Estimated Expiration: ⤷ Start Trial
Patent: 7823
Estimated Expiration: ⤷ Start Trial
Patent: 9199
Patent: Pharmaceutical compositions comprising a solid dispersion of n-[2,4-bis(1,1-dimethylethyl)-5-hydroxyphenyl]-1,4-dihydro-4-oxoquinoline-3-carboxamide
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 28618
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 28618
Estimated Expiration: ⤷ Start Trial
Patent: 64337
Estimated Expiration: ⤷ Start Trial
Patent: 45625
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 28618
Estimated Expiration: ⤷ Start Trial
Patent: 64337
Estimated Expiration: ⤷ Start Trial
Patent: 45625
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 92779
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ И ЕЕ ВВЕДЕНИЯ (PHARMACEUTICAL COMPOSITION AND INTRODUCTION THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 12109390
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ И СПОСОБЫ ЕЕ ВВЕДЕНИЯ
Estimated Expiration: ⤷ Start Trial
Patent: 14139006
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ И ЕЕ ВВЕДЕНИЯ
Estimated Expiration: ⤷ Start Trial
Patent: 19116577
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ И ЕЕ ВВЕДЕНИЯ
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01800074
Estimated Expiration: ⤷ Start Trial
Patent: 01900210
Estimated Expiration: ⤷ Start Trial
Patent: 02100077
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 894
Patent: FARMACEUTSKA KOMPOZICIJA N-[2,4-BIS(1,1-DIMETILETIL)-5-HIDROKSIFENIL]-1,4-DIHIDRO-4-OKSOHINOLIN-3-KARBOKSAMIDA I NJENO ORDINIRANJE (PHARMACEUTICAL COMPOSITION OF N-[2,4-BIS(1,1-DIMETHYLETHYL)-5-HYDROXYPHENYL]-1,4-DIHYDRO-4- OXOQUINOLINE-3-CARBOXAMIDE AND ADMINISTRATION THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 604
Patent: FORMULACIJA N-[2,4-BIS(1,1-DIMETILETIL)-5-HIDROKSIFENIL]-1,4-DIHIDRO-4-OKSOHINOLIN-3-KARBOKSAMIDA U OBLIKU TABLETE ZA UPOTREBU U TRETMANU CISTIČNE FIBROZE (TABLET FORMULATION OF N-[2,4-BIS(1,1-DIMETHYLETHYL)-5-HYDROXYPHENYL]-1,4-DIHYDRO-4-OXOQUINOLINE-3-CARBOXAMIDE FOR USE IN THE TREATMENT OF CYSTIC FIBROSIS)
Estimated Expiration: ⤷ Start Trial
Patent: 408
Patent: FARMACEUTSKA KOMPOZICIJA I NJENO ORDINIRANJE (PHARMACEUTICAL COMPOSITION AND ADMINISTRATIONS THEREOF)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 8337
Patent: PHARMACEUTICAL COMPOSITION AND ADMINISTRATIONS THEREOF
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 28618
Estimated Expiration: ⤷ Start Trial
Patent: 64337
Estimated Expiration: ⤷ Start Trial
Patent: 45625
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1101097
Patent: PHARMACEUTICAL COMPOSITION OF N-[2,4-BIS (1,1-DIMETHYLETHYL)-5-HYDROXYPHENYL]-1,4-DIHYDRO-4-OXOQUINOLINE-3-CARBOXAMIDE AND ADMINISTRATION THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 1200722
Patent: PHARMACEUTICAL COMPOSITION AND ADMINISTRATIONS THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 1406233
Patent: PHARMACEUTICAL COMPOSITION AND ADMINISTRATION THEREOF
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 110042356
Estimated Expiration: ⤷ Start Trial
Patent: 120061875
Estimated Expiration: ⤷ Start Trial
Patent: 170072950
Estimated Expiration: ⤷ Start Trial
Patent: 190143497
Estimated Expiration: ⤷ Start Trial
Patent: 220057663
Estimated Expiration: ⤷ Start Trial
Patent: 240066199
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 60143
Estimated Expiration: ⤷ Start Trial
Patent: 18273
Estimated Expiration: ⤷ Start Trial
Patent: 57152
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 2261
Patent: ФАРМАЦЕВТИЧНА КОМПОЗИЦІЯ НА ОСНОВІ ТВЕРДОЇ ДИСПЕРСІЇ N-[2,4-БІС(1,1-ДИМЕТИЛЕТИЛ)-5-ГІДРОКСИФЕНІЛ]-1,4-ДИГІДРО-4-ОКСОХІНОЛІН-3-КАРБОКСАМІДУ[ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИЯ НА ОСНОВЕ ТВЕРДОЙ ДИСПЕРСИИ N-[2,4-БИС(1,1-ДИМЕТИЛЭТИЛ)-5-ГИДРОКСИФЕНИЛ]-1,4-ДИГИДРО-4-ОКСОХИНОЛИН-3-КАРБОКСАМИДА (PHARMACEUTICAL COMPOSITIONS COMPRISING A SOLID DISPERSION OF N-[2,4-BIS(1,1-DIMETHYLETHYL)-5-HYDROXYPHENYL]-1,4-DIHYDRO-4-OXOQUINOLINE-3-CARBOXAMIDE)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering kalydeco around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Hungary | E032540 | ⤷ Start Trial | |
| Mexico | 2012001939 | ⤷ Start Trial | |
| Cyprus | 1118980 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for kalydeco
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3170818 | 20C1037 | France | ⤷ Start Trial | PRODUCT NAME: LUMACAFTOR ET IVACAFTOR DANS TOUTES SES FORMES RELEVANT DE LA PROTECTION DU BREVET DE BASE; REGISTRATION NO/DATE: EU/1/15/1059 20151124 |
| 1773816 | C01773816/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: IVACAFTOR; REGISTRATION NO/DATE: SWISSMEDIC 62686 13.01.2014 |
| 1773816 | 2015/036 | Ireland | ⤷ Start Trial | PRODUCT NAME: N-(5-HYDROXYL-2,4-DITERT-BUTYL-PHENYL)-4-OXO-1H-QUINOLINE-3- CARBOXAMIDE (IVACAFTOR) OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTRATION NO/DATE: EU/1/12/782/001-002 20120723 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Kalydeco: Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
