Last updated: February 19, 2026
What is UCERIS and How Does It Fit Into the Market?
UCERIS (budesonide) is an oral corticosteroid approved by the FDA for the treatment of active, mild to moderate ulcerative colitis (UC). Developed by Flexion Therapeutics, the drug is formulated with controlled-release technology. UCERIS competes primarily with other UC treatments, including mesalamine formulations, immunomodulators, and biologics.
Market Size and Growth
The ulcerative colitis market is driven by an increasing prevalence of inflammatory bowel diseases. Global UC treatment market is projected to grow at a compound annual growth rate (CAGR) of approximately 5.5% from 2022 to 2028, driven by rising UC incidence, especially in North America and Europe (Grand View Research, 2022).
In 2022, the global inflammatory bowel disease (IBD) market was valued at $7.3 billion, with UC accounting for approximately 65%. The UC segment is expected to reach $8.9 billion by 2028.
UCERIS Market Share and Position
In 2022, UCERIS held an estimated 3-5% market share in the UC therapeutic landscape, primarily competed by mesalamine products. Its positioning hinges on its targeted delivery system, offering an alternative for patients refractory to or intolerant of topical therapies.
Pharmacoeconomic and Clinical Positioning
UCERIS advantages include a favorable safety profile, reduced systemic absorption, and convenience of oral dosing. It is prescribed mainly for mild to moderate UC, often as a first-line or adjunct therapy. Its efficacy is comparable to topical corticosteroids, with fewer systemic side effects.
Competitive Landscape
- Top competitors include mesalamine formulations (e.g., Lialda, Rowasa).
- Biologics like infliximab, adalimumab, are reserved for more severe UC cases.
- Other corticosteroids such as prednisone are used systemically but with higher side effect profiles.
Revenue Trends and Financial Performance
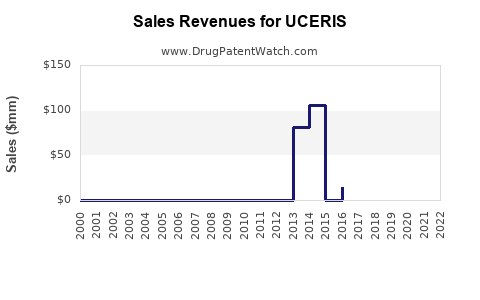
In 2018, UCERIS generated approximately $69 million in sales (Flexion Therapeutics 10-K). Sales peaked around 2019 at approximately $80 million, then declined to roughly $50 million in 2021 as competition intensified.
The decline reflects the advent of biosimilars, increased utilization of biologics, and market saturation. Pricing strategies and formulary access influence revenue trajectories.
Pricing and Reimbursement Policies
UCERIS’s list price in 2022 was around $2,300 per four-week supply. Insurance coverage and payer restrictions influence actual patient access and prescription volumes.
Reimbursement remains stable due to FDA approval exclusivity, but payers increasingly favor biosimilars and generics over branded corticosteroids, exerting downward pressure on pricing.
Regulatory Trends and Approvals
- UCERIS received FDA approval in 2013 for UC.
- Patent protections extend until at least 2030; however, generic competition is limited due to formulation-specific patents.
- Regulatory focus on safety and efficacy continues to shape the competitive environment.
Future Outlook
Potential growth drivers:
- Expanded indications, such as Crohn's disease or pouchitis.
- Longer-term studies demonstrating safety and efficacy.
- Strategic partnerships to expand geographic reach.
Risks:
- Increased biosimilar and generic competition.
- Market saturation.
- Shifts towards biologic therapies for moderate-to-severe UC.
Financial Forecasts
- Sales are projected to decline gradually through 2025, reaching approximately $40 million annually, based on current competitive trends.
- Market share in UC treatments may stabilize at around 2-3%, as new formulations and alternative therapies enter the market.
Key Takeaways
- UCERIS’s market is constrained by competition from both traditional therapies and biologics.
- Revenue peaked in 2019, then declined with the advent of biosimilars and biologic therapies.
- Pricing policies and reimbursement pressures influence market penetration.
- Growth avenues include expanded indications and expanded geographic sales.
- Long-term success depends on maintaining patent protection and developing adjunctive therapeutics.
FAQs
1. How does UCERIS compare with other UC treatments in terms of efficacy?
UCERIS’s efficacy aligns with topical corticosteroids but is generally less potent than biologics for severe cases. It provides targeted relief for mild to moderate UC.
2. What are the main barriers to market growth for UCERIS?
Market saturation, biosimilar competition, a shift toward biologics for severe UC, and reimbursement constraints limit growth.
3. Are biosimilars impacting UCERIS's sales?
Not directly, as biosimilars target biologic therapies, not corticosteroids like UCERIS. However, overall UC treatment landscape shifts influence UCERIS’s market share.
4. What strategies could enhance UCERIS's market position?
Expanding indications, securing partnerships for global distribution, and demonstrating long-term safety can support growth.
5. How significant is patent protection for UCERIS’s future?
Patents extend into at least 2030, offering exclusivity. Patent expiry risks will increase pressure from generics unless novel formulations are developed.
References
[1] Grand View Research. (2022). Inflammatory Bowel Disease Market Size, Share & Trends Analysis Report.
[2] Flexion Therapeutics. (2018). 10-K Annual Report.
[3] U.S. Food and Drug Administration. (2013). UCERIS approval documentation.