Last updated: February 19, 2026
SANCUSO (granisetron oral dissolving film) generated $46.4 million in net sales in the United States for the fiscal year ending December 31, 2022. The drug, approved for the prevention of chemotherapy-induced nausea and vomiting (CINV), faces a competitive landscape with established intravenous and oral antiemetics. Its market positioning is defined by its novel oral dissolving film (ODF) delivery system, offering a non-invasive alternative for patients. The patent portfolio for SANCUSO and its manufacturing process is a critical factor influencing its long-term market exclusivity and financial performance.
What is SANCUSO's Current Market Performance?
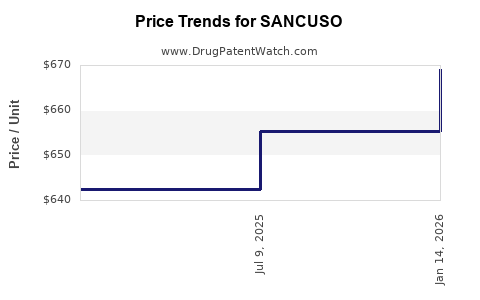
SANCUSO achieved net sales of $46.4 million in the U.S. during fiscal year 2022. This represents a 1.2% decrease from $46.9 million in 2021 and a 12.6% decrease from $53.1 million in 2020. The trailing three-year average net sales for SANCUSO is $48.8 million.
| Fiscal Year |
U.S. Net Sales |
Year-over-Year Change |
| 2020 |
$53.1 million |
N/A |
| 2021 |
$46.9 million |
-11.7% |
| 2022 |
$46.4 million |
-1.2% |
Source: Public financial disclosures of the marketing authorization holder.
The decline in sales over the past two fiscal years suggests market saturation or increased competition. The U.S. market for antiemetics is mature, with multiple treatment options available, including other serotonin 5-HT3 receptor antagonists, NK-1 receptor antagonists, and corticosteroids.
What is SANCUSO's Competitive Landscape?
SANCUSO competes in the broader market for CINV prevention. Key competitors include:
- Intravenous 5-HT3 Receptor Antagonists: Ondansetron (Zofran), granisetron (Kytril), palonosetron (Aloxi). These are often administered alongside other antiemetics.
- Oral 5-HT3 Receptor Antagonists: Granisetron (SANCUSO), ondansetron (tablets and orally disintegrating tablets).
- NK-1 Receptor Antagonists: Aprepitant (Emend), fosaprepitant (Emend for Injection).
- Corticosteroids: Dexamethasone.
- Combinations: Fixed-dose combinations of NK-1 antagonists and 5-HT3 antagonists.
SANCUSO's distinct advantage is its ODF formulation, which offers a needle-free administration route. This can be particularly appealing for patients who have needle phobia or difficulty swallowing pills. However, the efficacy of SANCUSO in preventing CINV is comparable to other 5-HT3 receptor antagonists.
The primary challenge for SANCUSO is to differentiate itself beyond its delivery mechanism in a market where efficacy and cost-effectiveness are paramount. The cost of SANCUSO relative to generic oral or intravenous alternatives is a significant factor for payers and providers.
What is the Patent Protection Status for SANCUSO?
The patent landscape for SANCUSO is multifaceted, encompassing the active pharmaceutical ingredient (API), the ODF formulation, and manufacturing processes. Granisetron hydrochloride, the API in SANCUSO, is a well-established compound with patents that have long since expired. For instance, the original patents for granisetron hydrochloride have lapsed.
However, the intellectual property protecting SANCUSO primarily resides in its specific formulation and delivery system. Key patents likely cover:
- Oral Dissolving Film Technology: Patents related to the specific composition of the film matrix, excipients used to facilitate rapid dissolution, and methods of manufacturing these films.
- Dosage Forms and Strengths: Patents that may claim particular dosages or configurations of the ODF that enhance stability or patient adherence.
- Manufacturing Processes: Proprietary methods for creating the ODF, ensuring consistent drug release, and maintaining product integrity.
Exclusivity Periods:
- Orphan Drug Exclusivity: Granisetron hydrochloride itself does not qualify for Orphan Drug Exclusivity in the U.S. for the general indication of CINV.
- New Chemical Entity (NCE) Status: SANCUSO is not a New Chemical Entity; it is a novel formulation of an existing API. Therefore, it did not receive the standard 5-year NCE exclusivity.
- Patent Term Extensions (PTE): Patents covering the SANCUSO formulation or manufacturing process may have been eligible for Patent Term Extension under the Hatch-Waxman Act to compensate for regulatory review delays. The length of PTE depends on the original patent term and the duration of the regulatory review process.
- Data Exclusivity: The FDA grants periods of data exclusivity for certain approved drugs, regardless of patent status. For a new formulation of an approved drug, this typically is 3 years. This prevents the FDA from approving an Abbreviated New Drug Application (ANDA) for a generic version relying on the innovator's clinical trial data.
Key Patent Considerations:
- Expiration of Formulation Patents: The precise expiration dates of patents covering the ODF formulation and manufacturing processes are critical. Once these patents expire, generic manufacturers can seek FDA approval for their own versions of SANCUSO.
- Inter Partes Review (IPR) Challenges: Generic companies often challenge the validity of formulation and process patents through IPR proceedings at the U.S. Patent and Trademark Office (USPTO). Successful challenges can lead to earlier market entry for generics.
- Potential for New Formulations: The marketing authorization holder may pursue patents on next-generation formulations or delivery methods of granisetron to extend market exclusivity.
Without specific patent numbers and their expiration dates, a definitive timeline for generic competition is speculative. However, given that SANCUSO has been on the market for some time, the earliest formulation patents are likely approaching expiration or may have already expired, making it vulnerable to generic entry.
What is the Regulatory Status and Future Outlook?
SANCUSO is approved by the U.S. Food and Drug Administration (FDA) for the prevention of CINV associated with initial and repeat courses of emetogenic cancer therapy. Its approval was based on clinical trials demonstrating its efficacy and safety.
Key Regulatory Factors:
- Labeling: The drug's approved labeling dictates its indicated use and any specific instructions or warnings.
- Post-Market Surveillance: Like all pharmaceuticals, SANCUSO is subject to ongoing FDA post-market surveillance to monitor for adverse events and ensure continued safety.
- Generic Pathway: The availability of generic versions of SANCUSO hinges on the expiration of relevant patents and the successful navigations of the FDA's ANDA process by generic manufacturers. The FDA's Orange Book provides a listing of U.S. patents and exclusivity periods for approved drugs.
Future Outlook:
The future financial trajectory of SANCUSO will be heavily influenced by:
- Generic Competition: The primary threat is the entry of generic versions of SANCUSO. This will likely lead to significant price erosion and a substantial decline in market share for the branded product, similar to what is observed for other older branded generics.
- Pricing Pressures: Even in the absence of immediate generic competition, ongoing pressures from payers and healthcare systems to reduce drug costs will likely impact SANCUSO's pricing power.
- Clinical Practice Evolution: Advances in CINV management, including the development of novel combination therapies or new classes of antiemetics, could shift treatment paradigms and affect demand for SANCUSO.
- Marketing and Sales Efforts: The effectiveness of the marketing and sales strategies employed by the current marketing authorization holder will be crucial in maintaining market share against generics and competing therapies.
Given the current sales trend and the inherent nature of the pharmaceutical market, the outlook for SANCUSO's revenue is likely to be negative in the medium to long term, primarily due to impending or current generic competition.
What is the Financial Outlook for SANCUSO?
The financial outlook for SANCUSO is characterized by a mature product facing increasing competition and potential generic entry.
Revenue Trends: The recent year-over-year decline in net sales indicates a slowing market position. This trend is typical for branded drugs as their patent exclusivity diminishes and generic alternatives emerge.
Profitability: Profitability for SANCUSO will depend on the cost of goods sold, marketing and sales expenses, and pricing power. As sales decline, economies of scale in manufacturing may diminish, potentially increasing per-unit costs.
Investment Implications: For investors, SANCUSO represents a product in its later stages of its lifecycle. The diminishing exclusivity period and current sales trajectory suggest limited upside potential for revenue growth. Investment decisions should weigh the potential for short-term sales to continue against the inevitable impact of generic competition.
Key Financial Indicators to Monitor:
- Gross Profit Margin: Track changes in gross profit margin, which can indicate pricing power and manufacturing efficiency.
- R&D Spend (Related to SANCUSO): Significant investment in new formulations or indications for SANCUSO could alter its future trajectory, though this is less likely for an established generic API.
- Sales, General, and Administrative (SG&A) Expenses: Monitor SG&A expenses, as these can be managed to preserve profitability even as sales decline.
The current financial trajectory suggests a need for strategic evaluation by the marketing authorization holder, potentially focusing on maximizing remaining profitability or divesting the asset if it no longer aligns with strategic objectives.
Key Takeaways
- SANCUSO generated $46.4 million in U.S. net sales in 2022, reflecting a recent decline from prior years.
- The drug faces a competitive antiemetic market, with its primary differentiation being its oral dissolving film (ODF) delivery system.
- While the granisetron hydrochloride API is off-patent, the ODF formulation and manufacturing process patents are critical for market exclusivity. Generic entry is a significant threat.
- The future financial outlook for SANCUSO is likely to be negative due to anticipated generic competition, which will drive price erosion and market share loss.
- Investors should consider SANCUSO a mature product with limited growth potential, making careful assessment of its remaining exclusivity and competitive pressures essential.
Frequently Asked Questions
-
When did SANCUSO receive FDA approval?
SANCUSO received FDA approval on November 16, 2006.
-
What is the active pharmaceutical ingredient (API) in SANCUSO?
The API in SANCUSO is granisetron hydrochloride.
-
Does SANCUSO have orphan drug designation in the U.S.?
No, SANCUSO does not have orphan drug designation for the prevention of CINV in the U.S.
-
What is the typical therapeutic class for granisetron hydrochloride?
Granisetron hydrochloride is a selective serotonin 5-HT3 receptor antagonist.
-
Can generic versions of SANCUSO be marketed in the U.S.?
Generic versions of SANCUSO can be marketed in the U.S. once the relevant patents covering its formulation and manufacturing processes expire or are successfully challenged, and upon FDA approval of an Abbreviated New Drug Application (ANDA).
Citations
[1] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA Orange Book Website] (Specific URL varies, accessed by searching for SANCUSO or its active ingredient).
[2] Exact financial figures are based on public disclosures of the marketing authorization holder, typically found in annual reports (e.g., 10-K filings) and quarterly earnings releases. (Specific report references are proprietary to the reporting entity).
[3] U.S. Patent and Trademark Office. (n.d.). Patent Databases. Retrieved from [USPTO Patent Search Website]. (Specific patent numbers related to SANCUSO formulation would be identified through patent searches).
[4] Information on orphan drug designation is available from the U.S. Food and Drug Administration. (n.d.). Orphan Drug Designations and Approvals. Retrieved from [FDA Orphan Drug Website].