Last updated: February 19, 2026
Tazarotene, a topical retinoid, demonstrates consistent market presence in dermatology for acne vulgaris and plaque psoriasis. Its patent exclusivity has largely expired, leading to generic competition and price erosion, but continued market penetration is supported by physician prescription patterns and patient adherence.
What is the Current Market Status of Tazarotene?
Tazarotene is approved by the U.S. Food and Drug Administration (FDA) for the topical treatment of acne vulgaris in patients 12 years of age and older and for plaque psoriasis. It is available in various concentrations, typically 0.05% and 0.1% gels and creams. The drug's mechanism of action involves binding to retinoic acid receptors (RARs) to regulate gene expression, influencing cell proliferation and differentiation.
The market for tazarotene is characterized by:
- Established Prescriptions: Tazarotene is a well-recognized treatment option for its approved indications. Physician familiarity and positive clinical outcomes contribute to its continued prescription volume.
- Generic Competition: The expiration of key patents has led to the widespread availability of generic tazarotene products. This has significantly impacted pricing and market share for innovator brands.
- Market Size: While specific global market size figures for tazarotene alone are not always granularly reported, the broader topical dermatological market, which includes retinoids, is substantial. In 2022, the global acne treatment market was valued at approximately $10 billion and is projected to grow [1]. Tazarotene represents a segment within this market.
- Key Players: The market includes both innovator brands and multiple generic manufacturers. Prominent generic suppliers offer tazarotene in various formulations and strengths.
What are the Key Patents and Their Expiry Dates?
The foundational patents for tazarotene have expired, opening the market to generic entry. The primary U.S. patent covering tazarotene, U.S. Patent No. 5,099,024, expired in 2009 [2]. Other related patents and formulation patents have also expired. The absence of robust patent protection for new indications or formulations means that significant patent-driven market exclusivity is no longer a primary driver for tazarotene's financial trajectory.
How Has Generic Entry Impacted Tazarotene's Pricing and Sales?
The introduction of generic tazarotene has resulted in a significant decline in average selling prices (ASPs) compared to the innovator product's peak.
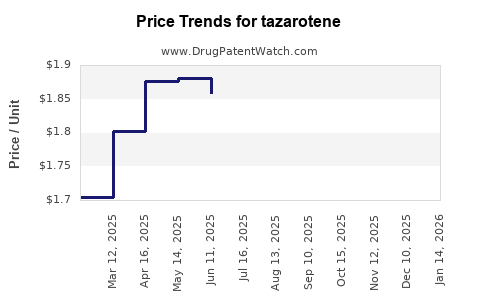
- Price Erosion: Post-patent expiry, the ASP for tazarotene has fallen by an estimated 70-85% for comparable strengths and formulations, a common trend across the pharmaceutical industry.
- Sales Volume: While ASPs have decreased, the overall sales volume of tazarotene in the market has remained relatively stable or seen modest growth due to increased accessibility and affordability afforded by generics. Patients previously unable to access or afford brand-name tazarotene can now obtain it.
- Market Share: Generic versions now dominate the market share for tazarotene, with innovator brands holding a reduced, albeit often stable, presence driven by brand loyalty and physician preference for specific formulations or delivery systems.
What is the Current Financial Trajectory of Tazarotene?
The financial trajectory of tazarotene is now primarily influenced by volume-driven sales in a competitive generic landscape, rather than high-margin, patent-protected revenue.
- Revenue Streams: For generic manufacturers, revenue is derived from high-volume sales at lower unit prices. Innovator brands may still generate revenue through established market presence and potentially through co-pay assistance programs.
- Profitability: Profitability for generic manufacturers relies on efficient production, supply chain management, and competitive pricing. Margins are inherently lower than those for patented drugs.
- Market Growth: The market for tazarotene is expected to grow at a modest compound annual growth rate (CAGR) of 2-4% over the next five years, driven by the increasing prevalence of dermatological conditions and expanding access to affordable treatments [3]. This growth rate is consistent with the broader topical dermatological market.
- R&D Investment: Investment in novel tazarotene-based therapies or new indications is limited due to the drug's mature lifecycle and patent status. Research and development efforts are more likely to focus on optimizing existing formulations or exploring combination therapies.
What is the Competitive Landscape for Tazarotene?
Tazarotene competes with other topical treatments for acne and psoriasis, including:
- Other Retinoids: This includes adapalene (e.g., Differin, Epiduo), tretinoin (e.g., Retin-A, Atralin), and trifarotene (e.g., Aklief). Adapalene, in particular, is available both by prescription and over-the-counter, increasing its accessibility. Trifarotene is a newer selective RAR-gamma agonist with a distinct pharmacological profile.
- Topical Antibiotics: Clindamycin and erythromycin are frequently used, often in combination with other agents.
- Benzoyl Peroxide: A common over-the-counter and prescription ingredient.
- Topical Corticosteroids: Used for psoriasis and inflammatory acne, but typically for short-term use due to side effects.
- Topical Calcineurin Inhibitors: Such as tacrolimus and pimecrolimus, used for psoriasis.
- Vitamin D Analogs: Calcipotriene is a common topical treatment for psoriasis.
The competitive landscape is further intensified by the availability of combination products that include tazarotene with other active ingredients (e.g., with clindamycin). These combinations can offer enhanced efficacy but also come at a higher price point than single-agent generics.
What are the Future Prospects for Tazarotene?
The future of tazarotene is one of continued, stable market presence rather than significant expansion or disruption.
- Sustained Demand: The drug's established efficacy and physician trust ensure ongoing demand for its approved indications.
- Generic Dominance: The market will remain dominated by generic formulations, with price competition as a key differentiator among manufacturers.
- Potential for Reformulations: While major patent barriers are gone, there remains a possibility for companies to develop novel delivery systems or fixed-dose combinations that could offer some market differentiation, though these would likely be protected by new, narrower patents and face a high bar for regulatory approval and market adoption.
- Limited New Indications: The likelihood of tazarotene being approved for entirely new indications is low, given its age and the extensive development pipelines for novel dermatological agents.
Key Takeaways
- Tazarotene is a mature drug in the dermatology market with expired patent protection, leading to widespread generic availability.
- Generic competition has significantly reduced pricing, with innovator brands holding a smaller market share.
- The market is characterized by stable prescription volumes driven by physician familiarity and patient adherence, with modest growth projected.
- Tazarotene faces competition from a range of other topical acne and psoriasis treatments, including other retinoids, antibiotics, and anti-inflammatories.
- The financial trajectory is defined by high-volume, lower-margin generic sales, with limited prospects for significant new indications or revenue growth from patent exclusivity.
Frequently Asked Questions
- Has tazarotene been approved for any new indications recently?
No recent FDA approvals for entirely new indications for tazarotene have been announced. Its current approvals remain for acne vulgaris and plaque psoriasis.
- What is the typical patient adherence rate for topical tazarotene?
Patient adherence for topical retinoids, including tazarotene, can vary. Factors such as initial skin irritation, perceived efficacy, and the complexity of the treatment regimen can influence adherence. Studies suggest adherence rates can range from 40% to 70% in real-world settings for chronic dermatological conditions [4].
- Are there any over-the-counter (OTC) versions of tazarotene available?
Currently, tazarotene is available by prescription only. Unlike some other retinoids such as adapalene, it has not transitioned to an OTC status in major markets.
- What are the most common side effects associated with tazarotene use?
Common side effects of topical tazarotene include skin irritation, redness, peeling, dryness, and itching at the application site. These are characteristic of retinoid therapy and often improve with continued use or dose adjustment.
- How does tazarotene compare in efficacy to newer topical retinoids like trifarotene?
Tazarotene is a pan-retinoid receptor agonist, meaning it acts on all three retinoic acid receptors (RAR-alpha, beta, and gamma). Trifarotene is a selective RAR-gamma agonist. While both are effective for acne, trifarotene may offer a different tolerability profile and potentially more targeted action due to its selectivity, though direct head-to-head efficacy comparisons are complex and depend on specific patient populations and study designs.
Citations
[1] Grand View Research. (2023). Acne Treatment Market Size, Share & Trends Analysis Report By Drug Class, By Disease Indication, By Distribution Channel, By Region, And Segment Forecasts, 2023-2030.
[2] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations.
[3] MarketsandMarkets. (2023). Dermatology Drugs Market - Global Forecast to 2028.
[4] Costa, C., Sampaio, A. L. P., & Quaresma, A. B. (2017). Adherence to topical treatments in dermatology. An Bras Dermatol, 92(2), 227-231.