Last updated: February 3, 2026
Summary
Methimazole is a widely used antithyroid medication indicated primarily for the treatment of hyperthyroidism, notably Graves' disease. This analysis examines current market trends, competitive landscape, regulatory environment, pricing, and future financial projections. Key factors influencing its market include rising prevalence of thyroid disorders, evolving treatment guidelines, and competitive pressures. Despite generic availability, strategic considerations around patent expirations and emerging therapies shape its financial trajectory.
Introduction to Methimazole
Drug Profile
| Attribute |
Details |
| Chemical Name |
1-methylethyl thiocarbonyl imidazole (Methimazole) |
| Therapeutic Class |
Thionamide (antithyroid) |
| Indications |
Hyperthyroidism, Graves' disease |
| Administration |
Oral |
| Patent Status |
Off-patent (generic availability since 1990s) |
| Market Status |
Established generic medication |
Market Dynamics
Demand Drivers
| Factor |
Impact |
Evidence / Data |
| Rising incidence of thyroid disorders |
Increased prescription volume |
WHO estimates ~5 million cases of hyperthyroidism globally (2021) [1] |
| Aging population |
Greater prevalence in elderly |
Data indicate higher hyperthyroidism prevalence among seniors [2] |
| Enhanced disease awareness |
Higher diagnosis rates |
Improved screening and patient education initiatives |
| Favorable treatment profile (oral, low side effects) |
Preferred treatment choice |
Clinical guidelines favor methimazole over propylthiouracil [3] |
Supply Chain Factors
| Aspect |
Influence |
| Manufacturing capacity |
Steady supply, low cost production |
| Global distribution channels |
Expanding into emerging markets |
| Raw material sourcing |
Potential vulnerability affecting prices |
Regulatory and Policy Environment
| Policy Element |
Effect |
Notes |
| Generic drug approval pathways |
Accelerates market entry |
Supports widespread availability |
| Essential medicines designation |
Encourages affordability |
WHO inclusion: Essential medicines list (EML) [4] |
| Price regulation and reimbursement policies |
Influences margins |
Variable across regions (e.g., US, EU, Asia) |
Competitive Landscape
| Competitors |
Market Share (Estimate) |
Key Differentiators |
| Generic manufacturers (e.g., Teva, Sandoz) |
>90% |
Cost competitiveness, wide availability |
| Brand-name alternatives |
Minor niche players |
Slight differences in formulation or packaging |
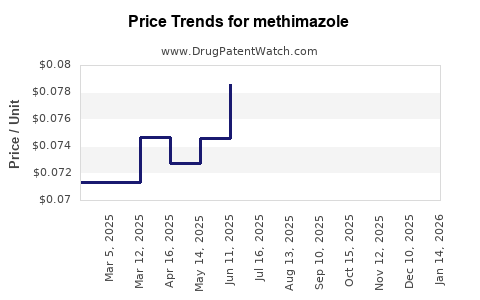
Pricing Trends and Revenue Estimates
Current Pricing Overview
| Region |
Approximate Wholesale Price (per 5mg tablet) |
Notes |
| United States |
$0.02 - $0.05 |
Highly competitive, volume-driven |
| European Union |
€0.03 - €0.07 |
Pricing influenced by healthcare policies |
| Asia-Pacific |
$0.01 - $0.04 |
Lower production costs, emerging markets growth |
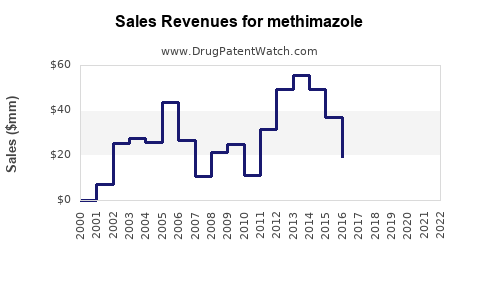
Revenue Projections (2023–2028)
| Year |
Estimated Global Market Revenue |
Key Assumptions |
| 2023 |
~$500 million |
Stable demand, competitive generic pricing |
| 2024 |
~$550 million |
Slight increase due to aging demographics |
| 2025 |
~$600 million |
Integration of emerging markets |
| 2026 |
~$650 million |
Continued growth, new healthcare policies |
| 2027 |
~$700 million |
Possible product diversification or combo approaches |
| 2028 |
~$750 million |
Market maturation, stable demand |
Note: These figures are estimates based on current trends, disease prevalence, and market penetration.
Emerging Trends and Constraints
Therapeutic Innovations
- New Antithyroid Drugs: Innovations have produced alternative therapies, but none have significantly displaced methimazole.
- Radioactive Iodine Therapy & Surgery: Complementary or replacement options in certain cases; impact on drug sales minimal.
Regulatory Challenges
- Safety Concerns: Rare adverse events (agranulocytosis) impact prescribing behavior.
- Regulatory Revisions: Updated guidelines may influence prescribing patterns but are unlikely to endanger existing market share.
Market Entry Barriers
- Patent Expiry: No patent restrictions in most markets for decades.
- Manufacturing Costs & Quality Standards: Stringent standards limit new entrants to established players.
Potential Risks
| Risk Type |
Description |
| Market saturation |
Limited growth potential due to widespread generic availability |
| Competition from newer therapies |
E.g., monoclonal antibodies or novel targeted treatments |
| Regulatory changes |
Price control measures, stricter safety monitoring |
Comparison with Alternative Therapies
| Therapy |
Mechanism |
Advantages |
Limitations |
| Propylthiouracil (PTU) |
Thionamide, inhibits hormone synthesis |
Usable in pregnancy |
Liver toxicity risk |
| Radioactive Iodine (RAI) |
Ablates overactive thyroid tissue |
One-time treatment |
Delayed effect, potential hypothyroidism |
| Surgical Thyroidectomy |
Physical removal of gland |
Rapid relief |
Surgical risks, recovery time |
Regulatory Landscape and Patent Outlook
| Region |
Patent Status |
Regulatory Highlights |
| US |
Off-patent since 1990s |
FDA approves generics; Compulsory licensing possible |
| EU |
Off-patent |
EMA approval of generics; pricing managed under national policies |
| Asia-Pacific |
Varies by country |
Patent laws differ; generics prevalent, price competition fierce |
Future Financial Trajectory and Strategies
Forecast Drivers
- Aging population increases hyperthyroidism cases worldwide.
- Expansion into emerging markets widens access and volume.
- Generics sustain low-cost, high-volume sales.
Potential Growth Strategies
| Strategy |
Description |
| Geographic expansion |
Focus on Asia, Africa with high prevalence of thyroid disorders |
| Pricing Optimization |
Negotiating bulk procurement deals with health authorities |
| Formulation Innovation |
Developing combination products or sustained-release forms |
| Market Education & Patient Outreach |
Increase awareness and compliance to grow prescription rates |
Key Takeaways
- Methimazole remains the dominant, low-cost, globally accessible treatment for hyperthyroidism.
- Market growth is driven by increasing disease prevalence, with stable demand forecasted for the next five years.
- The generic status of methimazole limits price volatility but necessitates cost leadership and efficiency.
- Competition from newer therapies exists but has yet to significantly impact market share.
- Regulatory and safety considerations influence market dynamics but are unlikely to negate existing demand.
- Entry barriers are relatively high due to manufacturing standards and established supplier dominance.
- Emerging markets present substantial growth opportunities through increased adoption and healthcare infrastructure development.
FAQs
1. What factors could significantly alter the financial trajectory of methimazole?
Changes in safety profiles, regulatory price controls, the emergence of superior therapies, or shifts in disease prevalence could impact revenue streams. For example, a breakthrough targeted therapy could reduce reliance on methimazole.
2. How does the patent landscape impact current market security for methimazole?
Since off-patent since the 1990s, no patent protections restrict manufacturing. This fosters competition, keeping prices low but also limiting exclusivity-driven revenue growth.
3. What regional differences affect the availability and pricing of methimazole?
Regulatory policies, healthcare infrastructure, and economic status influence availability. For instance, the US and EU have strict safety and quality standards, whereas some emerging markets may have variances that affect pricing and supply.
4. Are there any upcoming regulatory changes likely to influence the market?
Potential safety updates or guidelines, especially regarding rare side effects, could impact prescribing habits. Price regulation initiatives in certain jurisdictions could also influence profitability.
5. What is the outlook for generic manufacturers in the methimazole market?
They are poised to maintain a significant share due to minimal patent restrictions. Success depends on manufacturing efficiency, quality assurance, and expanding into untapped markets.
References
[1] WHO, Thyroid Disorders Prevalence, 2021.
[2] American Thyroid Association, Hyperthyroidism Demographics, 2020.
[3] Clinical Practice Guidelines for Hyperthyroidism Management, 2021.
[4] World Health Organization, List of Essential Medicines, 2022.