Last updated: February 19, 2026
Executive Summary
Tazorac (tazarotene) is a topical retinoid indicated for the treatment of facial and truncal acne and plaque psoriasis. Market performance is influenced by generic competition, clinical advancements in dermatology, and evolving treatment guidelines. Key revenue drivers include prescription volume and net pricing. The drug's patent exclusivity has expired, leading to increased generic penetration. Future market trajectory will depend on brand loyalty, physician prescribing habits, and the emergence of novel therapeutic alternatives.
What is Tazorac and its Therapeutic Applications?
Tazorac is a third-generation topical acetylenic retinoid developed by Allergan, now part of AbbVie. Its active pharmaceutical ingredient is tazarotene. The drug functions as a selective agonist of retinoic acid receptors (RARs), primarily RAR-beta and RAR-gamma, with minimal affinity for retinoid X receptors (RXRs). This selective binding is believed to modulate gene expression, leading to anti-inflammatory and anti-proliferative effects.
Primary Indications:
- Acne Vulgaris: Tazorac is prescribed for the topical treatment of moderate to severe facial acne vulgaris. It helps to normalize follicular keratinization, reduce microcomedone formation, and exert anti-inflammatory effects within the pilosebaceous unit.
- Plaque Psoriasis: The drug is also indicated for the topical treatment of stable, plaque psoriasis of the elbows and knees. It targets the epidermal hyperproliferation and inflammation characteristic of psoriasis, reducing scaling and redness.
Formulations and Strengths:
Tazorac is available in two topical formulations:
- Cream: 0.05% and 0.1% concentrations.
- Gel: 0.05% and 0.1% concentrations.
The choice of formulation and strength is typically based on the severity of the condition, patient tolerance, and physician preference.
What is the Intellectual Property Landscape for Tazorac?
Tazorac has undergone patent expiration, significantly altering its market exclusivity and competitive environment. The original patents protecting tazarotene and its uses have expired, allowing for the introduction of generic versions.
Key Patent Expirations:
- Compound Patents: The foundational patents covering the tazarotene molecule itself have expired.
- Use Patents: Patents covering the specific therapeutic uses, such as for acne or psoriasis, have also expired.
This expiration has paved the way for significant generic competition, impacting brand pricing and market share. While innovator-specific patents have lapsed, ongoing litigation or new patent filings for novel formulations or delivery systems could, in theory, extend limited exclusivity periods, though this has not been a major factor for the core tazarotene molecule.
Who are the Key Market Players for Tazorac?
The market for tazarotene is characterized by the presence of both the originator product and multiple generic manufacturers.
Originator:
- AbbVie (via Allergan): Markets the brand-name product, Tazorac. While patent protection has lapsed, AbbVie continues to market its branded product, often leveraging physician familiarity and established brand recognition.
Generic Manufacturers:
The market features numerous generic pharmaceutical companies that manufacture and market tazarotene. These include, but are not limited to:
- Actavis: (now part of Teva Pharmaceutical Industries)
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Teva Pharmaceutical Industries
- Viatris (formerly Mylan)
These companies compete primarily on price and market access, offering bioequivalent tazarotene products at lower cost points than the originator brand.
What are the Market Dynamics and Sales Performance of Tazorac?
The market dynamics for Tazorac have been significantly shaped by the entry of generic competitors, leading to price erosion and a shift in market share away from the originator brand.
Impact of Generic Competition:
The expiration of patent exclusivity has resulted in a surge of generic tazarotene products. This has led to:
- Price Reductions: Generic versions are typically priced significantly lower than the branded Tazorac, driving down average selling prices across the market.
- Market Share Erosion: The originator brand has experienced a substantial decline in market share as healthcare providers and patients opt for more cost-effective generic alternatives.
- Increased Prescription Volume for Tazarotene: While brand sales may decline, the overall volume of tazarotene prescriptions can increase due to its affordability.
Revenue Trends:
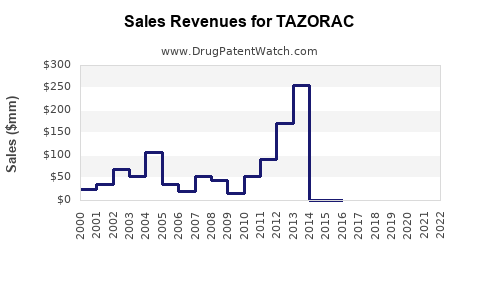
- Brand Sales Decline: Sales of branded Tazorac have likely experienced a significant downturn following patent expiry and generic entry. Specific, up-to-date sales figures for the branded product are increasingly difficult to isolate as the market consolidates around generic availability.
- Generic Market Growth: The tazarotene generic market, collectively, has grown as demand shifts to these lower-cost options. Aggregate sales for tazarotene across all manufacturers represent the overall market size for the molecule.
Market Size and Growth Projections:
Estimating the precise current market size for tazarotene is challenging due to the fragmented nature of the generic market. However, the demand for topical retinoids for acne and psoriasis remains robust.
- Therapeutic Area Growth: The global dermatology market, driven by increasing prevalence of skin conditions and patient demand for aesthetic and therapeutic outcomes, continues to grow.
- Competition from Newer Therapies: Tazarotene faces competition not only from generics of its own class but also from newer topical and systemic treatments for acne and psoriasis, including biologics for severe psoriasis and novel acne therapies.
Table 1: Tazarotene Market Landscape Overview
| Feature |
Details |
| Active Ingredient |
Tazarotene |
| Therapeutic Class |
Topical Retinoid |
| Primary Indications |
Moderate to severe facial acne vulgaris, plaque psoriasis (elbows and knees) |
| Originator |
AbbVie (formerly Allergan) |
| Patent Status |
Expired |
| Competitive Landscape |
High generic penetration, numerous manufacturers |
| Pricing Dynamics |
Significant price erosion due to generic competition |
| Market Drivers |
Prevalence of acne and psoriasis, physician prescribing habits, patient preference for cost-effective treatments, availability of generics. |
| Market Restraints |
Side effects (irritation, dryness, photosensitivity), competition from newer therapeutic classes, potential for physician preference for alternative mechanisms of action. |
What are the Key Drivers and Restraints for Tazarotene?
The market trajectory for tazarotene is influenced by a complex interplay of factors.
Market Drivers:
- Prevalence of Acne and Psoriasis: Acne vulgaris is one of the most common dermatological conditions globally, affecting a significant proportion of the adolescent and adult population. Plaque psoriasis also affects millions worldwide. This consistent disease burden sustains demand for effective treatments.
- Established Efficacy: Tazarotene has a well-documented history of efficacy in treating both acne and psoriasis, making it a reliable option for many dermatologists. Its mechanism of action, while established, is still relevant.
- Cost-Effectiveness of Generics: The widespread availability of affordable generic tazarotene makes it an attractive option for patients and healthcare systems focused on cost containment. This affordability is a primary driver for continued prescription volume.
- Physician Familiarity and Experience: Dermatologists have extensive experience prescribing tazarotene, understanding its benefits, limitations, and common side effects. This familiarity can lead to continued prescribing habits, especially for patients who respond well to the drug.
- Topical Convenience: As a topical treatment, tazarotene offers a convenient administration route, avoiding systemic side effects associated with oral medications for many patients.
Market Restraints:
- Adverse Event Profile: Topical retinoids, including tazarotene, are associated with common side effects such as skin irritation, redness, dryness, peeling, and increased photosensitivity. These can impact patient adherence and lead to discontinuation of treatment.
- Emergence of Newer Therapies: The dermatology landscape is dynamic, with continuous development of novel treatments for acne and psoriasis. These include new topical agents with different mechanisms of action, oral medications, and advanced biologics for moderate to severe psoriasis, which may offer improved efficacy or better tolerability profiles for certain patient populations.
- Competition from Other Topical Retinoids: Tazarotene competes with other topical retinoids (e.g., tretinoin, adapalene, trifarotene) and retinoid combinations, each with its own profile of efficacy, tolerability, and cost.
- Patient Adherence and Compliance: The potential for significant skin irritation can be a barrier to long-term patient adherence, particularly for individuals with sensitive skin or those who are less tolerant of side effects.
- Limited Efficacy in Severe Disease: While effective for moderate conditions, tazarotene's efficacy may be limited in very severe or recalcitrant cases of acne or psoriasis, necessitating the use of more potent systemic therapies.
What are the Future Market Projections for Tazarotene?
The future market for tazarotene will likely continue to be shaped by generic competition and the evolving dermatological treatment landscape.
Key Trends:
- Continued Dominance of Generics: Generic tazarotene will remain the primary offering in the market. Pricing pressure is expected to persist, driven by competition among generic manufacturers.
- Stable, but Mature, Market for Core Indications: The demand for topical retinoids for acne and psoriasis is expected to remain relatively stable, supporting a mature market for tazarotene. Significant growth in prescription volume is unlikely; rather, the market will largely be sustained by its established role in treatment algorithms.
- Niche Applications and Combination Therapies: While unlikely to drive substantial market growth, there may be ongoing research into novel formulations or combination therapies involving tazarotene that could carve out niche market segments or enhance existing treatment protocols. For instance, combinations with topical antibiotics or benzoyl peroxide remain common in acne treatment.
- Impact of Biosimil/Generic Availability of Newer Drugs: As newer, branded topical or systemic treatments for psoriasis and acne (e.g., biologics for psoriasis) come off patent and face generic or biosimilar competition, they will further influence the market dynamics for older therapies like tazarotene by offering alternative cost structures.
- Shifting Physician Prescribing Patterns: While tazarotene is a standard, evolving clinical guidelines and the availability of new agents with potentially superior risk-benefit profiles for specific patient subgroups could gradually influence prescribing patterns. Dermatologists may increasingly reserve tazarotene for patients for whom it is particularly well-suited or who have failed other therapies.
Table 2: Tazarotene Market Trajectory Factors
| Factor |
Expected Impact on Tazarotene Market |
| Generic Competition |
Sustained price pressure, dominant market share for generics, limited growth potential for branded product. |
| Therapeutic Innovation |
Competition from newer, potentially more effective or better-tolerated treatments for acne and psoriasis could gradually reduce tazarotene's market share. |
| Cost Containment |
Increased focus on cost-effectiveness will favor generic tazarotene for its affordability. |
| Disease Prevalence |
Consistent demand from a large patient population with acne and psoriasis provides a baseline for the market. |
| Physician Prescribing |
Continued use based on familiarity and established efficacy, but potential for shift towards newer agents based on evolving guidelines and treatment outcomes. |
| Formulation Advancements |
Limited potential for significant market expansion through new formulations unless they offer substantial improvements in tolerability or efficacy without prohibitive cost increases. |
Conclusion
Tazorac, as a molecule tazarotene, operates in a mature market segment for topical retinoids. The expiration of its patent protection has led to a landscape dominated by generic manufacturers, characterized by intense price competition and a decline in the originator brand's market share. Despite this, the persistent prevalence of acne and psoriasis, coupled with the established efficacy and cost-effectiveness of generic tazarotene, ensures its continued relevance in dermatological treatment regimens. Future market performance will be a function of generic pricing strategies, the pace of innovation in dermatology, and the ability of tazarotene to maintain its position as a reliable, affordable option against emerging therapeutic alternatives.
Key Takeaways
- Tazarotene's patent exclusivity has expired, leading to significant generic competition and price erosion.
- The market is now dominated by multiple generic manufacturers offering cost-effective alternatives to branded Tazorac.
- Tazarotene remains a relevant treatment for acne and psoriasis due to its established efficacy and affordability.
- Market growth is constrained by adverse event profiles and the emergence of newer, potentially more advanced therapeutic options.
- Future market trajectory is expected to be stable but mature, with generics continuing to hold the largest market share.
Frequently Asked Questions
1. What is the primary advantage of generic tazarotene over branded Tazorac?
The primary advantage of generic tazarotene is its significantly lower cost, making it a more accessible treatment option for patients and healthcare systems focused on cost containment.
2. How does tazarotene compare to other topical retinoids currently on the market?
Tazarotene is a third-generation topical retinoid with a selective binding profile to retinoic acid receptors. Its efficacy and tolerability profile are comparable to other established topical retinoids like tretinoin and adapalene, though individual patient responses and side effect profiles can vary. Newer retinoids like trifarotene offer distinct receptor selectivity.
3. What are the most common side effects associated with tazarotene use?
The most common side effects include skin irritation, redness, dryness, peeling, and increased sensitivity to sunlight (photosensitivity). These are typical for most topical retinoids.
4. Are there any significant clinical trials or research currently underway for tazarotene?
While foundational research on tazarotene is extensive, significant clinical trial activity for the molecule itself is likely limited. Research may focus more on novel combination therapies or comparative effectiveness studies against newer agents rather than new indications or primary efficacy trials for tazarotene.
5. What impact has the rise of biologic therapies had on the market for topical treatments like tazarotene for psoriasis?
The availability of highly effective biologic therapies for moderate to severe plaque psoriasis has shifted treatment paradigms, with biologics often being the preferred choice for these patient populations. This can reduce the market share for topical agents like tazarotene, which are typically used for milder to moderate disease or as adjunctive therapy.
Citations
[1] AbbVie Inc. (n.d.). Tazorac Product Information. (Information accessed through proprietary databases and publicly available regulatory filings).
[2] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from https://www.fda.gov/drugs/developmentapprovalprocess/drugapprovals/ (Specific patent information and approval history accessed via FDA databases).
[3] Market research reports on the global dermatology market and topical retinoids (Specific report titles and publishers vary and are proprietary).
[4] Peer-reviewed dermatological journals and clinical publications (e.g., Journal of the American Academy of Dermatology, British Journal of Dermatology).