Last updated: February 19, 2026
Summary: Zofran (ondansetron hydrochloride), developed by GlaxoSmithKline (GSK), is a 5-HT3 receptor antagonist primarily used to prevent nausea and vomiting caused by cancer chemotherapy, radiation therapy, and surgery. Since its initial approval, Zofran has faced patent expirations, leading to significant generic competition. This has shifted its market position from a blockbuster innovator drug to a mature, cost-competitive product. Financial performance is now largely driven by market share within the generics sector and indications for its use.
What is Zofran and its Mechanism of Action?
Zofran is an antiemetic medication. Its active pharmaceutical ingredient is ondansetron hydrochloride. It functions by selectively blocking the action of serotonin (5-HT3 receptors) in the brain and the gastrointestinal tract. Serotonin is a neurotransmitter released by damaged cells in the gut during chemotherapy or radiation, which can trigger the vomiting reflex. By inhibiting these receptors, ondansetron prevents the signaling that leads to nausea and vomiting.
When Was Zofran First Approved and By Which Regulatory Bodies?
The U.S. Food and Drug Administration (FDA) approved Zofran (ondansetron) for oral use on December 22, 1993. It was initially approved for the prevention of nausea and vomiting associated with initial and re-treatment cycles of emetogenic cancer therapy. The FDA approved Zofran for intravenous use in 1996.
The European Medicines Agency (EMA) also approved ondansetron. Initial marketing authorization in the United Kingdom was granted in 1990, with broader European approvals following.
What Were Zofran's Key Indications at Launch and How Have They Evolved?
At its launch, Zofran's primary indications were:
- Prevention of nausea and vomiting associated with highly emetogenic cancer chemotherapy.
- Prevention of nausea and vomiting associated with radiation therapy.
Over time, its indications expanded:
- Prevention of postoperative nausea and vomiting (PONV). This indication became a significant driver of Zofran's market growth and remains a key use.
- Prevention of nausea and vomiting associated with moderately emetogenic cancer chemotherapy.
What Was Zofran's Peak Market Performance and Revenue?
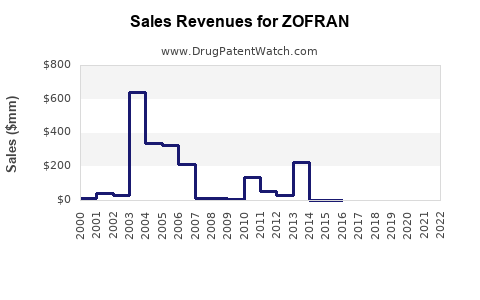
Zofran achieved blockbuster status shortly after its introduction. While specific peak annual revenue figures for the branded Zofran are subject to proprietary reporting, industry analyses indicate it was a significant contributor to GSK's sales. In 2003, for example, ondansetron-based products were reported to generate over $1 billion in annual sales for GSK, positioning it as one of the company's top-selling drugs at that time [1]. This revenue was driven by its efficacy and established safety profile in critical patient populations.
When Did Zofran's Key Patents Expire?
The primary patents protecting branded Zofran began to expire in the early to mid-2000s. For instance, a key patent for ondansetron was set to expire around 2005, opening the door for generic manufacturers. The exact expiry dates varied depending on specific formulations and geographical markets. This patent cliff marked a critical transition point for Zofran's market exclusivity.
Who Are the Major Generic Manufacturers of Ondansetron?
Following patent expirations, numerous pharmaceutical companies entered the market with generic versions of ondansetron. Key players in the generic ondansetron market include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Aurobindo Pharma Ltd.
- Apھار Pharma Limited
These companies compete on price, formulation, and distribution reach.
What is the Current Market Landscape for Ondansetron?
The market for ondansetron is now largely a genericized one. The branded Zofran product, while still available, holds a significantly smaller market share compared to its peak. The landscape is characterized by:
- Intense Price Competition: Generic ondansetron is available at a fraction of the cost of branded Zofran. This has made it a highly accessible treatment option.
- High Volume Sales: Despite lower per-unit revenue, the sheer volume of ondansetron prescribed and dispensed globally contributes to substantial overall market value.
- Fragmented Market Share: Market share among generic manufacturers is distributed, with Teva, Mylan, and Sun Pharma often ranking among the top suppliers due to their extensive portfolios and manufacturing capabilities.
- Continued Demand: The established efficacy of ondansetron for its primary indications ensures sustained demand, particularly in hospital and oncology settings.
- Formulation Diversity: Generic manufacturers offer ondansetron in various dosage forms including oral tablets, orally disintegrating tablets (ODTs), oral solution, and injectable solutions.
How Has the Shift to Generics Impacted Ondansetron's Pricing and Profitability?
The shift to generics has dramatically altered ondansetron's pricing structure.
- Pricing: Branded Zofran was priced as a premium specialty drug. Generic ondansetron prices are significantly lower, driven by competition and the general pricing pressures in the generic pharmaceutical market. For example, a 30-day supply of generic ondansetron tablets can range from under $10 to $50, depending on the specific manufacturer, dosage, and pharmacy, whereas branded Zofran would have been considerably more.
- Profitability: For GSK, the profitability of branded Zofran declined sharply after patent expiry due to the loss of market exclusivity and pricing power. For generic manufacturers, profitability is derived from achieving economies of scale in manufacturing, efficient supply chain management, and securing large contracts with healthcare systems and wholesalers. Profit margins on individual units are lower, but aggregate profits can be substantial due to high sales volumes.
What Are the Key Market Drivers for Ondansetron Today?
The primary market drivers for ondansetron include:
- Prevalence of Cancer: The ongoing and increasing incidence of cancer globally necessitates the use of antiemetics like ondansetron during chemotherapy.
- Surgical Procedures: The high volume of surgical procedures performed worldwide, coupled with the risk of postoperative nausea and vomiting, drives demand for ondansetron.
- Cost-Effectiveness: The low cost of generic ondansetron makes it a preferred choice for healthcare providers and payers, especially in budget-conscious healthcare systems.
- Established Efficacy and Safety Profile: Decades of clinical use have solidified ondansetron's reputation for reliable efficacy and a well-understood safety profile, even if off-label concerns have emerged.
- Accessibility: Broad availability through multiple generic manufacturers ensures consistent supply and accessibility.
What are the Potential Market Threats or Challenges for Ondansetron?
Despite its established position, ondansetron faces several market threats:
- Emergence of Newer Antiemetics: Development of newer classes of antiemetics, such as NK-1 receptor antagonists (e.g., aprepitant), which may offer improved efficacy in certain patient populations or reduced side effects, presents a competitive challenge. These newer agents are often used in combination with 5-HT3 antagonists like ondansetron for highly emetogenic regimens.
- Off-Label Concerns and Litigation: Zofran has been subject to litigation alleging risks of birth defects when taken during pregnancy. While the FDA has maintained that the drug is generally safe when prescribed appropriately for its approved indications, these concerns and ongoing legal challenges can create market uncertainty and impact prescribing patterns, particularly in women of childbearing age [2].
- Generic Price Erosion: The highly competitive generic market can lead to continued price erosion, putting pressure on the profitability of even high-volume generic manufacturers.
- Reimbursement Pressures: Healthcare payers continuously seek to manage costs, which can lead to formulary restrictions or preferred generic status, further intensifying competition.
- Development of Alternative Therapies: While less common for its primary indications, ongoing research into non-pharmacological approaches to managing nausea and vomiting could theoretically impact demand.
What is the Financial Trajectory and Outlook for Ondansetron?
The financial trajectory for ondansetron has transitioned from high-growth innovator sales to stable, volume-driven generic revenue.
- Past Trajectory: Dominated by rapid growth and blockbuster status for branded Zofran, followed by a sharp decline in revenue for GSK upon patent expiry.
- Current Trajectory: Characterized by sustained demand, consistent sales volumes, and moderate revenue generation for generic manufacturers. Profitability for generic players hinges on operational efficiency and market share.
- Future Outlook: The outlook for ondansetron remains stable. Its fundamental utility in managing common and critical conditions ensures ongoing demand. While significant revenue growth is unlikely, the market is expected to persist due to its cost-effectiveness and established clinical role. Competitive pressures will continue to shape profitability for generic manufacturers. The impact of any future litigation outcomes or significant advancements in alternative therapies remains a potential variable.
How Does Ondansetron Compare to Other Antiemetic Agents?
Ondansetron belongs to the 5-HT3 receptor antagonist class. It is often compared to other drugs within this class and other antiemetic classes:
| Drug Class |
Example Agents |
Mechanism |
Key Differentiators vs. Ondansetron |
| 5-HT3 Receptor Antagonists |
Ondansetron, Granisetron, Palonosetron, Dolasetron |
Blocks serotonin receptors in the GI tract and brain. |
Palonosetron has a longer half-life and demonstrated higher efficacy in preventing delayed nausea/vomiting in some studies [3]. |
| NK-1 Receptor Antagonists |
Aprepitant, Fosaprepitant, Rolapitant |
Blocks the action of substance P, a neurotransmitter involved in vomiting. |
Often used in combination with 5-HT3 antagonists for highly emetogenic chemotherapy to target different pathways [4]. |
| Corticosteroids |
Dexamethasone |
Mechanism not fully understood but contributes to antiemetic effects. |
Commonly used as an adjunct therapy in chemotherapy-induced nausea and vomiting (CINV). |
| Dopamine Antagonists |
Prochlorperazine, Metoclopramide |
Blocks dopamine receptors in the chemoreceptor trigger zone. |
May have a broader spectrum of activity but also different side effect profiles, including extrapyramidal symptoms. |
| Antihistamines/Anticholinergics |
Diphenhydramine, Scopolamine |
Block histamine and muscarinic receptors. |
Primarily used for motion sickness and mild nausea, less effective for severe chemotherapy-induced nausea. |
Ondansetron remains a foundational therapy due to its balance of efficacy, tolerability, and cost, particularly when compared to newer, more expensive agents for less severe forms of nausea and vomiting.
Key Takeaways
- Zofran (ondansetron hydrochloride) transitioned from a blockbuster branded drug to a mature, highly competitive generic market following patent expirations in the early to mid-2000s.
- Its primary market drivers remain the prevalence of cancer, the volume of surgical procedures, and its established cost-effectiveness and safety profile.
- The market is characterized by intense price competition among numerous generic manufacturers.
- While branded Zofran's revenue for GSK declined, ondansetron continues to generate significant revenue globally through high-volume generic sales.
- Emerging newer antiemetic classes and ongoing litigation related to off-label use present potential market challenges.
- The future outlook for ondansetron is stable, with sustained demand driven by its clinical utility and economic advantages.
Frequently Asked Questions
1. What is the primary revenue source for ondansetron today?
The primary revenue source for ondansetron today is from the sales of generic formulations by multiple pharmaceutical manufacturers.
2. How has the patent expiry of Zofran impacted GlaxoSmithKline's financials?
GlaxoSmithKline experienced a significant decline in revenue from its branded Zofran product following patent expiry due to the entry of generic competitors, which drastically reduced its pricing power and market exclusivity.
3. Are there any significant research and development activities ongoing for ondansetron?
Major R&D activity for ondansetron itself is limited, as it is a well-established molecule. Current research tends to focus on combination therapies or comparing its efficacy and cost-effectiveness against newer agents rather than developing novel ondansetron formulations.
4. What is the typical market share distribution among generic ondansetron manufacturers?
The market share distribution is fragmented, with major generic players like Teva, Mylan (Viatris), and Sun Pharma often holding substantial portions due to their scale and product portfolios. However, many smaller and regional players also contribute to the competitive landscape.
5. What is the most significant regulatory or legal challenge facing ondansetron currently?
Ongoing litigation alleging links between ondansetron use during pregnancy and birth defects remains a significant legal challenge, potentially impacting prescribing practices and creating market uncertainty, despite the drug's approved indications and general safety profile when used as prescribed.
Citations
[1] A. K. Singh, S. K. Singh, V. K. Singh, & P. Singh. (2012). Ondansetron: A review. Journal of Drug Delivery and Therapeutics, 2(3), 108-114.
[2] U.S. Food and Drug Administration. (2019, October 1). FDA Drug Safety Communication - Certain Zofran (ondansetron) Products. Retrieved from [FDA website] (Note: Specific URL may change, but FDA communications are searchable).
[3] Scarsbrook, A. F., Guppy, M. J., & Mulroy, P. D. (2009). A review of palonosetron in the management of chemotherapy-induced nausea and vomiting. Therapeutics and Clinical Risk Management, 5, 135–147.
[4] De Wit, R., Herrstedt, J., & Van Zuylen, L. (2013). Combination therapy with NK1 receptor antagonists and 5-HT3 receptor antagonists for chemotherapy-induced nausea and vomiting. Supportive Care in Cancer, 21(1), 299-307.