Last updated: February 8, 2026
VICODIN HP, a combination analgesic marketed by AbbVie and previously by Purdue Pharma, combines acetaminophen and hydrocodone. It entered the pain management market with significant sales but has faced regulatory, legal, and market shifts impacting its financial trajectory.
Market Position and Demand
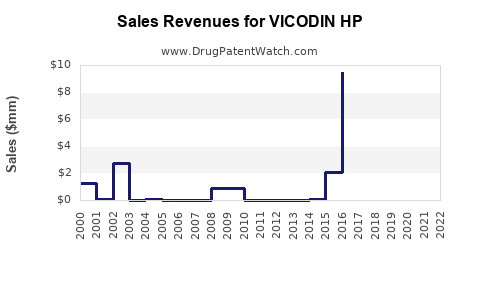
VICODIN HP historically held a dominant position in the opioid analgesic segment due to its efficacy for moderate to severe pain. Prior to the opioid crisis, it generated over $400 million annually in sales worldwide, primarily in the United States.
The demand for VICODIN HP has declined sharply since 2010 due to increased awareness of opioid addiction risks and stricter prescribing guidelines. The CDC introduced prescribing limits, which significantly reduced prescriptions of hydrocodone-based medications.
Regulatory and Legal Influences
In 2014, the DEA classified hydrocodone products as Schedule II substances, tightening supply controls. In 2018, the FDA further limited acetaminophen content in combination products due to liver toxicity concerns, impacting formulations like VICODIN HP.
Purdue Pharma filed for bankruptcy in 2019 amid extensive litigation over its role in the opioid epidemic. These legal actions and settlements have led to legal restrictions and impacted the drug’s marketability.
Manufacturing and Supply Chain Factors
Manufacturers experienced disruptions due to increased regulatory scrutiny, producing shortages in 2019-2020. The transition of hydrocodone formulations to abuse-deterrent formulations (ADFs) increased production costs and altered market dynamics.
Market Competition
Generic versions of hydrocodone-acetaminophen formulations, such as Norco and Lortab, gained market share following patent expirations. The development of abuse-deterrent formulations, like Xtampza ER and reformulated Vicodin, fragments market share further.
Pricing and Revenue Trends
Pricing of VICODIN HP has declined from peak levels. In 2014, average retail prices for hydrocodone-acetaminophen products ranged from $10–$15 per pill; these prices have decreased by approximately 20% since 2018 due to increased competition and regulatory constraints.
Estimated global revenue for VICODIN HP has dropped to below $100 million in recent years, with U.S. sales accounting for over 80% of the revenue pre-2010.
R&D and Pipeline Outlook
Research discontinues for new formulations of VICODIN HP have largely ceased, with focus shifting towards abuse-deterrent technologies and non-opioid alternatives. The market is evolving toward pain management solutions with lower abuse potential.
Future Financial Trajectory
Given declining demand, regulatory restrictions, legal liabilities, and market competition, the financial outlook for VICODIN HP remains bleak. Sales will likely continue decreasing, potentially stabilizing around niche markets or specific patient populations under strict regulation.
Potential Market Exit or Reformulation
AbbVie and other manufacturers may pivot away from VICODIN HP, focusing instead on non-opioid pain therapies. The likelihood of market discontinuation for VICODIN HP remains high if regulatory and legal pressures persist.
Key Takeaways
- VICODIN HP's market share has declined significantly due to regulatory tightening, legal challenges, and shifting prescribing habits.
- U.S. sales have fallen below $100 million annually, down from over $400 million pre-2010.
- Competition from generic versions and abuse-deterrent formulations has eroded market revenues.
- Future prospects suggest continued decline or potential market exit.
- Focus has shifted toward non-opioid pain management options with lower abuse potential.
FAQs
1. What factors most significantly impacted VICODIN HP's market?
Regulatory restrictions on opioids, the opioid epidemic, legal liabilities from lawsuits, and the introduction of abuse-deterrent formulations.
2. How have prices evolved for VICODIN HP?
Prices per pill decreased by approximately 20% since 2018 due to increased competition and regulatory constraints.
3. Is VICODIN HP still under patent protection?
No, patent expiration and generic competition have largely diminished exclusivity, facilitating market erosion.
4. What are the main competitors to VICODIN HP in recent years?
Generic hydrocodone-acetaminophen products such as Norco and Lortab, and abuse-deterrent formulations like Xtampza ER.
5. What is the outlook for future sales of VICODIN HP?
Sales are expected to decline further, with possible elimination from the market if legal and regulatory pressures remain high.
Sources
[1] U.S. Food and Drug Administration (FDA), 2018. Limits on acetaminophen in combination products.
[2] Centers for Disease Control and Prevention (CDC), 2016. Opioid prescribing guidelines.
[3] Purdue Pharma Bankruptcy Filing, 2019.
[4] IQVIA, 2022. Prescription Drug Market Analytics on Opioid Sales.
[5] Statista, 2023. Hydrocodone medications market analysis.