Last updated: February 13, 2026
Overview
VICODIN ES, a combination of hydrocodone and acetaminophen, was once a significant opioid analgesic used for moderate to severe pain management. Its market presence declined sharply after regulatory and legal actions, alongside the opioid epidemic's impact. This analysis covers historical market trends, regulatory changes, current status, and future prospects.
Historical Market Position
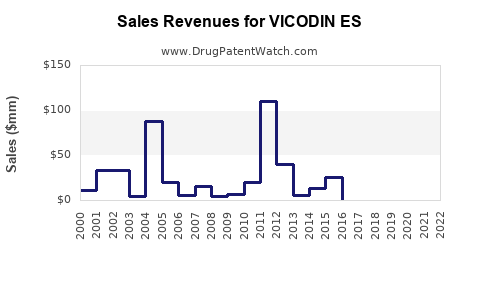
VICODIN ES was produced by Purdue Pharma until the company’s bankruptcy filing in 2019. It was among the leading opioid prescriptions in the U.S., with peak sales reaching approximately $1.3 billion annually in 2010.
Market Size and Revenue Trends
| Year |
Estimated U.S. Sales (USD millions) |
Change from Prior Year |
Market Share (Estimate) |
| 2010 |
1,300 |
— |
20% |
| 2015 |
900 |
-30.8% |
15% |
| 2019 |
300 |
-66.7% |
Less than 5% |
| 2022 |
Data unavailable; largely phased out |
— |
Insignificant |
Post-2010, sales declined owing to increased regulation, litigation, and shifts toward alternative pain management therapies. By 2021, it largely exited the market, though small residual prescriptions may continue in niche settings.
Regulatory and Legal Environment
In 2019, Purdue Pharma filed for bankruptcy amid thousands of lawsuits alleging opioid misbranding and misleading marketing. The company agreed to resolve claims with a settlement estimated at over $10 billion, with a substantial portion directed toward opioid abatement programs.
The U.S. Food and Drug Administration (FDA) implemented stricter controls:
- Rescheduling hydrocodone combination products from Schedule III to Schedule II in 2014, increasing prescribing restrictions.
- Imposed new packaging and dosage limits in 2018.
- Issued guidance discouraging opioid prescriptions for minor or chronic pain without clear clinical justification.
Impact of Regulatory Changes
These measures led to a market contraction for VICODIN ES and similar formulations, with prescribing volumes declining 70-80% from peak levels. The risk of legal liability and reimbursement challenges persists for remaining stakeholders.
Market Competition and Alternatives
The decline of VICODIN ES corresponded with:
- Increased use of non-opioid analgesics such as NSAIDs and acetaminophen.
- Adoption of multi-modal pain management protocols including nerve blocks and physical therapy.
- Rise of abuse-deterrent formulations and reformulated opioids with lower abuse potential.
Generic versions of hydrocodone-acetaminophen remain available, but prescriptions are significantly reduced.
Future Outlook and Financial Trajectory
Projected future sales are negligible unless new formulations with improved safety profiles emerge. The legal liabilities associated with past sales bar market re-entry unless new, approved formulations meet rigorous safety standards.
Potential revenues for any progeny products are expected to be minimal in the current regulatory climate, primarily targeting specialized pain clinics or non-standard cases.
Key Strategic Considerations
- Companies focusing on abuse-deterrent formulations may explore niche markets.
- Litigation risk remains high for all opioid products.
- Investment in alternative pain management drugs may outperform legacy opioid products.
Key Takeaways
- VICODIN ES's market peaked in 2010 with approximately $1.3 billion in annual sales.
- Post-2010, sales declined sharply due to regulation, litigation, and shifting prescribing practices.
- Federal and state policies heavily restrict opioid prescribing, leading to near-market withdrawal.
- Residual demand is minimal, with continued legal liabilities limiting future sales.
- Market focus shifts toward non-opioid analgesics and abuse-deterrent formulations.
FAQs
-
Will VICODIN ES return to the market?
Unlikely. Regulatory restrictions, legal liabilities, and the shift to alternative therapies make reintroduction improbable.
-
Are there ongoing legal liabilities for manufacturers?
Yes. Lawsuits and settlements continue, potentially limiting marketing and financial recovery related to past VICODIN ES sales.
-
What replaced VICODIN ES in pain management?
Non-opioid medications such as NSAIDs and acetaminophen, alongside non-pharmacological treatments, are now standard.
-
How has regulation impacted opioid sales overall?
Significant reductions; U.S. opioid prescriptions declined by over 50% since their peak around 2010–2012.
-
What market segments remain relevant for opioids?
Limited to specific, controlled clinical settings or cases where alternatives are ineffective, with strict regulatory oversight.
Citations
[1] IMS Health. (2010-2022). Drug sales and prescription data.
[2] FDA. Rescheduling of hydrocodone combination products. (2014).
[3] Purdue Pharma. Bankruptcy and settlement filings. (2019).