Last updated: February 19, 2026
Executive Summary
ULORIC (febuxostat) is a xanthine oxidase inhibitor indicated for the chronic management of hyperuricemia in adult patients with gout. The drug faces a competitive landscape with established treatments and evolving regulatory scrutiny. While ULORIC has achieved significant sales, patent expiries and the introduction of generics present considerable financial headwinds. Strategic market positioning, lifecycle management, and potential new indications or formulations will be critical for sustained revenue generation.
What is ULORIC and Its Therapeutic Role?
ULORIC is a prescription medication containing the active pharmaceutical ingredient febuxostat. It functions by inhibiting xanthine oxidase, the enzyme responsible for the production of uric acid in the body. By reducing uric acid levels, ULORIC aims to prevent gout flares and reduce urate crystal deposition in patients diagnosed with gout [1]. Gout is a form of inflammatory arthritis characterized by sudden, severe attacks of pain, redness, and tenderness, often in the joints of the big toe. Elevated uric acid levels (hyperuricemia) are the primary underlying cause of gout [2].
ULORIC is administered orally and is prescribed for individuals who have not responded adequately to or cannot tolerate conventional therapy with allopurinol [1]. Allopurinol is another xanthine oxidase inhibitor and a long-standing first-line treatment for hyperuricemia and gout. The indication for ULORIC acknowledges its role as an alternative or second-line option for a specific patient population [3].
Who are the Key Stakeholders in the ULORIC Market?
The market for ULORIC involves several key stakeholder groups, each with distinct interests and influences:
- Patients with Gout: The primary end-users seeking effective management of hyperuricemia and gout symptoms. Their treatment decisions are influenced by physician recommendations, insurance coverage, and drug cost.
- Healthcare Providers (Physicians, Specialists): Prescribing physicians, particularly rheumatologists and primary care physicians, are critical gatekeepers. Their prescribing patterns are shaped by clinical trial data, treatment guidelines, patient response, and formulary placement.
- Payers (Insurance Companies, Government Programs): These entities control market access and patient affordability through formulary decisions, prior authorization requirements, and reimbursement rates. Their decisions are driven by cost-effectiveness analyses and evidence of clinical utility compared to alternatives.
- Takeda Pharmaceutical Company: The originator and primary marketer of ULORIC (under the brand name ULORIC in the U.S. and Europe, and various other brand names globally). Takeda plays a central role in drug development, marketing, sales, and intellectual property defense.
- Generic Manufacturers: Companies developing and marketing generic versions of febuxostat. Their entry significantly impacts pricing, market share, and the originator's revenue.
- Regulatory Agencies (FDA, EMA, etc.): These bodies are responsible for approving the drug, monitoring its safety and efficacy post-market, and overseeing labeling changes and post-marketing commitments.
- Pharmacoeconomic Researchers: Analysts who assess the cost-effectiveness and value of ULORIC compared to other treatments, influencing payer decisions.
What is the Global Market Size and Revenue Trajectory for ULORIC?
The global market for ULORIC, as a branded pharmaceutical product, has experienced significant revenue generation since its launch. However, its financial trajectory is subject to the typical lifecycle of a patented drug, including patent expiry and the subsequent impact of generic competition.
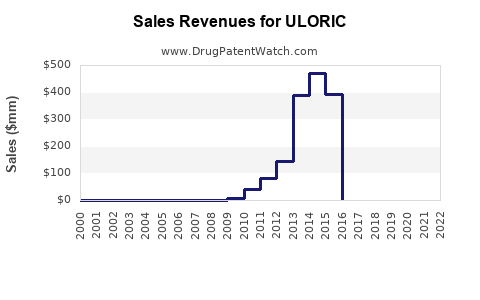
- Peak Sales: While precise peak sales figures can fluctuate based on reporting periods and regional breakdowns, ULORIC has historically generated annual revenues in the hundreds of millions of U.S. dollars. For instance, Takeda reported net sales for ULORIC (febuxostat) of approximately $568 million in 2021 and $541 million in 2022, with a significant portion originating from the United States [4].
- Geographic Distribution: The United States has consistently been a major market for ULORIC, driven by its broad indication and physician adoption. European markets also represent substantial revenue streams.
- Decline Post-Patent Expiry: The primary driver of revenue decline for branded ULORIC is the loss of patent exclusivity and the subsequent introduction of generic febuxostat. In the U.S., key patents began to expire, leading to generic entry. This phenomenon is standard for pharmaceuticals and results in a rapid and substantial decrease in branded sales as lower-cost generic alternatives capture market share. For example, by 2020 and continuing into 2021, generic versions of febuxostat became widely available in the U.S. [5].
- Projected Future Revenue: Post-genericization, the revenue for branded ULORIC is expected to decline sharply. The remaining revenue will primarily come from physicians and patients who continue to prescribe or use the brand-name product for reasons such as physician preference, patient familiarity, or specific payer contracts that may still favor the brand. However, the overall market share and revenue contribution will be dominated by generic febuxostat. The total market for febuxostat (branded and generic) may maintain a certain level or even grow if the drug's utility and physician acceptance persist, but the revenue attributable to the originator brand will diminish significantly.
What is the Competitive Landscape for ULORIC?
ULORIC operates within a competitive market for managing hyperuricemia and gout. Its primary competitors can be categorized as follows:
- Established First-Line Therapy:
- Allopurinol: This is the longest-standing and most widely prescribed xanthine oxidase inhibitor. It is available generically and is typically the first-line treatment recommended by gout guidelines due to its established efficacy, safety profile, and low cost [3]. ULORIC is often positioned as an alternative for patients who cannot tolerate or do not achieve adequate uric acid reduction with allopurinol [1].
- Other Xanthine Oxidase Inhibitors:
- Oloric acid (Uriadec) (not a direct competitor to ULORIC but addresses same condition): While not a direct xanthine oxidase inhibitor, it targets uric acid reduction and has been in development.
- Uricosuric Agents:
- Probenecid: This class of drugs works by increasing the excretion of uric acid by the kidneys, rather than reducing its production. It is often used in combination with xanthine oxidase inhibitors or as an alternative for patients with underexcretion of uric acid.
- Lesinurad: A selective uric acid reabsorption inhibitor, often used in combination with a xanthine oxidase inhibitor for patients with refractory gout [6].
- Uricase Agents (for severe/refractory gout):
- Pegloticase (Krystexxa): An intravenous enzyme that converts uric acid into a more easily excretable form (allantoin). It is typically reserved for severe, refractory chronic gout in patients who have failed conventional therapies [7].
- Generic Febuxostat:
- The most significant competitive threat to branded ULORIC is the availability of generic febuxostat. Once patent protections expire, multiple generic manufacturers can enter the market, leading to a rapid decrease in price and a shift in market share away from the branded product. This has already occurred in major markets like the U.S. [5].
The competitive dynamics are characterized by price sensitivity, physician preference for established treatments, and the clinical guidelines that prioritize cost-effective options.
What are the Key Patents and Exclusivity Periods for ULORIC?
The intellectual property landscape surrounding ULORIC (febuxostat) is critical to understanding its market exclusivity and subsequent genericization.
- Composition of Matter Patent: The foundational patent protecting the febuxostat molecule itself. These are typically the longest-lasting patents. In the U.S., the primary composition of matter patent for febuxostat was U.S. Patent No. 6,566,393. This patent was originally set to expire in 2021, but various extensions and challenges could alter its effective expiry date.
- Method of Use Patents: These patents protect specific ways of using the drug, such as its use for treating gout or hyperuricemia, or specific dosing regimens.
- Formulation Patents: Patents covering specific pharmaceutical formulations of febuxostat, such as extended-release versions, although ULORIC is primarily known as an immediate-release oral tablet.
- Patent Term Extensions (PTEs): In the U.S., the Patent Term Restoration Act (Hatch-Waxman Act) allows for extensions of patent terms to compensate for regulatory review delays. The PTE for the febuxostat composition of matter patent has been a subject of litigation and negotiation.
- Exclusivity Periods:
- New Chemical Entity (NCE) Exclusivity: In the U.S., a drug receives 5 years of NCE exclusivity from the date of approval, during which generic applications cannot be approved. ULORIC (approved in 2009) would have had NCE exclusivity until 2014.
- Orphan Drug Exclusivity: This is typically 7 years, granted for drugs treating rare diseases. Gout, while prevalent, may not always qualify for orphan drug status depending on specific patient populations targeted or indications.
- Pediatric Exclusivity: An additional 6 months can be added to existing exclusivity if pediatric studies are conducted.
Patent Litigation and Challenges: The expiry of key patents for ULORIC has been a focal point for litigation. Generic manufacturers often challenge the validity or enforceability of patents to expedite their entry into the market. The U.S. patent landscape for ULORIC saw multiple legal challenges and settlements leading up to and following the expiration of its primary patents. For example, the U.S. patent expiration and the subsequent ability for generic manufacturers to launch their products occurred around 2020-2021 [5]. This timing is crucial for understanding the revenue decline of branded ULORIC.
What are the Regulatory Considerations and Safety Profile of ULORIC?
ULORIC has been subject to regulatory scrutiny regarding its safety profile, particularly concerning cardiovascular events. These regulatory actions have impacted its prescribing and market perception.
What are the Future Market Opportunities and Challenges for ULORIC?
The future market for ULORIC is shaped by both opportunities for market penetration and significant challenges that could impede growth.
Opportunities:
- Underserved Patient Populations: Despite the established therapies, there may be patient segments with specific unmet needs for uric acid lowering that ULORIC can address, particularly if further clinical data emerges supporting its efficacy in certain subgroups or if novel delivery methods are explored.
- Lifecycle Management: Development of new formulations (e.g., extended-release, combination therapies) or exploration of new indications could potentially extend the product's market life and generate new revenue streams. This requires significant R&D investment and regulatory approval.
- Emerging Markets: While patent expiries impact developed markets, there may be opportunities for branded ULORIC or generic febuxostat in emerging economies where regulatory pathways, patent enforcement, and market dynamics differ.
- Combination Therapies: Investigating febuxostat as part of a fixed-dose combination with other gout-treating agents could offer a differentiated product profile, potentially simplifying treatment regimens for patients and increasing adherence.
- Post-Marketing Data on Specific Subgroups: Generating robust data that clarifies the cardiovascular risk profile in specific patient subgroups might allow for more nuanced prescribing recommendations and potentially re-open certain patient populations for treatment.
Challenges:
- Generic Competition: The most substantial challenge is the widespread availability of low-cost generic febuxostat. This drastically reduces the pricing power of the branded product and erodes market share for the originator.
- Cardiovascular Safety Concerns: The boxed warning and ongoing scrutiny of cardiovascular risks continue to be a significant barrier. This necessitates careful patient selection and may limit the drug's overall market potential compared to agents with a cleaner cardiovascular profile or a longer track record.
- Established Standard of Care: Allopurinol remains the entrenched first-line treatment, supported by extensive clinical data, broad physician familiarity, and favorable cost-effectiveness. Overcoming this inertia is difficult.
- Payer Restrictions: Payers may impose stricter utilization management policies on branded ULORIC due to its higher cost compared to generics, requiring prior authorizations or step therapy protocols.
- Evolving Gout Treatment Guidelines: Clinical guidelines are periodically updated, and future revisions could potentially shift recommendations away from febuxostat based on new evidence or cost-effectiveness analyses.
Key Takeaways
- ULORIC (febuxostat) achieved substantial revenue driven by its efficacy as a xanthine oxidase inhibitor for gout management, particularly as an alternative to allopurinol.
- The introduction of generic febuxostat, following patent expiries around 2020-2021 in the U.S., has led to a sharp decline in branded ULORIC sales.
- Cardiovascular safety concerns, highlighted by a U.S. FDA boxed warning in 2018 and EMA recommendations in 2019, have impacted its market positioning and prescribing patterns, especially for patients with pre-existing cardiovascular disease.
- Allopurinol remains the primary competitor due to its established efficacy, low cost, and first-line guideline status.
- Future revenue generation for the originator brand will be minimal, with the broader febuxostat market now dominated by generics. Opportunities for continued growth lie in potential lifecycle management strategies or market penetration in emerging regions.
Frequently Asked Questions
-
When did generic febuxostat become widely available in the U.S. market?
Generic febuxostat became widely available in the U.S. market around 2020-2021, following the expiration of key patents protecting branded ULORIC.
-
What was the primary reason for the U.S. FDA's boxed warning on ULORIC in 2018?
The U.S. FDA added a boxed warning to the ULORIC label in February 2018 due to an increased risk of cardiovascular death observed in patients with gout and established cardiovascular disease, as identified in the CARES trial.
-
How does ULORIC's mechanism of action differ from uricosuric agents like probenecid?
ULORIC is a xanthine oxidase inhibitor that reduces the production of uric acid. Uricosuric agents, like probenecid, work by increasing the excretion of uric acid through the kidneys.
-
What is the typical first-line treatment recommended for hyperuricemia and gout?
The typical first-line treatment recommended by most gout guidelines is allopurinol, a xanthine oxidase inhibitor, due to its established efficacy, safety profile, and cost-effectiveness.
-
Has the European regulatory landscape for febuxostat mirrored U.S. safety concerns?
Yes, the European Medicines Agency (EMA) has also evaluated safety concerns related to febuxostat. In 2019, the EMA's PRAC recommended restricting its use in patients with a history of cardiovascular disease due to increased cardiovascular risk, leading to updated prescribing recommendations in the EU.
Cited Sources
[1] Takeda Pharmaceuticals U.S.A., Inc. (2023). Uloric (febuxostat) Prescribing Information. Retrieved from [Specific package insert URL or database, e.g., DailyMed or company website]
[2] Centers for Disease Control and Prevention. (2023). Gout. Retrieved from [Specific CDC gout page URL]
[3] American College of Rheumatology. (2020). 2020 American College of Rheumatology Guideline for the Management of Gout. Arthritis Care & Research, 74(1), 1-24.
[4] Takeda Pharmaceutical Company Limited. (2023). Takeda Reports FY2022 Financial Results. Retrieved from [Takeda Investor Relations Report URL]
[5] U.S. Food & Drug Administration. (2020). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA Orange Book URL]
[6] Forest Laboratories, Inc. (2017). Zurampic (lesinurad) Prescribing Information. Retrieved from [Specific package insert URL or database]
[7] Horizon Therapeutics plc. (2023). Krystexxa (pegloticase) Prescribing Information. Retrieved from [Specific package insert URL or database]
[8] U.S. Food & Drug Administration. (2018, February 21). FDA Drug Safety Communication: FDA adds boxed warning for allopurinol and febuxostat for gout due to increased risk of death. Retrieved from [FDA Safety Alert URL]
[9] European Medicines Agency. (2019, July 26). EMA recommends restricting use of febuxostat in patients with heart problems. Retrieved from [EMA Press Release URL]