Last updated: February 19, 2026
Tropicamide, a synthetic anticholinergic medication, is primarily utilized in ophthalmology for its mydriatic (pupil-dilating) and cycloplegic (paralyzing accommodation) properties. Its market is characterized by established generic competition and stable demand, driven by routine diagnostic procedures and post-operative care. Financial performance is primarily influenced by volume sales and pricing within the ophthalmic pharmaceutical segment.
What is Tropicamide's Current Market Position?
Tropicamide is a well-established ophthalmic drug with a mature market presence. Its utility is centered on diagnostic ophthalmology, enabling detailed examination of the retina and fundus. It is also used to facilitate optometric procedures and pre- or post-operative management of certain eye conditions. The drug's mechanism of action involves blocking muscarinic acetylcholine receptors in the iris sphincter and ciliary muscle, leading to pupil dilation and temporary paralysis of the eye's focusing ability.
Key Market Segments and Applications
Tropicamide is predominantly used in the following areas:
- Diagnostic Ophthalmology: This represents the largest segment. Dilating the pupil allows ophthalmologists and optometrists to gain a clear, unobstructed view of the retina, optic nerve, and blood vessels. This is crucial for detecting conditions such as diabetic retinopathy, macular degeneration, glaucoma, and retinal detachments.
- Pre- and Post-Operative Care: Tropicamide can be employed before or after certain ophthalmic surgeries, such as cataract surgery or laser procedures, to aid in visualization and manage intraocular inflammation or spasm.
- Pediatric Eye Exams: Its rapid onset and relatively short duration of action make it suitable for dilating pupils in children, facilitating eye examinations.
Competitive Landscape
The market for tropicamide is highly competitive and dominated by generic manufacturers. Several pharmaceutical companies produce and distribute tropicamide formulations, primarily as ophthalmic solutions. Brand-name exclusivity expired decades ago, leading to widespread generic availability.
- Major Generic Suppliers: Companies such as Akorn, Inc., Bausch Health Companies Inc., and various smaller regional manufacturers are active in this market.
- Pricing Pressures: The presence of numerous generic alternatives exerts significant downward pressure on pricing. Manufacturers compete primarily on cost and supply chain reliability.
- Limited Differentiation: Product differentiation is minimal, with most products offering similar concentrations (e.g., 0.5% and 1.0%) and delivery systems (e.g., multi-dose vials, single-use droppers).
What is Tropicamide's Financial Trajectory and Market Value?
Tropicamide's financial trajectory is indicative of a mature pharmaceutical product. Revenue generation is driven by high-volume sales rather than significant price appreciation or new market expansion. The market value is largely stable, reflecting consistent demand for its established clinical applications.
Revenue Drivers
- Volume Sales: The primary driver of revenue for tropicamide is the sheer volume of units sold annually. Its widespread use in routine eye exams by a large number of practitioners globally ensures consistent demand.
- Geographic Distribution: Sales are distributed across developed and emerging markets, with demand correlating to the prevalence of eye care services and the number of ophthalmologists and optometrists.
- Procurement Channels: Significant volumes are procured through hospital systems, large ophthalmology clinics, and government health programs, often through tender processes that emphasize cost-effectiveness.
Pricing Dynamics
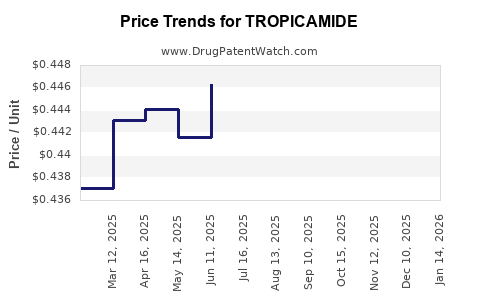
- Generic Pricing: As a generic drug, tropicamide prices are determined by supply and demand, manufacturing costs, and competitive bidding. Prices per unit are generally low.
- Contractual Agreements: A substantial portion of tropicamide sales occurs through negotiated contracts with healthcare providers and distributors, where volume discounts are common.
- Inflationary Impact: While overall healthcare costs may rise, the pricing of highly commoditized generics like tropicamide tends to lag behind general inflation, or even experience minor declines due to intense competition.
Estimated Market Size and Growth
Precise global market size figures for tropicamide are not separately reported by major market research firms, as it is often aggregated within broader ophthalmic drug categories or as part of the generic ophthalmics market. However, based on its application and competitive landscape, its market value is estimated to be in the tens of millions of U.S. dollars annually.
- Growth Rate: The market for tropicamide is expected to exhibit low single-digit growth, primarily driven by population growth and an aging population that often requires more frequent eye examinations. It is not a product with significant expansion potential through new indications or novel formulations. The growth is largely tied to the expansion of ophthalmic diagnostic services globally.
- Market Value: While specific figures are proprietary, it is reasonable to infer a stable but not high-growth market. For example, if a manufacturer sells 10 million units annually at an average price of $0.50 per unit (a plausible wholesale price for a generic ophthalmic solution), this alone would generate $5 million in revenue for that specific product. Considering multiple manufacturers, the global market is likely in the $50 million to $150 million range.
What are the Regulatory and Intellectual Property Considerations?
The regulatory landscape for tropicamide is mature, with established approval pathways for generic versions. Intellectual property protection for the original compound has long expired, meaning no patent barriers exist for generic manufacturers.
Regulatory Approvals
- FDA and EMA: Tropicamide is approved by major regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for its indicated uses. Generic versions undergo abbreviated new drug applications (ANDAs) in the U.S., demonstrating bioequivalence to the reference listed drug.
- Manufacturing Standards: Manufacturers must adhere to Good Manufacturing Practices (GMP) to ensure product quality, safety, and efficacy. Regulatory inspections are a standard component of maintaining manufacturing compliance.
- Pharmacopoeial Standards: Tropicamide formulations must meet the specifications outlined in major pharmacopoeias, such as the United States Pharmacopeia (USP) and the European Pharmacopoeia (Ph. Eur.), regarding identity, purity, and potency.
Intellectual Property Status
- Expired Patents: The original patents covering tropicamide and its synthesis have long since expired. This has allowed for the widespread development and marketing of generic versions by multiple pharmaceutical companies.
- No New Market Exclusivity: There are no active patent exclusivities that would prevent generic competition. This is a defining characteristic of tropicamide's market.
- Formulation Patents: While the active pharmaceutical ingredient (API) is off-patent, there could theoretically be patents on novel formulations (e.g., extended-release, preservative-free variants) or specific manufacturing processes. However, for standard ophthalmic solutions, such patents are unlikely to be a significant barrier to entry or to command substantial market premiums.
What are the Future Outlook and Potential Challenges?
The future outlook for tropicamide is characterized by continued stable demand and the persistent threat of generic competition. Key challenges will involve maintaining profitability in a highly price-sensitive market and ensuring supply chain resilience.
Market Outlook
- Sustained Demand: Demand for tropicamide is expected to remain stable, driven by the ongoing need for mydriatic and cycloplegic agents in routine eye care. Global aging populations and increasing awareness of eye health will continue to support this demand.
- Generic Dominance: The market will continue to be dominated by generic products. Innovation in this space is unlikely to significantly alter market dynamics, as current formulations are highly effective and cost-efficient.
- Emerging Markets: Growth in emerging markets, where access to eye care services is expanding, could provide incremental increases in volume sales.
Potential Challenges
- Price Erosion: Ongoing price erosion due to intense competition is the most significant challenge. Manufacturers must optimize production costs to remain competitive.
- Supply Chain Vulnerabilities: Like many generic pharmaceuticals, tropicamide supply chains can be vulnerable to disruptions, including API shortages, manufacturing issues, or geopolitical events, which could impact availability and potentially lead to price spikes in specific regions.
- Regulatory Scrutiny: While established, regulatory oversight of manufacturing and quality control remains rigorous. Any lapse in compliance can lead to product recalls and reputational damage.
- Competition from Alternatives: Although less common for routine diagnostics, the theoretical possibility of alternative diagnostic technologies or even other classes of drugs emerging that could reduce the need for mydriatics presents a long-term, low-probability risk. For example, advancements in handheld imaging devices that require less pupil dilation could, over time, affect demand.
Key Takeaways
Tropicamide is a mature generic ophthalmic drug with stable demand driven by diagnostic ophthalmology. The market is characterized by intense price competition among generic manufacturers, limiting revenue growth potential. Intellectual property for the active ingredient has expired, ensuring continued generic availability. Future growth is expected to be low single-digit, primarily from emerging markets, while challenges include price erosion and supply chain stability.
Frequently Asked Questions
- Is tropicamide still protected by any active patents?
No, all primary patents covering tropicamide as an active pharmaceutical ingredient have expired.
- What is the primary driver of tropicamide sales revenue?
Revenue is driven by high-volume sales, as pricing for this generic drug is highly competitive.
- Are there significant opportunities for market expansion with tropicamide?
Market expansion opportunities are limited; growth is expected to be marginal, primarily tied to the expansion of ophthalmic diagnostic services globally.
- What is the typical concentration of tropicamide in ophthalmic solutions?
Tropicamide ophthalmic solutions are commonly available in 0.5% and 1.0% concentrations.
- What is the main challenge faced by tropicamide manufacturers?
The primary challenge is persistent price erosion due to the highly competitive generic market.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/approved-drug-products-therapeutic-equivalence-evaluations-orange-book
[2] European Medicines Agency. (n.d.). Human medicines. Retrieved from https://www.ema.europa.eu/en/medicines
[3] United States Pharmacopeia. (n.d.). Pharmacopeial Forum. Retrieved from https://www.usppf.com/
[4] European Pharmacopoeia. (n.d.). Homepage. Retrieved from https://www.edqm.eu/en/european-pharmacopoeia-ph-eur