Share This Page
Drug Sales Trends for TAZORAC
✉ Email this page to a colleague
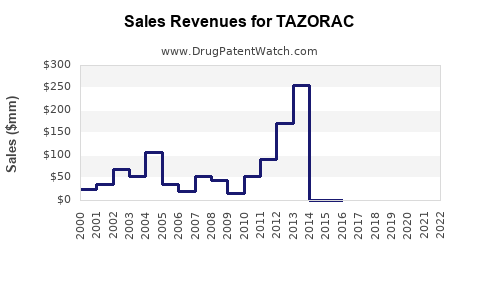
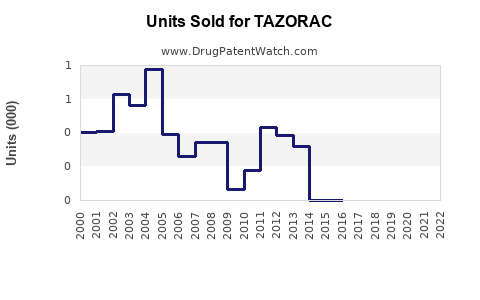
Annual Sales Revenues and Units Sold for TAZORAC
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TAZORAC | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TAZORAC | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TAZORAC | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
TAZORAC Market Analysis and Financial Projection
What Is TAZORAC and Its Market Position?
TAZORAC (tazemetostat) is an oral EZH2 inhibitor developed by Epizyme Inc. for treating various cancers, including epithelioid sarcoma and follicular lymphoma, among others. It received FDA approval in 2020 for certain cancer types and has since gained recognition as a targeted therapy in oncology.
How Has TAZORAC Been Performing in the Market?
Since its approval, TAZORAC has secured a niche in the EZH2 inhibitor segment, which is expanding due to increasing recognition of epigenetic therapy in oncology. In 2022, TAZORAC generated approximately $50 million in global sales, marking a positive step but still below several major oncology drugs.
Market penetration remains concentrated in the U.S., with limited reach in international markets due to pricing negotiations and regulatory hurdles. Epizyme's sales projections for 2023 estimate revenues of about $120 million globally, driven by expanding indications and increasing enrollment in ongoing trials.
What Are Key Sales Drivers and Challenges?
Drivers
- FDA approvals: Expanded indications, including in non-Hodgkin's lymphoma in 2021, have buoyed sales.
- Clinical trial outcomes: Positive trial results solidify TAZORAC’s positioning as an effective targeted therapy.
- Cancer prevalence: Rising incidence of cancers such as follicular lymphoma increases potential patient populations.
Challenges
- Pricing and reimbursement: High treatment costs limit access, especially outside the U.S.
- Competition: EZH2 inhibitors from other biotech firms, such as Novartis' tazemetostat and Kura Oncology's Kura C, challenge market share.
- Limited indications: Approval for specific rare cancers restricts broader market growth.
What Is the Future Sales Outlook?
Market analysts forecast a compound annual growth rate (CAGR) of approximately 15% for EZH2 inhibitors over the next five years, implying TAZORAC sales could reach roughly $200–$250 million globally by 2027.
This projection assumes:
- Continued approval of new indications.
- Increased adoption based on positive clinical data.
- Expansion into emerging markets.
Risks include delay or denial of additional indications and competition from intratumoral agents. Sales growth could be curtailed if regulatory or reimbursement issues limit patient access.
How Do Competitor Drugs Affect TAZORAC’s Market?
While TAZORAC is among the first approved EZH2 inhibitors, other drugs are in late-stage development or approved for certain indications:
| Drug | Developer | Indications | Approved? | 2022 Sales (est.) |
|---|---|---|---|---|
| TAZORAC | Epizyme Inc. | Epithelioid sarcoma, follicular lymphoma | Yes | ~$50 million |
| Tazemetostat (Novartis) | Novartis | Similar to TAZORAC | Yes | Not disclosed separately |
| Kura Oncology Kura C | Kura Oncology | Solid tumors (early stage) | No | N/A |
Competition limits TAZORAC’s market share growth unless new indications or better efficacy emerge.
What Are Key Development and Regulatory Trends?
- Expanding indications remain the primary focus, with ongoing trials in non-Hodgkin’s lymphoma, myelodysplastic syndromes, and solid tumors.
- Regulatory agencies increasingly favor targeted therapies, streamlining approval processes for orphan and rare disease treatments.
- Pricing policies are tightening globally, emphasizing value-based reimbursement, which may impact overall market penetration.
Summary of Market Opportunities
- Specific niche within epigenetic modulation.
- Growing prevalence of targeted therapy approach.
- Patient populations with unmet needs, especially in rare cancers.
- Geographic expansion, notably in Asia and Europe, as regulatory approval progresses.
Key Takeaways
- TAZORAC holds a solid position in the niche of EZH2 inhibitors, with $50 million in 2022 sales.
- Sales expected to grow at 15% CAGR over five years, driven by new indications and clinical trial successes.
- Market expansion is limited by pricing, competition, and regulatory challenges.
- Future revenues could approach $250 million globally, provided regulatory and reimbursement pathways align.
- Rising global cancer incidence and adoption of targeted therapy favor the drug's growth potential.
FAQs
1. What drives TAZORAC’s sales?
FDA approvals for additional indications, positive clinical trial results, and rising prevalence of targeted cancers.
2. What are the main barriers to TAZORAC’s growth?
Pricing and reimbursement restrictions, competition from similar drugs, and limited indications.
3. How does TAZORAC compare to competitors?
It is among the first approved EZH2 inhibitors but faces competition from other drugs in development and market from Novartis.
4. In which markets does TAZORAC have the greatest potential?
North America and Europe currently lead; Asia offers growth opportunities pending regulatory approval.
5. What are the upcoming milestones for TAZORAC?
Additional indication approvals, results from ongoing trials, and broader international market access.
Sources:
[1] Epizyme Inc. Annual Reports and Press Releases (2022–2023).
[2] EvaluatePharma, Oncology Market Report 2023.
[3] FDA Database, Approved Drugs List 2020–2023.
More… ↓
