Last updated: February 23, 2026
What is VICODIN and its current market status?
VICODIN is a combination analgesic medication containing hydrocodone and acetaminophen. It was widely prescribed for moderate to severe pain management. Its use has declined due to regulatory changes and increased awareness of opioid-related risks. Post-2020, the drug faces reduced market penetration, with prescriptions decreasing annually.
How have regulatory actions impacted VICODIN's availability?
In October 2014, the Drug Enforcement Administration (DEA) rescheduled hydrocodone combination products from Schedule III to Schedule II, imposing stricter prescribing regulations. This shift aimed to curb misuse but led to a reduction in prescriptions. Many providers shifted to alternative analgesics; pharmacies faced tighter controls on dispensing.
Since then, state-level policies have further restricted opioid prescriptions. The US FDA issued clarifications emphasizing safer use, impacting physician prescribing behavior.
What is the current market size and leading players?
The US pain management market, where VICODIN was dominant, was valued at approximately $24 billion in 2022. Hydrocodone products comprised a significant share within the opioid segment, but VICODIN’s market share has declined.
Leading competitors include:
- OxyContin (oxycodone)
- Percocet (oxycodone and acetaminophen)
- Tramadol (Ultram)
Generic versions accounting for 70% of prescriptions have entered the market, reducing brand-specific revenues.
How have sales figures evolved post-regulatory changes?
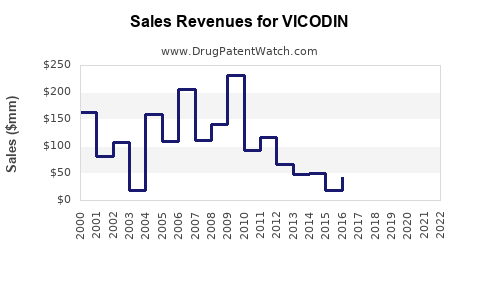
From peak sales of approximately $1.2 billion annually in the US (around 2010), VICODIN's revenue has fallen sharply. Between 2015 and 2020, sales declined by over 55%, owing to:
- Prescribing restrictions
- Physician avoidance of opioids
- Availability of alternatives with lower abuse potential
2022 retail sales are estimated below $300 million, predominantly from generic formulations.
What are the legal and ethical risks affecting future prospects?
Litigation and regulatory pressures increase risks:
- Multiple state and federal lawsuits allege opioid manufacturers contributed to the opioid epidemic.
- The Department of Justice has negotiated significant settlements, with some firms paying billions.
- Agencies continue to scrutinize promotional activities and prescribing practices.
These factors may constrain manufacturing and marketing strategies for opioids like VICODIN.
How might the future legal landscape influence VICODIN’s market?
Potential legislation could further restrict opioid prescribing or impose penalties. The CDC has issued guidelines favoring non-opioid pain management, influencing clinical practice.
Legal settlements could lead to mandatory marketing restrictions, impacting sales. Some jurisdictions may implement prescription monitoring programs that make distribution more difficult.
What are the financial implications for companies involved?
Manufacturers of VICODIN include AbbVie (through its acquisition of Abbott Laboratories assets before 2012) and generic producers.
Key financial effects:
- Revenue stream collapse for brand-name formulations
- Increased legal liabilities and settlement costs
- Shift in R&D toward non-opioid analgesics
Market contractions have led to revenue declines for primary producers, with some depleting opioid-related portfolios.
How does the trend in consumer behavior affect the market?
Patients and healthcare providers increasingly favor non-opioid options due to:
- Addiction concerns
- Regulatory restrictions
- Availability of safer medication alternatives
This trend diminishes demand for VICODIN and similar drugs.
Summary of market risks and opportunities
| Risks |
Opportunities |
| Regulatory restrictions |
Development of abuse-deterrent formulations |
| Litigation liabilities |
Expansion into non-opioid analgesic markets |
| Declining demand |
Innovation in alternative pain management therapies |
| Public perception |
Improving brand reputation via safer product profiles |
Key Takeaways
- VICODIN experienced a significant sales decline post-2014 drug scheduling and evolving prescribing guidelines.
- The US pain management market is shrinking for opioids, with generic formulations dominating.
- Legal and regulatory actions pose ongoing risks, including substantial settlement costs.
- Future growth prospects hinge on transitioning R&D toward non-opioid alternatives and safer formulations.
- Consumer preferences favor non-opioid options, impacting traditional narcotic analgesics' market share.
FAQs
1. What caused the decline in VICODIN sales?
Regulatory scheduling, increased prescribing restrictions, and a shift toward non-opioid therapies reduced demand.
2. Can VICODIN still be prescribed legally?
Yes, but with tighter controls and monitoring due to its Schedule II status, reducing prescribing flexibility.
3. Are there ongoing legal liabilities?
Yes, numerous lawsuits and settlement obligations exist, potentially impacting manufacturers financially.
4. Will VICODIN return to higher market levels?
Unlikely, given the public health focus on reducing opioid use and the availability of safer alternatives.
5. What are the future revenue prospects for opioids?
Limited, as policy and societal attitudes increasingly favor non-opioid analgesics; companies are shifting R&D accordingly.
References
[1] U.S. Drug Enforcement Administration. (2014). Final rule: Schedules of controlled substances: Rescheduling hydrocodone combination products from Schedule III to Schedule II. Retrieved from [DEA website].
[2] Centers for Disease Control and Prevention. (2022). Guideline for prescribing opioids for chronic pain. MMWR, 71(1), 1-22.
[3] MarketResearch.com. (2022). Pain management market analysis.
[4] FDA. (2018). Safety communication: FDA advises against using certain combination opioid pain medications. Retrieved from [FDA website].
[5] Thomson Reuters. (2021). Opioid litigation settlements and financial impact.