STEGLUJAN Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Steglujan, and when can generic versions of Steglujan launch?
Steglujan is a drug marketed by Msd Sub Merck and is included in one NDA. There are four patents protecting this drug.
This drug has one hundred and thirty-eight patent family members in fifty-two countries.
The generic ingredient in STEGLUJAN is ertugliflozin; sitagliptin phosphate. One supplier is listed for this compound. Additional details are available on the ertugliflozin; sitagliptin phosphate profile page.
DrugPatentWatch® Generic Entry Outlook for Steglujan
Steglujan was eligible for patent challenges on December 19, 2021.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be October 21, 2030. This may change due to patent challenges or generic licensing.
There have been twenty-nine patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
Summary for STEGLUJAN
| International Patents: | 138 |
| US Patents: | 4 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Formulation / Manufacturing: | see details |
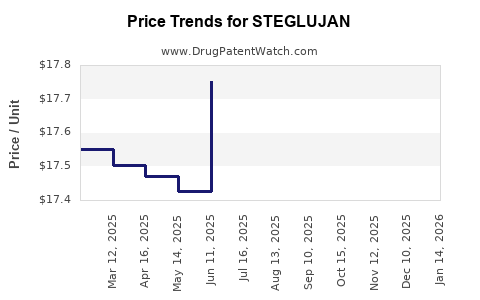
| Drug Prices: | Drug price information for STEGLUJAN |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for STEGLUJAN |
| What excipients (inactive ingredients) are in STEGLUJAN? | STEGLUJAN excipients list |
| DailyMed Link: | STEGLUJAN at DailyMed |
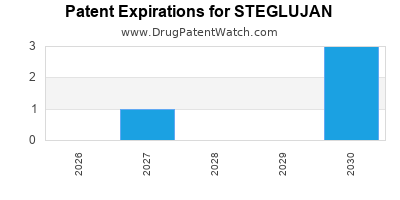
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for STEGLUJAN
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for STEGLUJAN
| Drug Class | Dipeptidyl Peptidase 4 Inhibitor |
| Mechanism of Action | Dipeptidyl Peptidase 4 Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for STEGLUJAN
US Patents and Regulatory Information for STEGLUJAN
STEGLUJAN is protected by four US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of STEGLUJAN is ⤷ Sign Up.
This potential generic entry date is based on patent ⤷ Sign Up.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting STEGLUJAN
Phosphoric acid salt of a dipeptidyl peptidase-IV inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Dioxa-bicyclo[3.2.1]octane-2,3,4-triol derivatives
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: AN ADJUNCT TO DIET AND EXERCISE TO IMPROVE GLYCEMIC CONTROL IN ADULTS WITH TYPE 2 DIABETES
Dioxa-bicyclo[3.2.1]octane-2,3,4-triol derivatives
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Dioxa-bicyclo[3.2.1]octane-2,3,4-triol derivatives
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: AN ADJUNCT TO DIET AND EXERCISE TO IMPROVE GLYCEMIC CONTROL IN ADULTS WITH TYPE 2 DIABETES
FDA Regulatory Exclusivity protecting STEGLUJAN
REVISION TO THE LABELING TO INCLUDE RESULTS FROM CLINICAL STUDY, MK-8835-004/B1521021, VERTIS CV
Exclusivity Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Msd Sub Merck | STEGLUJAN | ertugliflozin; sitagliptin phosphate | TABLET;ORAL | 209805-001 | Dec 19, 2017 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Msd Sub Merck | STEGLUJAN | ertugliflozin; sitagliptin phosphate | TABLET;ORAL | 209805-002 | Dec 19, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Msd Sub Merck | STEGLUJAN | ertugliflozin; sitagliptin phosphate | TABLET;ORAL | 209805-001 | Dec 19, 2017 | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for STEGLUJAN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Msd Sub Merck | STEGLUJAN | ertugliflozin; sitagliptin phosphate | TABLET;ORAL | 209805-002 | Dec 19, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| Msd Sub Merck | STEGLUJAN | ertugliflozin; sitagliptin phosphate | TABLET;ORAL | 209805-001 | Dec 19, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| Msd Sub Merck | STEGLUJAN | ertugliflozin; sitagliptin phosphate | TABLET;ORAL | 209805-001 | Dec 19, 2017 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for STEGLUJAN
When does loss-of-exclusivity occur for STEGLUJAN?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
African Regional IP Organization (ARIPO)
Patent: 99
Estimated Expiration: ⤷ Sign Up
Australia
Patent: 10310956
Estimated Expiration: ⤷ Sign Up
Canada
Patent: 77857
Estimated Expiration: ⤷ Sign Up
Costa Rica
Patent: 120289
Estimated Expiration: ⤷ Sign Up
Croatia
Patent: 0150107
Estimated Expiration: ⤷ Sign Up
Cyprus
Patent: 15949
Estimated Expiration: ⤷ Sign Up
Denmark
Patent: 96583
Estimated Expiration: ⤷ Sign Up
Ecuador
Patent: 12011946
Estimated Expiration: ⤷ Sign Up
Eurasian Patent Organization
Patent: 1983
Estimated Expiration: ⤷ Sign Up
Patent: 1290267
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 96583
Estimated Expiration: ⤷ Sign Up
Georgia, Republic of
Patent: 0146104
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 96156
Estimated Expiration: ⤷ Sign Up
Patent: 13509393
Estimated Expiration: ⤷ Sign Up
Montenegro
Patent: 016
Estimated Expiration: ⤷ Sign Up
New Zealand
Patent: 9945
Estimated Expiration: ⤷ Sign Up
Poland
Patent: 96583
Estimated Expiration: ⤷ Sign Up
Portugal
Patent: 96583
Estimated Expiration: ⤷ Sign Up
Serbia
Patent: 827
Estimated Expiration: ⤷ Sign Up
Slovenia
Patent: 96583
Estimated Expiration: ⤷ Sign Up
South Africa
Patent: 1203486
Estimated Expiration: ⤷ Sign Up
South Korea
Patent: 1426180
Estimated Expiration: ⤷ Sign Up
Patent: 120093321
Estimated Expiration: ⤷ Sign Up
Spain
Patent: 27179
Estimated Expiration: ⤷ Sign Up
Ukraine
Patent: 3416
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering STEGLUJAN around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Slovenia | 1654263 | ⤷ Sign Up | |
| Iceland | 2964 | ⤷ Sign Up | |
| Hungary | 0401104 | ⤷ Sign Up | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for STEGLUJAN
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1412357 | C200700039 | Spain | ⤷ Sign Up | PRODUCT NAME: SITAGLIPTINA; NATIONAL AUTHORISATION NUMBER: EU/1/07/383/001-018; DATE OF AUTHORISATION: 20070321; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/07/383/001-018; DATE OF FIRST AUTHORISATION IN EEA: 20070321 |
| 2334687 | PA2018510,C2334687 | Lithuania | ⤷ Sign Up | PRODUCT NAME: ERTUGLIFLOZINAS, PASIRINKTINAI KAIP KRISTALINE FORMA, YPAC KAIP KO-KRISTALAS SU L-PIROGLUTAMO RUGSTIMI, IR YPAC KAIP ERTUGLIFLOZINO L-PIROGLUTAMO RUGSTIS; REGISTRATION NO/DATE: EU/1/18/1267 20180321 |
| 1412357 | C300357 | Netherlands | ⤷ Sign Up | PRODUCT NAME: SITAGLIPTINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVA ARDBAAR ZOUT, IN HET BIJZONDER HET MONOFOSFAAT, EN METFORMINE DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER HET HYDROCHLORIDE; REGISTRATION NO/DATE: EU/1/08/455/001-014 20080716 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


