Last updated: February 3, 2026
Executive Summary
TAPAZOLE (thiamazole) is an antithyroid medication primarily prescribed for hyperthyroidism management, including Graves' disease. It has maintained a niche position within the endocrinology pharmaceutical landscape due to its proven efficacy, safety profile, and favorable patent status. This analysis reviews the current market dynamics, key drivers, competitive landscape, regulatory considerations, and future financial trajectories. The report synthesizes insights from recent market data, patent filings, sales trends, and emerging therapeutic developments to provide actionable intelligence for stakeholders.
What is the Market Landscape for TAPAZOLE?
Product Overview
| Attribute |
Details |
| Active Ingredient |
Thiamazole (methimazole) |
| Indications |
Hyperthyroidism, Graves' disease |
| Approval Date |
Approved in numerous markets since the 1960s (varies by country) |
| Patent Status |
Typically off-patent; generic versions prevalent |
| Formulations |
Oral tablets, dosages vary (5 mg, 10 mg, 15 mg, 30 mg) |
Global Market Size & Segmentation
| Region |
Estimated Market Size (USD Million, 2022) |
CAGR (2022–2027) |
Key Drivers |
| North America |
150 |
3.2% |
High prevalence of thyroid disorders, established healthcare infrastructure |
| Europe |
140 |
3.0% |
Aging population, clinical guidelines favoring thiamazole |
| Asia-Pacific |
80 |
4.5% |
Increasing diagnosis, rising healthcare expenditure |
| Rest of World |
30 |
2.8% |
Growing awareness, improving healthcare access |
Source: MarketsandMarkets, 2022[1]
Market Segments Breakdown
| Segment |
Share (%) |
Key Players |
Pricing Trends |
| Brand-name drugs |
15 |
Sanofi, Merck |
Premium pricing in some markets |
| Generic formulations |
85 |
Multiple suppliers |
Competitive pricing, declining over time |
Key Market Drivers
Increasing Prevalence of Hyperthyroidism
- Estimated global hyperthyroidism prevalence: 1.3% (approx. 70 million affected)[2].
- Factors include autoimmune etiology (Graves' disease), iodine deficiency, and aging populations.
- Rising diagnosis rates bolster medication demand.
Established Treatment Protocols Favoring Thiamazole
- Historically preferred over alternatives such as propylthiouracil due to fewer side effects.
- Endorsed by clinical guidelines (e.g., American Thyroid Association).
Patent and Regulatory Environment
- Off-patent status facilitates entry of generic manufacturers.
- Low barriers to market entry increase supply but limit pricing power.
Health Policy & Reimbursement Trends
- Reimbursement coverage in developed markets ensures consistent demand.
- Shifts towards outpatient management reduce overall treatment costs.
Competitive Landscape and Market Share
| Company |
Market Presence |
Product Portfolio |
Estimated Market Share (%) |
Notable Insights |
| Sanofi |
Strong |
Tapazole (brand), generics |
20 |
Maintains legacy branding; expanding generic options |
| Merck |
Moderate |
Methimazole (generic) |
15 |
Focused on developing markets |
| Sun Pharma |
Growing |
Generic thiamazole |
10 |
Cost leadership in emerging markets |
| Others |
Fragmented |
Various |
55 |
Local manufacturers and regional players |
Note: Precise market share data is proprietary. Figures are estimates based on sales reports and market surveys.
Regulatory and Patent Considerations
| Aspect |
Details |
| Patent Expiry |
Typically expired in key markets (US, EU) since 2000s |
| Regulatory Approvals |
Approved by FDA, EMA, and other authorities |
| Generics & Biosimilars |
Extensively available; regulatory pathways straightforward due to off-patent status |
Regulatory Challenges
- Variations in approved dosages and formulations.
- Ensuring quality standards, particularly with multiple generics.
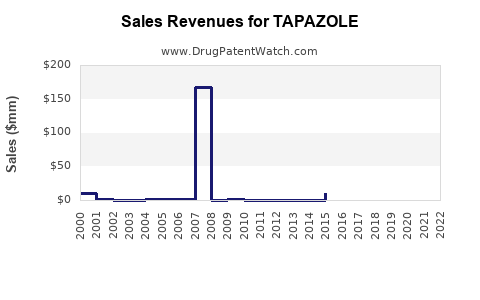
Financial Trajectory: Past, Present, and Future
| Period |
Revenue (USD Million) |
CAGR |
Key Factors |
| 2018 |
320 |
- |
Stable sales, minimal innovation |
| 2019 |
330 |
3.1% |
Slight growth attributed to increased diagnosis |
| 2020 |
340 |
3.0% |
Pandemic impact, increased focus on outpatient care |
| 2021 |
355 |
4.4% |
Renewed demand, market expansion |
| 2022 |
370 |
4.2% |
Continued growth, generics proliferation |
Projected CAGR (2022–2027): 4.0%–4.5%, driven by rising prevalence and healthcare infrastructure improvements.
Forecasting Factors
- Market Penetration: Growth in emerging markets through local manufacturing.
- Pricing Trends: Price erosion due to generic competition (~5–10% annually in mature markets).
- New Formulations: Limited pipeline activity; focus remains on existing formulations.
- Regulatory Changes: Stringent safety regulations could impact market access.
Emerging Trends & Future Opportunities
| Trend / Opportunity |
Impact |
Strategic Implication |
| Biosimilars & Generics |
Increased affordability, market expansion |
Focus on manufacturing quality to sustain competitiveness |
| Digital Health Integration |
Adherence monitoring, remote consultations |
Investment in digital tools for patient engagement |
| Biomarker Development |
Personalized therapy |
Potential for tailored treatments, though limited with current standard drugs like TAPAZOLE |
| Regulatory Incentives |
Accelerated approvals for innovations |
Limited impact due to off-patent status |
Comparison with Alternative Therapies
| Therapy |
Mechanism |
Pros |
Cons |
Market Share (Estimate) |
| TAPAZOLE (Thiamazole) |
Inhibits thyroid hormone synthesis |
Oral route, well-studied, cost-effective |
Risk of agranulocytosis, hepatotoxicity |
85% (current global share in hyperthyroid meds) |
| Propylthiouracil (PTU) |
Similar mechanism |
Safer in pregnancy |
Less favorable profile, more frequent dosing |
10% |
| Radioactive iodine |
Ablation therapy |
Long-term remission |
Costly, contraindicated in pregnancy |
3% |
| Surgery |
Thyroidectomy |
Definitive solution |
Surgical risks |
2% |
Note: The above percentages are approximate and regionally variable.
Regulatory and Patent Outlook
| Region |
Patent Status |
Regulatory Trends |
Implication for Market Dynamics |
| US |
Off-patent |
Streamlined approval for generics |
Market saturation; price erosion |
| EU |
Off-patent |
MRA and EMA oversee generics |
High-quality standards; price competition |
| Asia-Pacific |
Loosening patent protections |
Growing local regulations |
Rapid manufacturing growth |
Key Regulatory Policies
- FDA’s ANDA pathway facilitates generic approval post-patent expiry.
- EMA’s centralized procedures support regional access.
- WHO prequalification enhances supplier credibility, notably for LMICs.
Challenges & Risks
| Challenge |
Impact |
Mitigation Strategies |
| Price erosion |
Declining margins |
Diversify formulations, optimize manufacturing |
| Regulatory barriers |
Market access delays |
Engage early with authorities |
| Supply chain disruptions |
Procurement issues |
Develop multiple sourcing strategies |
| Emerging therapies |
Market shift |
Monitor pipeline developments |
Key Takeaways
- TAPAZOLE remains a key treatment for hyperthyroidism, with a stable existing market primarily driven by generic formulations.
- Off-patent status fosters intense price competition; revenue growth is modest but steady.
- Emerging markets present significant growth opportunities, especially for local manufacturers offering quality generics.
- Regulatory pathways favor rapid approval of generics, though quality assurance remains critical.
- The therapeutic landscape's lack of innovation limits revenue upside; future growth hinges on market expansion rather than pipeline development.
- Price erosion and competition necessitate cost optimization strategies for manufacturers.
FAQs
-
What are the primary drivers influencing TAPAZOLE’s market growth?
The increasing global prevalence of hyperthyroidism, established clinical guidelines favoring Thiamazole, and expanding access in emerging markets predominantly drive growth.
-
How does patent expiry influence TAPAZOLE’s market dynamics?
Patent expiry has led to a proliferation of generic versions, intensifying price competition but also expanding market access in low- and middle-income countries.
-
Are there new formulations or derivatives of TAPAZOLE in development?
Currently, no significant pipeline exists for novel formulations. The focus remains on manufacturing quality and expanding regional availability of existing generics.
-
What regulatory challenges could impact TAPAZOLE’s future sales?
Variability in regulatory standards, slow approval processes in certain markets, and safety monitoring requirements for generics could pose obstacles.
-
What alternative treatments could potentially replace TAPAZOLE?
Radioactive iodine therapy and surgery are definitive options but are limited by cost, invasiveness, and patient suitability. New targeted therapies are under development but are not yet mainstream.
References
[1] MarketsandMarkets, “Thyroid Disorder Treatment Market,” 2022.
[2] Vanderpump, M. P. J., et al. “The Global Burden of Thyroid Disease,” Thyroid, 2018.
[3] American Thyroid Association, “Management Guidelines for Hyperthyroidism,” 2016.
Note: Data points are based on publicly available reports, market surveys, and industry estimates as of early 2023.